Abstract
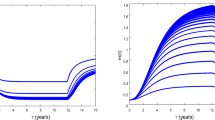
Cancer remains a major challenge for conventional treatments. This is due to the resistance mechanisms driven by cancer stem cells (CSCs) which sustain tumor growth. In this work, we investigate both analytically and computationally the effects of radioimmunotherapy (RIT), a cutting-edge technique that uses radiolabeled antibodies to precisely target and irradiate cancer cells. The work considers time delay modeling and the interactions between microRNAs and differentiated cancer cells (DCs). We evaluate the effects of extrapolated dose rates from four important radionuclides including yttrium-90 (\(^{90}\textrm{Y}\)), lutetium-177 (\(^{177}\textrm{Lu}\)), iodine-131 (\(^{131}\textrm{I}\)) and actinium-225 (\(^{225}\textrm{Ac}\)) in the preventive treatment of cancer before recurrence. A sensitivity analysis of model parameters is also performed to assess the robustness of the predictions and to identify the most influential biological and physical variables. Using the linear-quadratic formalism, we compare their biological effective dose, surviving fraction, and tumor control probability. The results demonstrate that an extrapolated initial dose of 165 \(\mathrm {Gy.year^{-1}}\) leads to an eradication of CSCs using \(^{225}\textrm{Ac}\) and \(^{177}\textrm{Lu}\) within 1.4636 year and 1.5736 year, respectively. Similarly, DCs are eliminated with \(^{225}\textrm{Ac}\) and \(^{177}\textrm{Lu}\) over treatment durations of 0.9396 year and 1.0496 year, respectively. These results highlight the potent effects of \(^{225}\textrm{Ac}\) and \(^{177}\textrm{Lu}\) in targeting CSCs and DCs at this dose rate. Under these conditions, microRNAs act as tumor suppressors, thus preventing pro-tumorigenic effects. Exceeding the dose threshold (beyond 165 \(\mathrm {Gy.year^{-1}}\)) disrupts the therapeutic balance with an efficacy which decreases progressively. For the doses above 326 \(\mathrm {Gy.year^{-1}}\), the overproliferation of CSCs and DCs is observed with an oncogenic behavior of microRNAs. We further examine the role of tumor oxygenation in modulating RIT efficacy. The results reveal that enhancing oxygen availability significantly increases CSC radiosensitivity, which is otherwise reduced under hypoxic conditions. The results of this work provide insight in optimizing RIT protocols using radiolabeled agents with improved pharmacokinetics and biological half-lives.
Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this paper.
References
Bray, F. et al. Global cancer statistics 2022: Globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 74(3), 229–263 (2024).
Masoudi, R. & Gopalan, V. Cancer stem cells in cancer initiation and progression, Cancer Stem Cells: Basic Concept and Therapeutic Implications 119–133 (Springer Nature Singapore, 2023).
Clevers, H. The cancer stem cell: Premises, promises and challenges. Nat. Med. 17(3), 313–319 (2011).
Valent, P. Cancer stem cell definitions and terminology: The devil is the details. Nat. Rev. Cancer 12(11), 767–775 (2012).
Chang, J. C. Cancer stem cells: Role in tumor growth, recurrence, metastasis, and treatment resistance. Medicine 95(1S), S20–S25 (2016).
Ayob, A. Z. & Ramasamy, T. S. Cancer stem cells as key drivers of tumour progression. J. Biomed. Sci. 25, 1–18 (2018).
Liu, Q. et al. Cancer stem cells and their niche in cancer progression and therapy. Cancer Cell Int. 23(1), 305 (2023).
Fatma, H. & Siddique, H. R. Cancer cell plasticity, stem cell factors, and therapy resistance: How are they linked?. Cancer Metastasis Rev. 43(1), 423–440 (2024).
Mengistu, B. A. et al. Comprehensive review of drug resistance in mammalian cancer stem cells: Implications for cancer therapy. Cancer Cell Int. 24(1), 406 (2024).
Dos Santos, S. N. & Witney, T. H. Molecular imaging of cancer stem cells and their role in therapy resistance. J. Nucl. Med. 66(1), 14–19 (2025).
Howell, R. W., Goddu, S. M. & Rao, D. V. Proliferation and the advantage of longer-lived radionuclides in radioimmunotherapy. Medical Physics 25(1), 37–42 (1998).
Rao, D. V. & Howell, R. W. Time dose fractionation in radioimmunotherapy: implications for selecting radionuclides. J. Nucl. Med. 34(10), 1801–1810 (1993).
Ahsun, R. et al. Complications following radioembolization with yttrium-90 microspheres: a comprehensive literature review. Journal of Vascular and Interventional Radiology 20(9), 1121–1130 (2009).
Salem, R. & Thurston, K. G. Radioembolization with 90y microspheres: A state-of-the-art brachytherapy treatment for primary and secondary liver malignancies. Part 1: Technical and methodologic considerations. J. Vasc. Interv. Radiol. 17(8), 1251–1278 (2006).
Schlumberger, M. et al. Strategies of radioiodine ablation in patients with low-risk thyroid cancer. N. Engl. J. Med. 366(18), 1663–1673 (2012).
Strosberg, J. et al. Phase 3 trial of \(^{177}{\sf L}u\)-dotatate for midgut neuroendocrine tumors. N. Engl. J. Med. 376(2), 125–135 (2017).
Kim, S. J. et al. The efficacy of \(\phantom{0}^{177 }\)lu-labelled peptide receptor radionuclide therapy in patients with neuroendocrine tumours: a meta-analysis. Eur. J. Nucl. Med. Mol. Imaging 42, 1964–1970 (2015).
Kratochwil, C. et al. \(\phantom{0}^{225}\)ac-psma-617 for psma-targeted alpha-radiation therapy of metastatic castration-resistant prostate cancer. J. Nucl. Med. 57(12), 1941–1944 (2016).
Jandl, T. et al. Melanoma stem cells in experimental melanoma are killed by radioimmunotherapy. Nucl. Med. Biol. 40(2), 177–181 (2013).
Giri, S. et al. Initial insights into the interaction of antibodies radiolabeled with lutetium-177 and actinium-225 with tumor microenvironment in experimental humain and canine osteosarcoma. Nucl. Med. Biol. 134–135, 108917 (2024).
Macey, D. J. et al. Uptake of indium-111-labeled monoclonal antibody zme-018 as a function of tumor size in a patient with melanoma. Am. J. Physiol. Imag. 3(1), 1–6 (1988).
Order, S. E. et al. Phase i-ii study of radiolabeled antibody integrated in the treatment of primary hepatic malignancies. Int. J. Radiat. Oncol. Biol. Phys. 6(6), 703–710 (1980).
Iznaga-Escobar, N., Mishra, A. K. & Perez-Rodriguez, R. Factors affecting pharmacokinetics of monoclonal antibodies: a review article. Methods and findings in experimental and clinical pharmacology 26 (2), 123–127 (2004).
Warfvinge, C. F. et al. Relationship between absorbed dose and response in neuroendocrine tumors treated with [177lu] lu-dotatate. J. Nucl. Med. 65 (7), 1070–1075 (2024).
Srinivasan, A. & Mukherji, S. K. Tositumomab and iodine i 131 tositumomab (bexaar). Am. J. Neuroradiol. 32(4), 637–638 (2011).
Rousseau, J., Lau, J. & Bénard, F., Radiolabeled antibodies for cancer radioimmunotherapy. Nucl. Med. Immunol. Eds.; Springer: Cham, Switzerland, 297–345 (2022).
Sun, N., Wang, T. & Zhang, S. Radionuclide-labelled nanoparticles for cancer combination therapy: a review. J. Nanobiotechnology 22(1), 728 (2024).
Witzig, T. E. et al. Phase i/ii trial of idec-y2b8 radioimmunotherapy for treatment of relapsed or refractory cd20\(\phantom{0}^{+}\)b-cell non-hodgkin’s lymphoma. J. Clin. Oncol. 17(12), 3793–3803 (1999).
Chinn, P. et al. Antibody therapy of non-hodgkin’s b-cell lymphoma. Cancer Immunol. Immunother. 52, 257–280 (2003).
Li, W. et al. Microrna-34a: Potent tumor suppressor, cancer stem cell inhibitor, and potential anticancer therapeutic. Front. Cell Dev. Biol. 9, 640587 (2021).
Yamamura, S. et al. Microrna-34a modulates c-myc transcriptional complexes to suppress malignancy in human prostate cancer cells. PLoS One 7(1), e29722 (2012).
Liu, C. et al. The microrna mir-34a inhibits prostate cancer stem cells and metastasis by directly repressing cd44. Nat. Med. 17(2), 211–215 (2011).
Pang, S.-T. et al. Urinary microrna-21 for prostate cancer detection using a silver nanoparticle sensor: A promising diagnostic tool. Biosensors 14(12), 599 (2024).
Li, W. et al. Microrna-34a, prostate cancer stem cells, and therapeutic development. Cancers 14(18), 4538 (2022).
Li, W. et al. Developing folate-conjugated mir-34a therapeutic for prostate cancer: Challenges and promises. Int. J. Mol. Sci. 25(4), 2123 (2024).
Pérez-Aliacar, M. et al. Modelling glioblastoma resistance to temozolomide. a mathematical model to simulate cellular adaptation in vitro. Comput. Biol. Med. 180, 108866 (2024).
Baez, J. & Kuang, K. Mathematical models of androgen resistance in prostate cancer patients under intermittent androgen suppressiontherapy. Appl. Sci. 6, 352 (2016).
Konstorum, A. et al. Feedback regulation in a cancer stem cell model can cause allee effect. Bull. Math. Biol. 78(4), 754–785 (2016).
Olmeda, F. & Ben Amar, M. Clonal pattern dynamics in tumor: The concept of cancer stem cells. Sci. Rep. 9(1), 15607 (2019).
Mori, L. & Ben Amar, M. Stochasticity and drug effects in dynamical model for cancer stem cells. Cancers 15(677), 15607 (2023).
Essongo, F. E., Mvogo, A. & Ben-Bolie, G. H. Dynamics of a diffusive model for cancer stem cells with time delay in microrna-differentiated cancer cell interactions and radiotherapy effects. Sci. Rep. 14(1), 1–17 (2024).
Fowler, J. F. The linear quadratic formula and progress in fractionated radiotherapy. Br. J. Radiol. 62, 679–694 (1989).
Dale, R. G. The application of the linear-quadratic dose-effect equation to fractionated and protracted radiotherapy. Br. J. Radiol. 58(690), 515–528 (1985).
Brown, J. M. & Wilson, W. R. Exploiting tumor hypoxia in cancer treatment. Nat. Rev. Cancer 4(6), 437–447 (2004).
Vaupel, P. & Mayer, A. Hypoxia in cancer: Significance and impact on clinical outcome. Cancer Metastasis Rev. 26, 225–239 (2007).
Brizel, D. M. et al. Tumor oxygenation predicts for the likehood of distant metastases in human soft tissue sarcoma. Cancer Res. 56(5), 941–943 (1996).
Chan, N., Milosevic, M. & Bristow, R. G. Tumor hypoxia, DNA repair and prostate cancer progression: New targets and new therapies. Future Oncol. 3(3), 329–341 (2007).
Visvader, J. E. & Lindeman, G. J. Cancer stem cells in solid tumors: Accumulating evidence and unresolved questions. Nat. Rev. Cancer 16(3), 225–238 (2015).
Zhang, T. & Zang, H. Delay-induced turing instability in reaction-diffusion equations. Phys. Rev. E 90, 05208 (2014).
Wang, X. et al. Delay-induced patterns in a reaction-diffusion system on complex networks. New J. Phys. 23(7), 12 (2021).
Howell, R. W., Goddu, S. M. & Rao, D. V. Application of the linear-quadratic model to radioimmunotherapy: further support for the advantage of longer-lived radionuclides. J. Nucl. Med. 35(11), 1861–1869 (1994).
Bernhardt, P. et al. Dosimetric analysis of the short-ranged particle emitter \(\phantom{0}^{161}\)tb for radionuclide therapy of metastatic prostate cancer. Cancers 13(9), 2011 (2021).
Friedrich, T. et al. Systematic analysis of rbe and related quantities using a database of cell survival experiments with ion beam irradiation. J. Radiat. Res. 54(3), 494–514 (2013).
Williams, M. V. et al. A review of alpha/beta ratios for experimental tumors: implications for clinical studies of altered fractionation. Int. J. Radiat. Oncol. Biol. Phys. 11(1), 87–96 (1985).
Kuznetsov, M. et al. Mathematical modeling unveils optimization strategies for targeted radionuclide therapy of blood cancers. Cancer Res. Commun. 4(11), 2955–2967 (2024).
Alper, T. & Howard-Flanders, P. The role of oxygen in modifying the radiosensitivity of e. coli b.. Nature 178, 978–979 (1956).
Wouters, B. G. & Brown, J. M. Cells at intermediate oxygen levels can be more important than the hypoxic fraction in determining tumor response to fractionated radiotherapy. Radiat. Res. 147(5), 541–550 (1997).
Daşu, A., Toma-Daşu, I. & Karlsson, M. Theoretical simulation of tumour oxygenation and results from acute and chronic hypoxia. Phys. Med. Biol. 48(17), 2829–2842 (2003).
Chen, Z. et al. Hypoxic microenvironment in cancer: molecular mechanisms and therapeutic interventions. Signal Transduct. Target. Ther. 8(1), 70 (2023).
Tannock, I. F. Oxygen diffusion and distribution of cellular radiosensitivity in tumours. Br. J. Radiol. 45(535), 515–24 (1972).
Lea, D. E. & Catcheside, D. G. The mechanism of induction by radiation of chromosome aberrations in transcendentia. J. Genet. 44, 216–245 (1942).
Joiner, M. & Bentzen, S. M. Time-dose relationships: the linear quadratic approach. In Steel c. g.(ed.)basic clinical radiobiology 55–64 (Edward Arnold, 2002).
Chadwick, K. H. & Leenhouts, H. P. A molecular theory of cell survival. Phys. Med. Biol. 18, 78–87 (1973).
Howell, R. W., Goddu, S. M. & Rao, D. V. Design and performance characteristics of an experimental cs-137 irradiator to simulate internal radionuclide dose rate patterns. J. Nucl. Med. 38(5), 727–731 (1997).
Liu, A. et al. On the modeling of the tumor uptake to determine the time-dose-fractionation (tdf) effect in radioimmunotherapy. J. Nucl. Med. 35(9), 1561–1562 (1994).
Rao, D. V. & Howell, R. W. On the modeling of the tumor uptake to determine the time-dose-fractionation (tdf) effect in radioimmunotherapy-reply. J. Nucl. Med. 35(9), 1562–1564 (1994).
Petit, S. F. et al. Intra-voxel heterogeneity influences the dose prescription for dose-painting with radiotherapy: A modelling study. Phys. Med. Biol. 54(7), 2179–2196 (2009).
Spoormans, K. et al. A review on tumor control probability (tcp) and preclinal dosimetry in targeted radionuclide therapy (trt). Pharmaceutics 14(10), 2007 (2022).
Royce, T. J. et al. Tumor control probability and systematic review of the litterature of stereotactic body radiotherapy for prostate cancer, International Journal of Radiation Oncology. Biology. Physics 110(1), 227–236 (2021).
Tucker, S. L., Thames, H. D. & Taylor, J. M. G. How well is the probability of tumour cure after fractionated irradiation described by poisson statistics?. Radiat. Res. 124(3), 273–282 (1990).
Tomé, W. A. & Fowler, J. F. Selective boosting of tumour subvolumes. Int. J. Radiat. Oncol. Biol. Phys. 48(2), 593–599 (2000).
Bachman Jeff, W. & Hillen, T. Mathematical optimization of the combination of radiation and differentiation therapies for cancer. Front. Oncol. 3, 42398 (2013).
Dasu, A. Toma-Dasu, Will infraction repair have negative consequences on extreme hypofractionation in prostate radiation therapy?. Br. J. Radiol. 88, 0588 (2015).
E. J. Hall, Radiobiology for the radiobiologist, Lippincott Williams and Wilkins 5\(\phantom{0}^{th}\) edition (2000).
Carlson, D.J., Stewart, R. D. & Semenenko, V. A. Effects of oxygen on intrinsic radiation sensitivity: A test of the relationship between aerobic and hypoxic linear-quadratic (lq) model parameters.. Med. Phys. 33(9), 3105–3115 (2006).
Iikuni, S. et al. Radiotheranostics using a novel \(\phantom{0}^{225}\)ac-labeled radioligand with improved pharmacokinetics targeting prostate-specific membrane antigen. J. Med. Chem. 64(18), 13429–13438 (2021).
Abusalem, M. et al. Current status of radiolabeled monoclonal antibodies targeting psma for imaging and therapy. Cancers 15(18), 4537 (2023).
Pandit-Taskar, N. et al. Antibody mass escalation study in patients with castration-resistantprostate cancer using \(\phantom{0}^{111}\)in-j591: Lesion detectability and dosimetric projections for \(\phantom{0}^{90}\)y radioimmunotherapy. J. Nucl. Med. 49(7), 1066–1074 (2008).
Zechmann, C. M. et al. Radiation dosimetry and first therapy results with a (124)i/ (131)i-labeled small molecule (mip-1095) targeting psma for prostate cancer therapy. Eur. J. Nucl. Med. Mol. Imaging 41(7), 1280–1292 (2014).
Schuchardt, C. et al. Prostate-specific membrane antigen radioligand therapy using \(\phantom{0}^{177}\)lu-psma i & t and \(\phantom{0}^{177}lu\)-psma-617 in patients with metastatic castration-resistant prostate cancer: comparison of safety, biodistribution, and dosimetry. J. Nucl. Med. 63(8), 1199–1207 (2022).
James, M. K. et al. A single dose of \(\phantom{0}^{225}\rm A c\)-rps-074 induces a complete tumor response in an lncap xenograft model. J. Nucl. Med. 60(5), 649–655 (2019).
Oberle, H. J. & pesch, H. J. Numerical treatment of delay differential equations by hermite interpolation. Numer. Math. 37, 235–255 (1981).
Ismail, F. et al. Numerical treatment of delay differential equations by runge-kutta method using hermite interpolation. Matematika 18(2), 79–90 (2002).
Zaider, M. & Minerbo, G. N. Tumor control probability: a formulation applicable to any temporal protocol of dose delivery. Phys. Med. Biol. 45(2), 279 (2000).
Brahme, A. Dosimetric precision requirements in radiation therapy. Acta Radiologica: Oncology 23(5), 379–391 (1984).
Wedenberg, M., From cell survival to dose response: modeling biological effects in radiation therapy. Karolinska Institutet. Thesis. (2013).
Graham, K. & Unger, E. Overcoming tumor hypoxia as a barrier to radiotherapy, chemotherapy and immunotherapy in cancer treatment. Int. J. Nanomed. 13, 6049–6058 (2018).
Pouysségur, J., Dayan, F. & Mazure, N. M. Hypoxia signalling in cancer and approaches to enforce tumour regression. Nature 441, 437–443 (2006).
Pugh, C. W. & Ratcliffe, P. J. Regulation of angiogenesis by hypoxia: role of the hif system. Nat. Med. 9, 677–684 (2003).
Najafi, M. et al. Hypoxia in solid tumors: a key promoter of cancer stem cell (csc) resistance. J. Cancer Res. Clin. Oncol. 146(1), 19–31 (2020).
Dewhirst, M. W., Cao, Y. & Moeller, B. Cycling hypoxia and free radicals rgulate angiogenesis and radiotherapy response. Nat. Rev. Cancer 8(6), 425–437 (2008).
Braun, R. D. et al. Comparison of tumor and normal tissue oxygen tension measurements using oxylite or microelectrodes in rodents. Am. J. Physiol. Heart Circ. Physiol. 280(6), H2533–H2544 (2001).
O’Connor, J. P. B., Robinson, S. P. & Waterton, J. C. Imaging tumor hypoxia with oxygen-enhanced mri and bold mri. Br. J. Radiol. 92(1096), 20180642 (2019).
Milotti, E. et al. Pulsation-limited oxygen diffusion in the tumour microenvironment. Sci. Rep. 7, 39762 (2017).
Semenza, G. L. Hyoxia-inducible factors in physiology and medicine. Cell 148(3), 399–408 (2012).
Horsman, M. R. et al. Therapeutic modification of hypoxia. Clin. Oncol. 33(11), 492–509 (2021).
Evans, S. M. & Koch, C. J. Prognostic significance of tumor oxygenation in humans. Cancer Lett. 195(1), 1–16 (2003).
G. Herrera, F. et al. 50-Gy stereotactic body radiation therapy to the dominant intraprostatic nodule: Results from a phase 1a/b trial. Int. J. Radiat. Oncol. Biol. Phys. 103(2), 320–334 (2019).
Wang, Y. et al. Necroptosis regulates tumor repopulation after radiotherapy via rip1/rip3/mlkl/jnk/il8 pathway. J. Exp. Clin. Cancer Res. 38(1), 461 (2019).
Jiang, H. et al. Low-dose radiation does not induce proliferation in tumor cells in vitro and in vivo. Radiat. Res. 170(4), 477–487 (2008).
Pei, J. et al. Sublethal dose of irradiation enhances invasion of malignant glioma cells through p53-mmp 2 pathway in u87mg mouse brain tumor model. Radiat. Oncol. 10, 164 (2015).
van der Doelen, M. J. et al. Clinical experience with psma-actinium-225 (ac-225) radioligand therapy (rlt) in end-stage metastatic castration-resistant prostate cancer (mcrpc) patients. J. Clin. Oncol. 36(6), 344–344 (2018).
Saleh, R. et al. Accel:[ac-225]-psma-62 phase ia/ib/ii clinical trial to characterize efficacy, safety, tolerability, and dosimetry in oligometastatic hormone-sensitive and metastatic castration-resistant prostate cancer. J. Clin. Oncol. 43(5), TPS282–TPS282 (2025).
Dale, L. L. et al. Preclinical development of an actinium-225-labeled antibody radio-conjugate directed against cd45 for targeted conditioning and radioimmunotherapy. Biology of Blood and Marrow Transplantation 26(3), S160–S161 (2020).
Jurcic, J. et al. Phase i trial of alpha-particle immunotherapy with \(\phantom{0}^{225}\)ac-lintuzumab and low-dose cytarabine in patients age 60 or older with untreated acute myeloid leukemia. J. Nucl. Med. 58, 456–456 (2017).
Csismazia, A. Correction to: Eanm’24 abstract book congress oct 19–23, 2024. Eur. J. Nucl. Med. Mol. Imaging 52, 374 (2024).
Therapeutics, A. Ariceum therapeutics presents outstanding data on its first-in-class radiopharmaceutical drug 225ac-satoreotide at the european association of nuclear medicine 2024, European Association of Nuclear Medicine (EANM) Annual Conference (2024).
Zhang, Y. & Wang, X. Targeting the wnt/\(\beta\)-catenin signaling pathway in cancer. J. Hematol. Oncol. 13(1), 165 (2020).
Pećina-Šlaus, N. et al. Wnt signaling inhibitors and their promising role in tumor treatment. Int. J. Mol. Sci. 24(7), 6733 (2023).
Acknowledgements
The work has greatly benefited from rich comments of reviewers.
Funding
Not applicable
Author information
Authors and Affiliations
Contributions
A.M. initiated the Project. F.E.E. performed the analytical results. F.E.E. and A.M. wrote the main manuscript text and prepared figures. A.M. and G.H.B.-B. performed numerical analysis. G.H.B.-B. supervised the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mvogo, A., Essongo, F.E. & Hubert Ben-Bolie, G. Cancer treatment by radioimmunotherapy: insights from a dynamical model of cancer stem cells and hypoxia effects. Sci Rep (2026). https://doi.org/10.1038/s41598-026-47796-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-47796-w