Abstract
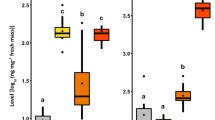
The spiraling whitefly, Aleurodicus dispersus Russell (Hemiptera: Aleyrodidae), is a highly polyphagous pest that poses a major threat to tropical and subtropical agriculture. Its rapid proliferation across multiple crop species is influenced by host plant quality, which significantly affects its development, reproduction, and survival. This study evaluated the influence of ten different host plants (cassava, cotton, guava, papaya, banana, mulberry, chilli (Capsicum annuum), Acalypha indica, eggplant, and Indian beech) on the developmental and reproductive parameters of A. dispersus under insect-proof screen house conditions. Developmental and reproductive traits varied significantly among host plants. The total developmental period (TDP) ranged from 24.2 ± 2.39 days on mulberry to 31.9 ± 3.35 days on banana (F = 6.06; df = 9, 90; P < 0.001). Egg hatchability and adult emergence were highest on cotton (98.7% and 98.2%, respectively) and lowest on Indian beech and eggplant. Fecundity was highest on cassava (28.5 ± 3.72 eggs per female) and lowest on eggplant (13.1 ± 1.79 eggs per female) (F = 27.20; df = 9, 90; P < 0.001). Female longevity was longest on cotton and cassava. A strong positive correlation was observed between egg hatchability and adult emergence (r = 0.85; P < 0.001), whereas a significant negative correlation was found between TDP and fecundity (r = − 0.78; P < 0.001). These results demonstrates host plant species significantly influence the biological performance of A. dispersus. Cassava, cotton, and guava supported comparatively faster development and higher reproductive output, indicating their suitability for population buildup. The findings provide useful insights for host-based pest management and highlight the potential role of less suitable host plants in reducing whitefly population growth within integrated pest management (IPM) programs.
Similar content being viewed by others
Data availability
Data is provided within the manuscript file.
Abbreviations
- A. dispersus :
-
Aleurodicus dispersus
- B. tabaci :
-
Bemisia tabaci
- CRD:
-
Completely randomized design
- IQR:
-
Interquartile range
- SD:
-
Standard deviation
- IBM:
-
International business machines
- NY:
-
New York
- USA:
-
United States of America
- F:
-
F value
- df:
-
Degrees of freedom
- P:
-
P value
- IPM:
-
Integrated pest management
- Ns:
-
Non-significant
- r:
-
Correlation coefficient
- Y:
-
Dependent variable
- X:
-
Independent variable
- TDP:
-
Total developmental period
- ANOVA:
-
Analysis of variance
- HSD:
-
Honest significant difference
- R2 :
-
Coefficient of determination
- η2 :
-
Eta-squared
- n:
-
Number
- TNAU:
-
Tamil Nadu Agricultural University
- PKM:
-
Periyakulam
- MR:
-
Mildew Resistant
- Co:
-
Coimbatore
- TRY:
-
Trichy
- ICAR-IARI:
-
Indian Council of Agricultural Research-Indian Agricultural Research Institute
- cm:
-
Centimeter
- h:
-
Hour
- χ2 :
-
Chi-squared
- R0 :
-
Net reproductive rate (offspring/individual)
- r :
-
Intrinsic rate of increase (day–1)
- λ :
-
Finite rate of increase (day–1)
References
Sundararaj, R. & David, B. V. On the genera Fippataleyrodes n. gen. and Taiwanaleyrodes Takahashi from India (Insecta, Homoptera, Sternorrhyncha: Aleyrodidae). Orient. Insects. 27, 233–270 (1992).
Mani, M., Ragunatha, R., Dinesh, M. S. & Krishnamoorthy, A. Spiralling whitefly Aleurodicus dispersus Russell in Hyderabad. Insect Environ. 7, 82 (2001).
Boopathi, T. New host plants, natural enemy complex and newly distributed potential areas of exotic spiralling whitefly (Hemiptera: Aleyrodidae) in India. Phytoparasitica. 49, 1–23. https://doi.org/10.1007/s12600-021-00907-2 (2021).
Byrne, D. N. & Bellows, T. S. Whitefly biology. Annu. Rev. Entomol. 36, 431–457 (1991).
Sathe, T. V. & Gangate, V. S. Host plants for a white fly Aleurodicus dispersus from Kolhapur region, India. Int. J. Recent. Sci. Res. 2, 28176–22820 (2015).
van Lenteren, J. C. & Noldus, L. P. J. J. Whitefly–plant relationships: Behavioral and ecological aspects. In Whiteflies: Their Bionomics, Pest Status and Management (ed. Gerling, D.) 47–89 (Intercept Ltd., 1990).
Awmack, C. S. & Leather, S. R. Host plant quality and fecundity in herbivorous insects. Annu. Rev. Entomol. 47, 817–844 (2002).
Oliveira, M. R. V., Henneberry, T. J. & Anderson, P. History, current status, and collaborative research projects for Bemisia tabaci. Crop Prot. 20, 709–723 (2001).
Musa, P. D. & Ren, S. X. Development and reproduction of Bemisia tabaci (Homoptera: Aleyrodidae) on three bean species. Insect Sci. 12, 25–30 (2005).
Kedar, S. C., Saini, R. K., Kumaranag, K. M. & Whitefly Bemisia tabaci (Gennadius) as influenced by host plants in Haryana. Indian J. Entomol. 80, 257–262 (2018).
Anjum, H. & Ahmed, S. I. An updated and consolidated review on Indian Aleyrodids fauna (Hemiptera: Aleyrodidae: Insecta) along with their host plant families and distributional records. Rec Zool. Surv. India. 119, 381–417 (2019).
Sampiano, K. F. S. & Aceres, L. V. The spiralling whitefly, Aleurodicus dispersus Russell (Hemiptera: Aleyrodidae): A Philippine perspective. Agrikultura CRI J. 2, 1–14 (2021).
Al-Zeyoud, F. & Sengonca, C. Development, longevity and fecundity of Bemisia tabaci (Genn.) (Homoptera: Aleyrodidae) on different host plants at two temperatures. Mitt Dtsch. Ges Allg Angew Entomol. 14, 373–376 (2004).
Manoharan, T., Jeyarani, S. & Arulraj, S. Host plant influence on the biological parameters of spiralling whitefly, Aleurodicus dispersus Russell. Indian J. Entomol. 80, 454–456 (2018).
Taggar, G. K. & Gill, R. S. Host plant resistance in Vigna sp. towards whitefly, Bemisia tabaci (Gennadius): A review. Entomol. Gen. 36, 1–24. https://doi.org/10.1127/entomologia/2016/0184 (2016).
Gamal, M. H. Comparative biological features of cotton whitefly, Bemisia tabaci (Hemiptera: Aleyrodidae) when reared on four host plants. Egypt. J. Plant. Prot. Res. Inst. 3, 804–815 (2020).
Sani, I. et al. A review of the biology and control of whitefly, Bemisia tabaci (Hemiptera: Aleyrodidae), with special reference to biological control using entomopathogenic fungi. Insects 11, 619. https://doi.org/10.3390/insects11090619 (2020).
Omondi, A. B., Obeng-Ofori, D., Kyerematen, R. A. & Danquah, E. Y. Host preference and suitability of some selected crops for two biotypes of Bemisia tabaci in Ghana. Entomol. Exp. Appl. 115, 393–400 (2005).
Shah, M. M. R., Zhang, S. & Liu, T. Whitefly, host plant and parasitoid: A review on their interactions. Asian J. Appl. Sci. Eng. 4, 48–61 (2015).
Huang, H., Zhao, H., Zhang, Y. M., Zhang, S. Z. & Liu, T. X. Influence of selected host plants on biology of castor whitefly, Trialeurodes ricini (Hemiptera: Aleyrodidae). J. Asia Pac. Entomol. 17, 745–751 (2014).
Manoharan, T. & Uthamasamy, S. Influence of host plants on the development and reproduction of spiralling whitefly, Aleurodicus dispersus Russell. Indian J. Entomol. 64, 421–426 (2002).
Srinivasa, M. V. Host plants of the spiralling whitefly, Aleurodicus dispersus. Pest Manag. Hortic. Ecosyst. 6, 79–105 (2000).
Inbar, M. & Gerling, D. Plant-mediated interactions between whiteflies, herbivores, and natural enemies. Annu. Rev. Entomol. 53, 431–448 (2008).
Walling, L. L. The myriad plant responses to herbivores. J. Plant. Growth Regul. 19, 195–216 (2000).
Hunter, M. D. A breath of fresh air: Beyond lab studies of plant volatiles and natural enemies. Agric. Entomol. 4, 81–86 (2002).
Chi, H. & Liu, H. Two new methods for the study of insect population ecology. Bull. Inst. Zool. Acad. Sin. 24, 225–240 (1985).
Chi, H. Life-table analysis incorporating both sexes and variable development rates among individuals. Environ. Entomol. 17, 26–34 (1988).
Acknowledgements
The authors are extremely thankful for providing facilities and support to the Head of the Department of Agricultural Entomology and the Director of the Centre for Plant Protection Studies, Tamil Nadu Agricultural University, Coimbatore, Tamil Nadu, India. The authors gratefully acknowledge the Director, ICAR–Indian Institute of Oilseeds Research, Rajendranagar, Hyderabad-500030, Telangana, India, for providing institutional support. Research was supported by the Indian Council of Agricultural Research, Department of Agricultural Research and Education, Government of India. During the preparation of this manuscript, the authors used ChatGPT (OpenAI) solely for language editing and improvement of clarity and readability. All scientific content, data interpretation and conclusions were generated and verified by the authors. The authors reviewed and edited all AI-assisted text and take full responsibility for the accuracy, originality and integrity of the manuscript.
Author information
Authors and Affiliations
Contributions
T.B. conceived, and designed research. T.B. performed all experiments. N.A., J.G.P. and T.B. analyzed data and wrote the manuscript. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Boopathi, T., Anusha, N. & Prasuna, J.G. Host plant influences on the developmental and reproductive biology of Aleurodicus dispersus and its implications for ecological pest management. Sci Rep (2026). https://doi.org/10.1038/s41598-026-47835-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-47835-6