Abstract
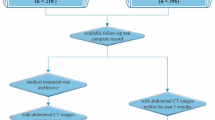
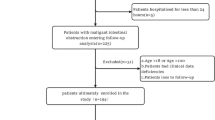
Despite advances in therapeutic strategies, including biologic and small-molecule agents, many Crohns disease (CD) patients in remission or with mild activity based on Crohns Disease Activity Index still require abdominal surgery, yet current predictive tools remain insufficient. This study developed and validated a practical prediction model using routine clinical data to assess long-term surgical risk in this population. This retrospective cohort study analyzed 615 incident CD patients diagnosed between 2016 and 2022. Using backward stepwise Cox regression, we developed a prediction model incorporating only readily available clinical parameters. Model performance was evaluated through Harrells C-index, time-dependent ROC analysis, and calibration curves, with bootstrap validation for internal verification. Five surgical predictors emerged: C-reactive protein (HR = 1.06, 95% CI: 1.03–1.10), fibrinogen (HR = 0.71, 95% CI: 0.59–0.85), albumin (HR: 0.67, 95% CI: 0.50–0.90), Montreal B classification (HR of B2 = 2.28, 95% CI: 1.30–4.00, HR of B3 = 4.13, 95% CI: 2.56–6.65), the use of advanced therapy (HR 0.63, 95% CI 0.42–0.92). The model demonstrated excellent predictive performance, with a Harrells C-index of 0.728 (95% CI: 0.677–0.779), and maintained time-dependent AUC values of 0.729–0.766 across 0.5–5 years. Bootstrap validation showed consistent performance (mean C-index: 0.733; time-dependent AUC: 0.731–0.773) with excellent calibration, confirming the models stability and clinical utility for long-term prediction. Risk stratification based on the cox model effectively categorized patients into low-, intermediate-, and high-risk groups (P < 0.001). Additionally, an online platform to enable clinical guidance: https://crohndisease.shinyapps.io/CDAI0-1/.The prediction model incorporating routinely available clinical variables to accurately predict abdominal surgery risk in CD patients in remission or with mild activity, supporting clinical decision-making.
Similar content being viewed by others
Data availability
The datasets utilized in the present study can be obtained from the corresponding author upon reasonable request.
Abbreviations
- ALB:
-
Albumin
- APTT:
-
Activated partial thromboplastin time
- BMI:
-
Body mass index
- CD:
-
Crohns disease
- CRP:
-
C-Reactive protein
- DD:
-
D-dimer
- ESR:
-
Erythrocyte sedimentation rate
- Fg:
-
Fibrinogen
- Hb:
-
Hemoglobin
- PT:
-
Prothrombin time
- TT:
-
Thrombin time
- WBC:
-
White blood cell count
References
Torres, J. et al. Crohns disease. Lancet 389(10080), 1741–1755 (2017).
Dolinger, M., Torres, J. & Vermeire, S. Crohns disease. Lancet 403(10432), 1177–1191 (2024).
Hracs, L. et al. Global evolution of inflammatory bowel disease across epidemiologic stages. Nature 642(8067), 458–466 (2025).
Solitano, V. et al. Shaping the future of inflammatory bowel disease: A global research agenda for better management and public health response. Nat. Rev. Gastroenterol. Hepatol. 22(6), 438–452 (2025).
Kaplan, G. G. The global burden of IBD: From 2015 to 2025. Nat. Rev. Gastroenterol. Hepatol. 12(12), 720–727 (2015).
Nagayama, M., Gogokhia, L. & Longman, R. S. Precision microbiota therapy for IBD: Premise and promise. Gut. Microbes. 17(1), 2489067 (2025).
Petagna, L. et al. Pathophysiology of Crohns disease inflammation and recurrence. Biol. Direct. 15(1), 23 (2020).
Rieder, F. et al. Results of the 4th scientific workshop of the ECCO (Group II): Markers of intestinal fibrosis in inflammatory bowel disease. J. Crohns Colitis 8(10), 1166–1178 (2014).
Bernstein, C. N. et al. Hospitalisations and surgery in Crohns disease. Gut 61(4), 622–629 (2012).
Du, J. et al. A novel computed tomography enterography radiomics combining intestinal and creeping fat features could predict surgery risk in patients with Crohns disease. Eur. J. Gastroenterol. Hepatol. 36(12), 1384–1392 (2024).
Toh, J. W. et al. Indications and surgical options for small bowel, large bowel and perianal Crohns disease. World J. Gastroenterol. 22(40), 8892–8904 (2016).
Patel, K. V. et al. Patient optimization for surgery relating to Crohns disease. Nat. Rev. Gastroenterol. Hepatol. 13(12), 707–719 (2016).
Tsai, L. et al. Contemporary risk of surgery in patients with Ulcerative colitis and Crohns disease: A meta-analysis of population-based cohorts. Clin. Gastroenterol. Hepatol. 19(10), 2031-2045. (2021).
Alós, R. & Hinojosa, J. Timing of surgery in Crohns disease: A key issue in the management. World. J. Gastroenterol. 14(36), 5532–5539 (2008).
Lee, J. S. et al. The importance of the Crohns Disease Activity Index in surgery for small bowel Crohns disease. J. Visc. Surg. 153(5), 339–345 (2016).
Meima-van Praag, E. M. et al. Surgical management of Crohns disease: A state of the art review. Int. J. Colorectal Dis. 36(6), 1133–1145 (2021).
Chirra, P. et al. Integrating Radiomics with clinicoradiological scoring can predict high-risk patients who need surgery in Crohns disease: A pilot study. Inflamm. Bowel. Dis. 29(3), 349–358 (2023).
Dong, Y. et al. A novel surgical predictive model for Chinese Crohns disease patients. Med. (Baltimore) 98(46), e17510 (2019).
Liu, R. et al. Development and validation of multiparametric models based on computed tomography enterography to determine endoscopic activity and surgical risk in patients with Crohns disease: A multi-center study. Heliyon. 9(9), e19942 (2023).
Madsen, G. R. et al. Intestinal Ultrasound Findings and Their Prognostic Value in Early Crohns Disease: A Copenhagen IBD Cohort Study. Clin. Gastroenterol. Hepatol. 23(8), 1398-1407. (2025).
Ruiqing, L. et al. A novel radiomics model integrating luminal and mesenteric features to predict mucosal activity and surgery risk in Crohns disease patients: A multicenter study. Acad. Radiol. 30(Suppl 1), S207-s219 (2023).
Yang, L. et al. Quantitative analysis of adipose tissue for predicting Crohns disease postoperative endoscopic recurrence and anastomotic ulcer. Int. J. Colorectal Dis. 38(1), 170 (2023).
Buisson, A. et al. Bowel wall healing assessed using magnetic resonance imaging predicts sustained clinical remission and decreased risk of surgery in Crohns disease. J. Gastroenterol. 54(4), 312–320 (2019).
Camp, R. L., Dolled-Filhart, M. & Rimm, D. L. X-tile: A new bio-informatics tool for biomarker assessment and outcome-based cut-point optimization. Clin. Cancer. Res. 10(21), 7252–7259 (2004).
Yang, L. et al. Epidemiological investigation of different types of Crohns disease patients undergoing first abdominal surgery in the era of biological agents: a population-based cohort study. Int. J. Colorectal. Dis. 39(1), 150 (2024).
Cerqueira, R. M. & Lago, P. M. Clinical factors predictive of Crohns disease complications and surgery. Eur. J. Gastroenterol. Hepatol. 25(2), 129–134 (2013).
Silverberg, M. S. et al. Toward an integrated clinical, molecular and serological classification of inflammatory bowel disease: Report of a Working Party of the 2005 Montreal World Congress of Gastroenterology. Can. J. Gastroenterol. 19(Suppl A), 5a–36a (2005).
Satsangi, J. et al. The Montreal classification of inflammatory bowel disease: controversies, consensus, and implications. Gut 55(6), 749–753 (2006).
Zhou, H. H. et al. C-reactive protein: Structure, function, regulation, and role in clinical diseases. Front. Immunol. 15, 1425168 (2024).
Yanai, H. et al. The Crohns disease exclusion diet for induction and maintenance of remission in adults with mild-to-moderate Crohns disease (CDED-AD): An open-label, pilot, randomised trial. Lancet. Gastroenterol. Hepatol. 7(1), 49–59 (2022).
Mosli, M. H. et al. C-Reactive Protein, Fecal Calprotectin, and Stool Lactoferrin for Detection of Endoscopic Activity in Symptomatic Inflammatory Bowel Disease Patients A Systematic Review and Meta-Analysis. Am. J. Gastroenterol. 110(6), 802–819 https://journals.lww.com/ajg/abstract/2015/06000/c_reactive_protein,_fecal_calprotectin,_and_stool.10.asp (2015).
Chen, X. F. et al. Predictive value of fibrinogen in identifying inflammatory bowel disease in active stage. BMC Gastroenterol. 21(1), 472 (2021).
Fu, L., Liu, Z. & Liu, Y. Fibrinogen-like protein 2 in inflammatory diseases: A future therapeutic target. Int. Immunopharmacol. 116, p. 109799 (2023).
Lin, H. et al. Elevated fibrinogen-like protein 2 in TNBS-induced colitis mice: association with Th17 and regulatory T cells. Mol. Med. Rep. 16(3), 3445–3454 (2017).
Hoppe, B. Fibrinogen and factor XIII at the intersection of coagulation, fibrinolysis and inflammation. Thromb. Haemost. 112(4), 649–658 (2014).
Kattula, S., Byrnes, J. R. & Wolberg, A. S. Fibrinogen and fibrin in hemostasis and thrombosis. Arterioscler. Thromb. Vasc. Biol. 37(3), e13–e21 (2017).
Davalos, D. & Akassoglou, K. Fibrinogen as a key regulator of inflammation in disease. Semin. Immunopathol. 34(1), 43–62 (2012).
Kearney, K. J., Ariëns, R. A. S. & Macrae, F. L. The role of fibrin(ogen) in wound healing and infection control. Semin. Thromb. Hemost. 48(2), 174–187 (2022).
Seltana, A. et al. Fibrin(ogen) is constitutively expressed by differentiated intestinal epithelial cells and mediates wound healing. Front. Immunol. 13, p. 916187 (2022).
Bayer, I. S. Advances in fibrin-based materials in wound repair: A review. Molecules https://doi.org/10.3390/molecules27144504 (2022).
Owczarek, D. et al. Reduced plasma fibrin clot permeability and susceptibility to lysis in patients with inflammatory bowel disease: A novel prothrombotic mechanism. Inflamm. Bowel Dis. 19(12), 2616–2624 (2013).
Molitoris, B. A. et al. Albumin uptake and processing by the proximal tubule: physiological, pathological, and therapeutic implications. Physiol. Rev. 102(4), 1625–1667 (2022).
Garcia-Martinez, R. et al. Albumin: Pathophysiologic basis of its role in the treatment of cirrhosis and its complications. Hepatology 58(5), 1836–1846 (2013).
Caraceni, P., OBrien, A. & Gines, P. Long-term albumin treatment in patients with cirrhosis and ascites. J. Hepatol. 76(6), 1306–1317 (2022).
Alvisi, P. et al. Major abdominal surgery for pediatric Crohns disease in the anti-TNF era: 10-year analysis of data from the IBD Registry of Italian Society of Pediatric Gastroenterology, Hepatology, and Nutrition. Inflamm. Bowel Dis. 30(11), 2087–2096 (2024).
Rivière, P. et al. Comparative effectiveness of Ustekinumab and anti-TNF agent as first-line biological therapy in luminal Crohns disease: A retrospective study from 2 referral centers. Inflamm. Bowel Dis. 29(6), 923–931 (2023).
Nagata, Y. et al. Anti-tumor necrosis factor therapy decreases the risk of initial intestinal surgery after diagnosis of Crohns disease of inflammatory type. J. Gastroenterol. 54(4), 330–338 (2019).
González-Lama, Y. et al. Timing of Thiopurine or anti-TNF initiation is associated with the risk of major abdominal surgery in Crohns disease: A retrospective cohort study. J. Crohns Colitis 10(1), 55–60 (2016).
Quaresma, A. B., Yamamoto, T. & Kotze, P. G. Biologics and surgical outcomes in Crohns disease: Is there a direct relationship?. Therap. Adv. Gastroenterol. 13, p. 1756284820931738 (2020).
Acknowledgements
We highly appreciate all the patients who were involved in the study and the guidance and dedication of the Pro. Yuan.
Author information
Authors and Affiliations
Contributions
Conceptualization: K.X., H.Y., L.Y., L.Y. Data curation: Z.J., H.L., Q.S. Formal analysis: K.X., H.Y., L.Y. Visualization: K.X., H.Y., L.Y. Writing – original draft: K.X., H.Y., Z.J., H.L., Q.S., L.Y., L.Y. Writing – review & editing: K.X., H.Y., Z.J., H.L., Q.S., L.Y., L.Y. Funding acquisition: L.Y. All authors reviewed the manuscript
Corresponding authors
Ethics declarations
Competing interest
The authors declare no competing interests.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Xiangya Second Hospital (Ethics ID: LYEC2025-0156), and followed the Declaration of Helsinki. Patient data were anonymized throughout the study. As this was a retrospective study, informed consent was waived.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xie, K., Yao, H., Jiang, Z. et al. A web-based prediction model using routinely available clinical variables to estimate surgical risk in Crohn’s disease during remission or mild activity. Sci Rep (2026). https://doi.org/10.1038/s41598-026-48056-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-48056-7