Abstract
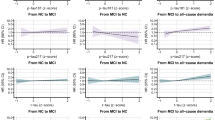
Background and Objectives: Impaired lung function (ILF) has been associated with cognitive decline and dementia risk in multiple cohorts, yet the role of circulating Alzheimer disease (AD) biomarkers in this relationship is not well understood. We aimed to assess the associations of ILF with AD-related blood biomarkers and to evaluate whether these biomarkers statistically accounted for part of the observed association between ILF and incident dementia. Methods: Serum p-Tau181 and plasma Aβ42/40, NfL, and GFAP were measured in 4,072 participants (mean age 66 ± 10 years; 59% women) in the 2016 Health and Retirement Study. Peak Expiratory Flow (PEF) was assessed in 2012/2014, and cognitive function was measured at four time points between 2014 and 2020 (every two years) to determine dementia status. ILF was defined as predicted PEF < 80%. Multivariable regression examined associations between ILF and AD biomarkers. Mediation models were used to evaluate the extent to which biomarkers statistically accounted for the association between ILF and incident dementia. Results: In total, 881 (21.6%) participants had ILF and 272 (6.8%) participants developed dementia. After adjusting for demographics, education, BMI, smoking, comorbidities, inflammation, eGFR and APOE e4, ILF was associated with a higher risk of dementia (HR = 1.74; 95% CI (1.34, 2.25)). Individuals with ILF had 0.10 SD higher NfL (SE = 0.03; p = 0.004) and 0.09 SD higher p-Tau 181 (SE = 0.03; p = 0.002) compared to those without ILF. In mediation models, higher NfL and p Tau181 accounted for modest proportions of the observed association between ILF and incident dementia, with estimated mediated proportions of 7.3% for NfL and 4.9% for p Tau181. Discussion: ILF was associated with higher levels of blood biomarkers reflecting neurodegeneration and with increased risk of dementia. NfL and p-Tau181 accounted for modest proportions of this association in mediation analyses, suggesting a limited and exploratory contribution. These findings support further investigation into biological pathways linking lung function and cognitive decline.
Similar content being viewed by others
Data availability
We used the HRS publicly available datasets and sensitive biomarker data for this study analysis. This data can be found here: https://hrsdata.isr.umich.edu/data-products/public-survey-data and https://hrsdata.isr.umich.edu/dataproducts/sensitive-health and can be accessed by completing required data use agreement.
References
Xiao, T. et al. Lung Function Impairment and the Risk of Incident Dementia: The Rotterdam Study. J. Alzheimer’s disease: JAD. 82 (2), 621–630. https://doi.org/10.3233/jad-210162 (2021).
Gilsanz, P. et al. Early Midlife Pulmonary Function and Dementia Risk. Alzheimer disease and associated disorders. Oct-Dec 32 (4), 270–275. https://doi.org/10.1097/wad.0000000000000253 (2018).
Xie, W. et al. Reduced Lung Function and Cognitive Decline in Aging: A Longitudinal Cohort Study. Annals Am. Thorac. Society Feb. 18 (2), 373–376. https://doi.org/10.1513/AnnalsATS.202009-1152RL (2021).
Joyce, B. T. et al. Pulmonary Function in Midlife as a Predictor of Later-Life Cognition: The Coronary Artery Risk Development in Adults (CARDIA) Study. Journals Gerontology: Ser. A. 77 (12), 2517–2523. https://doi.org/10.1093/gerona/glac026 (2022).
Zhou, L. et al. Association of impaired lung function with dementia, and brain magnetic resonance imaging indices: a large population-based longitudinal study. Age Ageing Nov. https://doi.org/10.1093/ageing/afac269 (2022).
Donahue, P. T., Xue, Q. L., Carlson & Series, A. Peak Expiratory Flow Predicts Incident Dementia in a Representative Sample of U.S. Older Adults: The National Health and Aging Trends Study (NHATS). J Gerontol Biol Sci Med Sci. 78(8):1427–1435. (2023). https://doi.org/10.1093/gerona/glac235
Shrestha, S. et al. Association of Lung Function With Cognitive Decline and Incident Dementia in the Atherosclerosis Risk in Communities Study. Am. J. Epidemiol. 192 (10), 1637–1646. https://doi.org/10.1093/aje/kwad140 (2023).
Pathan, S. S. et al. Association of lung function with cognitive decline and dementia: the Atherosclerosis Risk in Communities (ARIC) Study. European J. neurology Jun. 18 (6), 888–898. https://doi.org/10.1111/j.1468-1331.2010.03340.x (2011).
Lutsey, P. L. et al. Impaired Lung Function, Lung Disease, and Risk of Incident Dementia. American J. respiratory Crit. care medicine Jun. 1 (11), 1385–1396. https://doi.org/10.1164/rccm.201807-1220OC (2019).
Wang, J. et al. Poor pulmonary function is associated with mild cognitive impairment, its progression to dementia, and brain pathologies: A community-based cohort study. Alzheimer’s Dement. 18 (12), 2551–2559. (2022). https://doi.org/10.1002/alz.12625
Frenzel, S. et al. Associations of Pulmonary Function with MRI Brain Volumes: A Coordinated Multi-Study Analysis. J. Alzheimer’s disease: JAD. 90 (3), 1073–1083. https://doi.org/10.3233/jad-220667 (2022).
Ashton, N. J. et al. Diagnostic Accuracy of a Plasma Phosphorylated Tau 217 Immunoassay for Alzheimer Disease Pathology. JAMA Neurol. 81 (3), 255–263. https://doi.org/10.1001/jamaneurol.2023.5319 (2024).
Leuzy, A. et al. Blood-based biomarkers for Alzheimer’s disease. EMBO Mol. medicine Jan. 11 (1), e14408. https://doi.org/10.15252/emmm.202114408 (2022).
Mandal, P. K. et al. Blood Biomarkers in Alzheimer’s Disease. ACS Chem. neuroscience Nov. 15 (22), 3975–3978. https://doi.org/10.1021/acschemneuro.3c00641 (2023).
Graff-Radford, N. R. et al. Association of Low Plasma Aβ42/Aβ40 Ratios With Increased Imminent Risk for Mild Cognitive Impairment and Alzheimer Disease. Arch. Neurol. 64 (3), 354–362. https://doi.org/10.1001/archneur.64.3.354 (2007).
Chatterjee, P. et al. Plasma Aβ42/40 ratio, p-tau181, GFAP, and NfL across the Alzheimer’s disease continuum: A cross-sectional and longitudinal study in the AIBL cohort. Alzheimer’s & dementia: J. Alzheimer’s Association Apr. 19 (4), 1117–1134. https://doi.org/10.1002/alz.12724 (2023).
Xiao, Z. et al. Plasma p-tau217, p-tau181, and NfL as early indicators of dementia risk in a community cohort: The Shanghai Aging Study. Alzheimer’s & dementia (Amsterdam, Netherlands). Oct-Dec 15 (4), e12514. https://doi.org/10.1002/dad2.12514 (2023).
Stevenson-Hoare, J. et al. Plasma biomarkers and genetics in the diagnosis and prediction of Alzheimer’s disease. Brain: J. neurology Feb. 13 (2), 690–699. https://doi.org/10.1093/brain/awac128 (2023).
Teunissen, C. E. et al. Blood-based biomarkers for Alzheimer’s disease: towards clinical implementation. The Lancet Neurology Jan. 21 (1), 66–77. https://doi.org/10.1016/s1474-4422(21)00361-6 (2022).
Rebelos, E. et al. Circulating neurofilament is linked with morbid obesity, renal function, and brain density. Sci Rep May. 12 (1), 7841. https://doi.org/10.1038/s41598-022-11557-2 (2022).
Hermesdorf, M. et al. Longitudinal analyses of serum neurofilament light and associations with obesity indices and bioelectrical impedance parameters. Sci. Rep. 12 (1), 15863. (2022). https://doi.org/10.1038/s41598-022-20398-y
Yalachkov, Y. et al. Effect of Estimated Blood Volume and Body Mass Index on GFAP and NfL Levels in the Serum and CSF of Patients With Multiple Sclerosis. Neurol. Neuroimmunol. Neuroinflamm. https://doi.org/10.1212/nxi.0000000000200045
Sonnega, A. et al. Cohort Profile: the Health and Retirement Study (HRS). International J. epidemiology Apr. 43 (2), 576–585. https://doi.org/10.1093/ije/dyu067 (2014).
Crimmins, E. M., Kim, J. K., Langa, K. M. & Weir, D. R. Assessment of Cognition Using Surveys and Neuropsychological Assessment: The Health and Retirement Study and the Aging, Demographics, and Memory Study. Journals Gerontology: Ser. B. 66B (suppl_1), i162–i171. https://doi.org/10.1093/geronb/gbr048 (2011).
Crimmins, E. et al. Documentation of physical measures, anthropometrics and blood pressure in the Health and Retirement Study. HRS Doc. Rep. DR-011. 14 (1–2), 47–59 (2008).
Heeringa, S. G. & Connor, J. H. Technical description of the Health and Retirement Survey sample design. Ann Arbor: Univ. Michigan ; (1995).
Crimmins, E. et al. Venus blood collection and assay protocol in the 2016 Health and Retirement Study 2016 Vensu Blood Study (VBS). (2017). https://hrsdata.isr.umich.edu/sites/default/files/documentation/data-descriptions/HRS2016VBSDD.pdf. Accessed June 2024.
DeVrieze, B. W., Goldin, J. & Giwa, A. O. Peak Flow Rate Measurement. StatPearls. StatPearls Publishing. Copyright ©. StatPearls Publishing LLC.; 2025. (2025).
Hankinson, J. L., Odencrantz, J. R. & Fedan, K. B. Spirometric Reference Values from a Sample of the General U.S. Population. American J. respiratory Crit. care medicine 1999/01/01. 159 (1), 179–187. https://doi.org/10.1164/ajrccm.159.1.9712108 (1999).
Panikkar, D. et al. Pre-Analytical Variables Influencing Stability of Blood-Based Biomarkers of Neuropathology. J. Alzheimer’s disease: JAD. 95 (2), 735–748. https://doi.org/10.3233/jad-230384 (2023).
Meier, H. C. S., Mitchell, C., Karadimas, T. & Faul, J. D. Systemic inflammation and biological aging in the Health and Retirement Study. GeroScience Dec. 45 (6), 3257–3265. https://doi.org/10.1007/s11357-023-00880-9 (2023).
Faul, J., Smith, J. & Zhao, W. Health and retirement study: candidate gene and SNP data description. Health Retire Study, Univ Mich, Ann Arbor, MIhttp://hrsonline isr umich edu/sitedocs/genetics/candidategene/CandidateGeneSNPDataDescription . (2014).
Faul, J., Smith, J. & Zhao, W. Health and Retirement Study: Candidate genes for cognition/behavior. Ann Arbor MI: Univ. Michigan ; (2014).
Hoost, S. S. et al. Effects of Vascular Risk Factors on the Association of Blood-Based Biomarkers with Alzheimer’s Disease. Med. Res. Arch. https://doi.org/10.18103/mra.v11i9.4468 (2023).
Pichet Binette, A. et al. Confounding factors of Alzheimer’s disease plasma biomarkers and their impact on clinical performance. Alzheimer’s & dementia: J. Alzheimer’s Association Apr. 19 (4), 1403–1414. https://doi.org/10.1002/alz.12787 (2023).
Guo, X. et al. Midlife respiratory function related to white matter lesions and lacunar infarcts in late life: the Prospective Population Study of Women in Gothenburg, Sweden. Stroke Jul. 37 (7), 1658–1662. https://doi.org/10.1161/01.STR.0000226403.00963.af (2006).
Murray, A. D. et al. Brain White Matter Hyperintensities: Relative Importance of Vascular Risk Factors in Nondemented Elderly People. Radiology 237 (1), 251–257. (2005). https://doi.org/10.1148/radiol.2371041496
Liao, D. et al. Lower pulmonary function and cerebral subclinical abnormalities detected by MRI: the Atherosclerosis Risk in Communities study. Chest Jul. 116 (1), 150–156. https://doi.org/10.1378/chest.116.1.150 (1999).
Desai, P. et al. Examination of Neurofilament Light Chain Serum Concentrations, Physical Activity, and Cognitive Decline in Older Adults. JAMA Netw. open Mar. 1 (3), e223596. https://doi.org/10.1001/jamanetworkopen.2022.3596 (2022).
Zheng, G. Q., Wang, Y. & Wang, X. T. Chronic hypoxia-hypercapnia influences cognitive function: a possible new model of cognitive dysfunction in chronic obstructive pulmonary disease. Med. Hypotheses. 71 (1), 111–113. https://doi.org/10.1016/j.mehy.2008.01.025 (2008).
Areza-Fegyveres, R., Kairalla, R. A., Carvalho, C. R. R. & Nitrini, R. Cognition and chronic hypoxia in pulmonary diseases. Dement. Neuropsychol. 4 (1), 14–22. (2010) https://doi.org/10.1590/s1980-57642010dn40100003
Miners, S., Kehoe, P. G. & Love, S. Cognitive impact of COVID-19: looking beyond the short term. Alzheimer’s Res. Ther. 12 (1), 170. (2020). https://doi.org/10.1186/s13195-020-00744-w
Ortiz, G. G. et al. Alzheimer’s Disease and SARS-CoV-2: Pathophysiological Analysis and Social Context. Brain Sci. (2022). https://doi.org/10.3390/brainsci12101405
Kim, K. Y., Shin, K. Y. & Chang, K. A. GFAP as a Potential Biomarker for Alzheimer’s Disease: A Systematic Review and Meta-Analysis. Cells May. (2023). https://doi.org/10.3390/cells12091309
Funding
The author(s) acknowledge financial backing for the study, writing, and/or publication of this article. This project received support from the department grants and the Health and Retirement Study is sponsored by NIA through grant U01 AG009740.
Author information
Authors and Affiliations
Contributions
SV: Conceptualization, formal analysis and methodology, writing – original draft, review and editing. EC: Data collection, Writing – critical review and editing. JKK: Data development, Writing – critical review and editing. JF: Data collection, Writing – critical review and editing. DJ: Analysis methodology, Writing – critical review and editing.WG: Analysis methodology, Writing – critical review and editing. BT: Data collection, conceptualization, Writing – critical review and editing.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The venous blood study involving human samples was approved by University of Minnesota Institutional Review Board.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Vivek, S., Crimmins, E.M., Kim, J.K. et al. Impaired lung function is associated with elevated blood biomarkers of AD/ADRD: unraveling the interplay with risk of dementia. Sci Rep (2026). https://doi.org/10.1038/s41598-026-48115-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-48115-z