Abstract
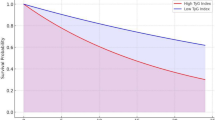
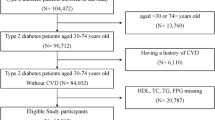
We defined triglyceride-glucose (TyG) index < 8.52 as a low TyG index and TyG index ≥ 8.52 as a high TyG index, and divided the subjects into nine groups based on the levels of TyG index and fasting blood glucose (FBG) levels: FBG levels < 90 mg/dL and low TyG index, FBG levels < 90 mg/dL and high TyG index, FBG levels of 90–94 mg/dL and low TyG index, FBG levels of 90–94 mg/dL and high TyG index, FBG levels of 95–99 mg/dL and low TyG index, FBG levels of 95–99 mg/dL and high TyG index, FBG levels of 100–125 mg/dL and low TyG index, FBG levels of 100–125 mg/dL and high TyG index, and diabetes mellitus. The odds ratios for endothelial dysfunction were significantly higher in groups with high TyG index and any levels of FBG, and the group with low TyG index and FBG levels of 95–99 mg/dL, the group with low TyG index and FBG levels of 100–125 mg/dL and the diabetes mellitus group than the FBG levels < 90 mg/dL and low TyG index group. Even at a low TyG index, subjects with FBG levels ≥ 95 mg/dL might have a higher risk for endothelial dysfunction.
Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are not publicly available due to protecting participant confidentiality but are available from the corresponding author on reasonable request.
References
Ross, R. Atherosclerosis–An inflammatory disease. N. Engl. J. Med. 340, 115–126. https://doi.org/10.1056/nejm199901143400207 (1999).
Higashi, Y., Noma, K., Yoshizumi, M. & Kihara, Y. Endothelial function and oxidative stress in cardiovascular diseases. Circ. J. 73, 411–418 (2009).
Yamaji, T. et al. Self-reported total sitting time on a non-working day is associated with blunted flow-mediated vasodilation and blunted nitroglycerine-induced vasodilation. Sci. Rep. 12, 6366. https://doi.org/10.1038/s41598-022-10242-8 (2022).
Oda, N. et al. Endothelial function is impaired in relation to alcohol intake even in the case of light alcohol consumption in Asian men; flow-mediated dilation Japan (FMD-J) study. Int. J. Cardiol. 230, 523–528. https://doi.org/10.1016/j.ijcard.2016.12.065 (2017).
Matsui, S. et al. Decreased frequency and duration of tooth brushing is a risk factor for endothelial dysfunction. Int. J. Cardiol. 241, 30–34. https://doi.org/10.1016/j.ijcard.2017.03.049 (2017).
Hashimoto, H. et al. Smoking status and endothelial function in Japanese men. Sci. Rep. 11, 95. https://doi.org/10.1038/s41598-020-80012-x (2021).
Higashi, Y. Noninvasive assessment of vascular function: From physiological tests to biomarkers. JACC. Asia 4, 898–911. https://doi.org/10.1016/j.jacasi.2024.09.015 (2024).
Maruhashi, T. et al. Relationship between flow-mediated vasodilation and cardiovascular risk factors in a large community-based study. Heart 99, 1837–1842. https://doi.org/10.1136/heartjnl-2013-304739 (2013).
Yamaji, T. et al. Pre-impaired fasting glucose state is a risk factor for endothelial dysfunction: Flow-mediated Dilation Japan (FMD-J) study. BMJ Open Diabetes Res. Care. https://doi.org/10.1136/bmjdrc-2020-001610 (2020).
Defronzo, R. A. Banting Lecture. From the triumvirate to the ominous octet: A new paradigm for the treatment of type 2 diabetes mellitus. Diabetes 58, 773–795. https://doi.org/10.2337/db09-9028 (2009).
Tao, L. C., Xu, J. N., Wang, T. T., Hua, F. & Li, J. J. Triglyceride-glucose index as a marker in cardiovascular diseases: Landscape and limitations. Cardiovasc. Diabetol. 21, 68. https://doi.org/10.1186/s12933-022-01511-x (2022).
Sasaki, N., Ueno, Y., Ozono, R., Nakano, Y. & Higashi, Y. Insulin resistance in the adipose tissue predicts future vascular resistance: The Hiroshima study on glucose metabolism and cardiovascular diseases. Atherosclerosis 393, 117547. https://doi.org/10.1016/j.atherosclerosis.2024.117547 (2024).
Bergman, R. N., Finegood, D. T. & Ader, M. Assessment of insulin sensitivity in vivo. Endocr. Rev. 6, 45–86. https://doi.org/10.1210/edrv-6-1-45 (1985).
Matthews, D. R. et al. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28, 412–419. https://doi.org/10.1007/bf00280883 (1985).
Unger, G., Benozzi, S. F., Perruzza, F. & Pennacchiotti, G. L. Triglycerides and glucose index: A useful indicator of insulin resistance. Endocrinol. Nutr. 61, 533–540. https://doi.org/10.1016/j.endonu.2014.06.009 (2014).
Kim, M. K. et al. Relationship between the triglyceride glucose index and coronary artery calcification in Korean adults. Cardiovasc. Diabetol. 16, 108. https://doi.org/10.1186/s12933-017-0589-4 (2017).
Ding, X., Wang, X., Wu, J., Zhang, M. & Cui, M. Triglyceride-glucose index and the incidence of atherosclerotic cardiovascular diseases: A meta-analysis of cohort studies. Cardiovasc. Diabetol. 20, 76. https://doi.org/10.1186/s12933-021-01268-9 (2021).
Son, D. H., Lee, H. S., Lee, Y. J., Lee, J. H. & Han, J. H. Comparison of triglyceride-glucose index and HOMA-IR for predicting prevalence and incidence of metabolic syndrome. Nutr. Metab. Cardiovasc. Dis. 32, 596–604. https://doi.org/10.1016/j.numecd.2021.11.017 (2022).
Zhang, M. et al. Cumulative increased risk of incident type 2 diabetes mellitus with increasing triglyceride glucose index in normal-weight people: The Rural Chinese Cohort Study. Cardiovasc. Diabetol. 16, 30. https://doi.org/10.1186/s12933-017-0514-x (2017).
Li, Y. et al. Association between triglyceride-glucose index and endothelial dysfunction. Endocrine 85, 717–723. https://doi.org/10.1007/s12020-024-03785-5 (2024).
Kajikawa, M. et al. Relationship between serum triglyceride levels and endothelial function in a large community-based study. Atherosclerosis 249, 70–75. https://doi.org/10.1016/j.atherosclerosis.2016.03.035 (2016).
Huang, R. et al. Prognostic value of triglyceride glucose (TyG) index in patients with acute decompensated heart failure. Cardiovasc. Diabetol. 21, 88. https://doi.org/10.1186/s12933-022-01507-7 (2022).
Cai, X. L. et al. Prognostic value of triglyceride glucose index in population at high cardiovascular disease risk. Cardiovasc. Diabetol. 22, 198. https://doi.org/10.1186/s12933-023-01924-2 (2023).
Vicent, D. et al. The role of endothelial insulin signaling in the regulation of vascular tone and insulin resistance. J. Clin. Invest. 111, 1373–1380. https://doi.org/10.1172/jci15211 (2003).
Veys, K. et al. Role of the GLUT1 glucose transporter in postnatal CNS angiogenesis and blood-brain barrier integrity. Circ. Res. 127, 466–482. https://doi.org/10.1161/circresaha.119.316463 (2020).
Kodama, K. et al. Ethnic differences in the relationship between insulin sensitivity and insulin response: A systematic review and meta-analysis. Diabetes Care 36, 1789–1796. https://doi.org/10.2337/dc12-1235 (2013).
Tomiyama, H. et al. A multicenter study design to assess the clinical usefulness of semi-automatic measurement of flow-mediated vasodilatation of the brachial artery. Int. Heart J. 53, 170–175 (2012).
Umemura, S. et al. The Japanese Society of Hypertension Guidelines for the Management of Hypertension (JSH 2019). Hypertens. Res. 42, 1235–1481. https://doi.org/10.1038/s41440-019-0284-9 (2019).
Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). Jama. 285, 2486–2497 (2001).
American Diabetes Association. Clinical practice recommendations 1999. Diabetes Care 22(Suppl 1), S1-114 (1999).
Maruhashi, T. et al. Nitroglycerine-induced vasodilation for assessment of vascular function: A comparison with flow-mediated vasodilation. Arterioscler. Thromb. Vasc. Biol. 33, 1401–1408. https://doi.org/10.1161/atvbaha.112.300934 (2013).
Acknowledgements
The authors would like to thank all of the patients who participated in this study. In addition, we thank Miki Kumiji, Megumi Wakisaka, Ki-ichiro Kawano and Satoko Michiyama for their excellent secretarial assistance; FMD-J investigators Takayuki Hidaka, MD, PhD; Shuji Nakamura, MD, PhD; Junko Soga, MD, PhD; Yuichi Fujii, MD, PhD; Naomi Idei, MD; Noritaka Fujimura, MD, PhD; Shinsuke Mikami, MD, PhD; Yumiko Iwamoto, MD; Akimichi Iwamoto, MD, PhD; Takeshi Matsumoto, MD, PhD; Nozomu Oda, MD, PhD (Department of Cardiovascular Medicine, Hiroshima University Graduate School of Biomedical Sciences, Hiroshima, Japan); Kana Kanai, PhD; Haruka Morimoto, PhD (Department of Cardiovascular Regeneration and Medicine, Research Institute for Radiation Biology and Medicine, Hiroshima University, Hiroshima, Japan); Tomohisa Sakashita, MD, PhD; Yoshiki Kudo, MD, PhD (Department of Obstetrics and Gynecology, Hiroshima University Graduate School of Biomedical Sciences, Hiroshima, Japan); Taijiro Sueda, MD, PhD (Department of Surgery, Hiroshima University Graduate School of Biomedical Sciences, Hiroshima, Japan); Akira Yamashina, MD, PhD (Department of Cardiology, Tokyo Medical University, Tokyo, Japan); Bonpei Takase, MD, PhD, FAHA (Division of Biomedical Engineering, National Defense Medical College Research Institute, Tokorozawa, Japan); Takahide Kohro, MD, PhD (Department of Cardiology, Tokyo Medical University, Tokyo, Japan); Toru Suzuki, MD, PhD (Cardiovascular Medicine, University of Leicester, Leicester, UK); Tomoko Ishizu, MD, PhD (Cardiovascular Division, Institute of Clinical Medicine, University of Tsukuba, Ibaraki, Japan); Shinichiro Ueda, MD, PhD (Department of Clinical Pharmacology and Therapeutics, University of the Ryukyu School of Medicine, Okinawa, Japan); Tsutomu Yamazaki, MD, PhD (Clinical Research Support Center, Faculty of Medicine, The University of Tokyo, Tokyo, Japan); Tomoo Furumoto, MD, PhD (Department of Cardiovascular Medicine, Hokkaido University Graduate School of Medicine, Hokkaido, Japan); Kazuomi Kario, MD, PhD (Division of Cardiovascular Medicine, Jichi Medical University School of Medicine, Tochigi, Japan); Teruo Inoue, MD, PhD (Department of Cardiovascular Medicine, Dokkyo Medical University, Mibu, Tochigi, Japan); Shinji Koba, MD, PhD (Department of Medicine, Division of Cardiology, Showa University School of Medicine, Tokyo, Japan); Kentaro Watanabe, MD, PhD (Department of Neurology, Hematology, Metaboism, Endocrinology and Diabetology (DNHMED), Yamagata University School of Medicine, Yamagata, Japan); Yasuhiko Takemoto, MD, PhD (Department of Internal Medicine and Cardiology, Osaka City University Graduate School of Medicine, Osaka, Japan); Takuzo Hano, MD, PhD (Department of Medical Education and Population-based Medicine, Postgraduate School of Medicine, Wakayama Medical University, Wakayama, Japan); Masataka Sata, MD, PhD (Department of Cardiovascular Medicine, Institute of Health Biosciences, The University of Tokushima Graduate School, Tokushima, Japan); Yutaka Ishibashi, MD, PhD (Department of General Medicine, Shimane University Faculty of Medicine, Izumo, Japan); Koichi Node, MD, PhD (Department of Cardiovascular and Renal Medicine, Saga University, Saga, Japan); Koji Maemura, MD, PhD (Department of Cardiovascular Medicine, Nagasaki University Graduate School of Biomedical Sciences, Nagasaki, Japan); Yusuke Ohya, MD, PhD (The Third Department of Internal Medicine, University of the Ryukyus, Okinawa, Japan); Taiji Furukawa, MD, PhD (Department of Internal Medicine, Teikyo University School of Medicine, Tokyo, Japan); Hiroshi Ito, MD, PhD (Department of Cardiovascular Medicine, Okayama University Graduate School of Medicine, Dentistry and Pharmaceutical Sciences, Japan); and Hisao Ikeda, MD, PhD (Faculty of Fukuoka Medical Technology, Teikyo University, Omuta, Japan).
Funding
Grant-in-Aid for Scientific Research from the Ministry of Education, Science and Culture of Japan (18590815 and 21590898 to Y.Higashi), a Grant-in-Aid of Japanese Arteriosclerosis Prevention Fund (to Y. Higashi), Hirose Fundation (to T. Yamaji).
Author information
Authors and Affiliations
Contributions
T.Yamaji. and Y.Higashi, drafting the article and conception of the study; T.Yamaji., F.M.Y., S.K., M.K., T.H., A.M., S.T., and T.M. acquiring subjects and/or data; Y.N. revising the article critically for important intellectual content. Y. Higashi is the guarantor of this work and, as such, had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of data analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yamaji, T., Yusoff, F.M., Kishimoto, S. et al. A cut-off value for endothelial dysfunction based on the triglyceride-glucose index. Sci Rep (2026). https://doi.org/10.1038/s41598-026-48550-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-48550-y