Abstract
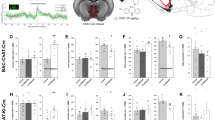
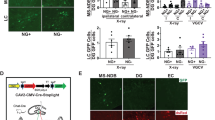
Cholinergic dysfunction is a key contributor to cognitive impairment observed in aging and neurodegenerative disorders such as Alzheimer’s disease (AD). Although acetylcholinesterase (AChE) inhibitors have been the mainstay of symptomatic treatment for over two decades, their limited efficacy and adverse effects underscore the need for alternative therapeutic approaches. Recent evidence indicates that mechanical stimulation can modulate neuronal and glial signaling through mechanotransduction, suggesting a potential strategy to enhance cognitive function via non-pharmacological means. Here, we developed a head-mounted vibrotactile stimulation system (HVSS) that delivers controlled vibration to the cranium and evaluated its effects in a pharmacological model of acute cholinergic dysfunction induced by scopolamine. To this end, male C57BL/6 mice received scopolamine (1 mg/kg, i.p.; on days 7, 14, and 28) and were exposed to daily vibrotactile stimulation at 20, 40, or 80 Hz for 28 days. Behavioral performance was assessed using passive avoidance and Morris water maze tests, followed by biochemical and histological analyses. HVSS at 40 Hz and 80 Hz significantly improved cognitive performance, enhanced hippocampal cholinergic function, reduced oxidative damage, and upregulated memory-related signaling genes, including BDNF, PI3K, AKt, ERK1/2, CREB, and CAMK4. These findings suggest that high-frequency HVSS improves memory hippocampal cholinergic function via activation of memory-related signaling pathways, highlighting its potential as a safe, non-pharmacological neuromodulatory strategy for cholinergic dysfunction-related cognitive decline.
Similar content being viewed by others
Data availability
The datasets used and/or analysed during the current study available from the corredsponding author on reasonable request.
References
Chuong, N. N. et al. Anti-amnesic effect of alkaloid fraction from Lycopodiella cernua (L.) Pic. Serm. on scopolamine-induced memory impairment in mice. Neurosci. Lett. 575, 42–46. https://doi.org/10.1016/j.neulet.2014.05.031 (2014).
Becker, R., Giacobini, E., Elble, R., McIlhany, M. & Sherman, K. Potential pharmacotherapy of Alzheimer disease. A comparison of various forms of physostigmine administration. Acta Neurol. Scand. Suppl. 116, 19–32. https://doi.org/10.1111/j.1600-0404.1988.tb07983.x (1988).
Blusztajn, J. K. & Wurtman, R. J. Choline and cholinergic neurons. Science 221, 614–620. https://doi.org/10.1126/science.6867732 (1983).
Watkins, P. B., Zimmerman, H. J., Knapp, M. J., Gracon, S. I. & Lewis, K. W. Hepatotoxic effects of tacrine administration in patients with Alzheimer’s disease. JAMA 271, 992–998 (1994).
Park, S. M. et al. Tacrine, an oral acetylcholinesterase inhibitor, induced hepatic oxidative damage, which was blocked by liquiritigenin through GSK3-beta inhibition. Biol. Pharm. Bull. 38, 184–192. https://doi.org/10.1248/bpb.b14-00430 (2015).
Sims, J. R. et al. Donanemab in early symptomatic Alzheimer disease: The TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA 330, 512–527. https://doi.org/10.1001/jama.2023.13239 (2023).
van Dyck, C. H. et al. Lecanemab in early Alzheimer’s disease. N. Engl. J. Med. 388, 9–21. https://doi.org/10.1056/NEJMoa2212948 (2023).
Cho, H. et al. Neural differentiation of umbilical cord mesenchymal stem cells by sub-sonic vibration. Life Sci. 90, 591–599. https://doi.org/10.1016/j.lfs.2012.02.014 (2012).
Lee, G. Y., Kim, O. H., Kim, E. R. & Lee, H. J. Biomechanical forces in the aged brain: Relationship to AD. Life Sci 312, 121237. https://doi.org/10.1016/j.lfs.2022.121237 (2023).
Martorell, A. J. et al. Multi-sensory gamma stimulation ameliorates Alzheimer’s-associated pathology and improves cognition. Cell 177, 256-271 e222. https://doi.org/10.1016/j.cell.2019.02.014 (2019).
Paulson, A. L., Zhang, L., Prichard, A. M. & Singer, A. C. 40 Hz sensory stimulation enhances CA3-CA1 coordination and prospective coding during navigation in a mouse model of Alzheimer’s disease. Proc. Natl. Acad. Sci. U. S. A. 122, e2419364122. https://doi.org/10.1073/pnas.2419364122 (2025).
Chen, X. et al. Unleashing the potential: 40 Hz multisensory stimulation therapy for cognitive impairment. J. Cent. Nerv. Syst. Dis. 17, 11795735251328029. https://doi.org/10.1177/11795735251328029 (2025).
Ahuja, G. et al. The effects of whole-body vibration therapy on immune and brain functioning: Current insights in the underlying cellular and molecular mechanisms. Front. Neurol. 15, 1422152. https://doi.org/10.3389/fneur.2024.1422152 (2024).
Park, M. et al. Effects of transcranial ultrasound stimulation pulsed at 40 Hz on Aβ plaques and brain rhythms in 5×FAD mice. Transl. Neurodegener. 10, 48. https://doi.org/10.1186/s40035-021-00274-x (2021).
Hu, J. R., Chun, Y. S., Kim, J. K., Cho, I. J. & Ku, S. K. Ginseng berry aqueous extract prevents scopolamine-induced memory impairment in mice. Exp. Ther. Med. 18, 4388–4396. https://doi.org/10.3892/etm.2019.8090 (2019).
Lee, S. et al. Sulforaphane alleviates scopolamine-induced memory impairment in mice. Pharmacol. Res. 85, 23–32. https://doi.org/10.1016/j.phrs.2014.05.003 (2014).
Kim, D.-W. et al. Anti-amnesic effect of curcumin spray dry powders on scopolamine-induced mouse memory impairment. J. Korean Soc. Food Sci. Nutr. 51, 751–764. https://doi.org/10.3746/jkfn.2022.51.8.751 (2022).
Kim, Y. R. et al. Beneficial effects of Gagam‐Palmultang on scopolamine‐induced memory deficits in mice. Evid. Based Complement. Alternat. Med. 2018, 3479083. https://doi.org/10.1155/2018/3479083 (2018).
Ryu, H. S. et al. Golden oyster mushroom extract ameliorates oxidative stress-induced cell death in neurons and scopolamine-induced cholinergic system impairment in mice. Mol. Neurobiol. https://doi.org/10.1007/s12035-025-05223-7 (2025).
Morris, R. Developments of a water-maze procedure for studying spatial learning in the rat. J. Neurosci. Methods. 11, 47–60. https://doi.org/10.1016/0165-0270(84)90007-4 (1984).
Hung, T. M. et al. Cholinesterase inhibitory and anti-amnesic activity of alkaloids from Corydalis turtschaninovii. J. Ethnopharmacol. 119, 74–80. https://doi.org/10.1016/j.jep.2008.05.041 (2008).
Nam, Y. & Lee, D. Ameliorating effects of constituents from Cortex Acanthopanacis Radicis on memory impairment in mice induced by scopolamine. J. Tradit. Chin. Med. 34, 57–62. https://doi.org/10.1016/s0254-6272(14)60055-8 (2014).
Del Rio, D., Stewart, A. J. & Pellegrini, N. A review of recent studies on malondialdehyde as toxic molecule and biological marker of oxidative stress. Nutr. Metab. Cardiovasc. Dis. 15, 316–328. https://doi.org/10.1016/j.numecd.2005.05.003 (2005).
Schmittgen, T. D. & Livak, K. J. Analyzing real-time PCR data by the comparative C(T) method. Nat. Protoc. 3, 1101–1108. https://doi.org/10.1038/nprot.2008.73 (2008).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25, 402–408. https://doi.org/10.1006/meth.2001.1262 (2001).
Kim, S. H., Chung, D. K., Lee, Y. J., Song, C. H. & Ku, S. K. Neuroprotective effects of Danggui-Jakyak-San on rat stroke model through antioxidant/antiapoptotic pathway. J. Ethnopharmacol. 188, 123–133. https://doi.org/10.1016/j.jep.2016.04.060 (2016).
Xu, Q. Q. et al. Sodium tanshinone IIA sulfonate attenuates scopolamine-induced cognitive dysfunctions via improving cholinergic system. BioMed Res. Int. 2016, 9852536. https://doi.org/10.1155/2016/9852536 (2016).
Wirtshafter, D. Cholinergic involvement in the cortical and hippocampal Fos expression induced in the rat by placement in a novel environment. Brain Res. 1051, 57–65. https://doi.org/10.1016/j.brainres.2005.05.052 (2005).
Fast, C. D., Flesher, M. M., Nocera, N. A., Fanselow, M. S. & Blaisdell, A. P. Learning history and cholinergic modulation in the dorsal hippocampus are necessary for rats to infer the status of a hidden event. Hippocampus 26, 804–815. https://doi.org/10.1002/hipo.22564 (2016).
Zhan, C. & Yang, J. Protective effects of isoliquiritigenin in transient middle cerebral artery occlusion-induced focal cerebral ischemia in rats. Pharmacol. Res. 53, 303–309. https://doi.org/10.1016/j.phrs.2005.12.008 (2006).
Sun, Y., Oberley, L. W. & Li, Y. A simple method for clinical assay of superoxide dismutase. Clin. Chem. 34, 497–500 (1988).
Mansoorali, K. P., Prakash, T., Kotresha, D., Prabhu, K. & Rama Rao, N. Cerebroprotective effect of Eclipta alba against global model of cerebral ischemia induced oxidative stress in rats. Phytomedicine 19, 1108–1116. https://doi.org/10.1016/j.phymed.2012.07.004 (2012).
Ghumatkar, P. J., Patil, S. P., Jain, P. D., Tambe, R. M. & Sathaye, S. Nootropic, neuroprotective and neurotrophic effects of phloretin in scopolamine induced amnesia in mice. Pharmacol. Biochem. Behav. 135, 182–191. https://doi.org/10.1016/j.pbb.2015.06.005 (2015).
Kim, T. W. et al. Acceleration-dependent effects of vibrotactile gamma stimulation on cognitive recovery and cholinergic function in a scopolamine-induced neurotoxicity mouse model. Biomedicines https://doi.org/10.3390/biomedicines13082031 (2025).
Leinenga, G. & Gotz, J. Scanning ultrasound removes amyloid-beta and restores memory in an Alzheimer’s disease mouse model. Sci. Transl. Med. 7, 278ra233. https://doi.org/10.1126/scitranslmed.aaa2512 (2015).
Oroszi, T. et al. Whole-body vibration affects hippocampal choline acetyltransferase and synaptophysin expression and improves spatial memory in young adult mice. J. Integr. Neurosci. 23, 173. https://doi.org/10.31083/j.jin2309173 (2024).
Park, S. S. et al. Physical exercise during exposure to 40-Hz light flicker improves cognitive functions in the 3xTg mouse model of Alzheimer’s disease. Alzheimers Res. Ther. 12, 62. https://doi.org/10.1186/s13195-020-00631-4 (2020).
Kong, L. et al. Assessment of vibration modulated regional cerebral blood flow with MRI. Neuroimage 269, 119934. https://doi.org/10.1016/j.neuroimage.2023.119934 (2023).
Seitz, R. J. & Roland, P. E. Vibratory stimulation increases and decreases the regional cerebral blood flow and oxidative metabolism: A positron emission tomography (PET) study. Acta Neurol. Scand. 86, 60–67. https://doi.org/10.1111/j.1600-0404.1992.tb08055.x (1992).
Buzsaki, G. & Wang, X. J. Mechanisms of gamma oscillations. Annu. Rev. Neurosci. 35, 203–225. https://doi.org/10.1146/annurev-neuro-062111-150444 (2012).
Ichim, A. M. et al. The gamma rhythm as a guardian of brain health. Elife https://doi.org/10.7554/eLife.100238 (2024).
Brown, P. Oscillatory nature of human basal ganglia activity: Relationship to the pathophysiology of Parkinson’s disease. Mov. Disord. 18, 357–363. https://doi.org/10.1002/mds.10358 (2003).
Iaccarino, H. F. et al. Gamma frequency entrainment attenuates amyloid load and modifies microglia. Nature 540, 230–235. https://doi.org/10.1038/nature20587 (2016).
Shin, C. H. et al. <article-title update=“added”>Vibrotactile stimulation at 40 Hz inhibits Aβ-induced changes in SH-SY5Y, BV2 cells, and pericytes. Brain Res. Bull. 220, 111138. https://doi.org/10.1016/j.brainresbull.2024.111138 (2025).
Herrmann, C. S. Human EEG responses to 1-100 Hz flicker: Resonance phenomena in visual cortex and their potential correlation to cognitive phenomena. Exp. Brain Res. 137, 346–353. https://doi.org/10.1007/s002210100682 (2001).
Tyler, W. J. The mechanobiology of brain function. Nat. Rev. Neurosci. 13, 867–878. https://doi.org/10.1038/nrn3383 (2012).
Zheng, Q. et al. Mechanical properties of the brain: Focus on the essential role of Piezo1-mediated mechanotransduction in the CNS. Brain Behav. 13, e3136. https://doi.org/10.1002/brb3.3136 (2023).
Park, J., Lee, C., Lee, S. & Im, C. H. 80 Hz but not 40 Hz, transcranial alternating current stimulation of 80 Hz over right intraparietal sulcus increases visuospatial working memory capacity. Sci. Rep. 12, 13762. https://doi.org/10.1038/s41598-022-17965-8 (2022).
Rostami, M. et al. Determining the effects of transcranial alternating current stimulation on corticomotor excitability and motor performance: A sham-controlled comparison of four frequencies. Neuroscience 568, 12–26. https://doi.org/10.1016/j.neuroscience.2025.01.016 (2025).
Merola, A. et al. 80 Hz versus 130 Hz subthalamic nucleus deep brain stimulation: Effects on involuntary movements. Parkinsonism Relat. Disord. 19, 453–456. https://doi.org/10.1016/j.parkreldis.2013.01.006 (2013).
Ricchi, V. et al. Transient effects of 80 Hz stimulation on gait in STN DBS treated PD patients: A 15 months follow-up study. Brain Stimul. 5, 388–392. https://doi.org/10.1016/j.brs.2011.07.001 (2012).
Liu, Y. et al. <article-title update=“added”>Non-invasive auditory and visual stimulation attenuates α-Synuclein deposition and improves motor and non-motor symptoms in PD mice. Exp. Neurol. 364, 114396. https://doi.org/10.1016/j.expneurol.2023.114396 (2023).
Nagarajan, N. & Capecchi, M. R. Optogenetic stimulation of mouse Hoxb8 microglia in specific regions of the brain induces anxiety, grooming, or both. Mol. Psychiatry. 29, 1726–1740. https://doi.org/10.1038/s41380-023-02019-w (2024).
Greenberg, S. M., Bax, F. & van Veluw, S. J. Amyloid-related imaging abnormalities: Manifestations, metrics and mechanisms. Nat. Rev. Neurol. 21, 193–203. https://doi.org/10.1038/s41582-024-01053-8 (2025).
Sohmer, H., Freeman, S., Geal-Dor, M., Adelman, C. & Savion, I. Bone conduction experiments in humans - A fluid pathway from bone to ear. Hear. Res. 146, 81–88. https://doi.org/10.1016/s0378-5955(00)00099-x (2000).
Lee, J. et al. Contralateral bone conducted sound wave propagation on the skull bones in fresh frozen cadaver. Sci. Rep. 13, 7479. https://doi.org/10.1038/s41598-023-32307-y (2023).
Freeman, S., Sichel, J. Y. & Sohmer, H. Bone conduction experiments in animals - Evidence for a non-osseous mechanism. Hear. Res. 146, 72–80. https://doi.org/10.1016/s0378-5955(00)00098-8 (2000).
Jindrak, K. F. & Jindrak, H. Mechanical effect of vocalization on human brain and meninges. Med. Hypotheses 25, 17–20. https://doi.org/10.1016/0306-9877(88)90040-0 (1988).
Kong, L. et al. Transcranial vibration stimulation at 40 Hz induced neural activity and promoted the coupling of global brain activity and cerebrospinal fluid flow. Neuroimage 308, 121074. https://doi.org/10.1016/j.neuroimage.2025.121074 (2025).
Acknowledgements
We express our sincere gratitude to all researchers and technical staff who contributed to this study. We also thank the personnel involved in animal care and data collection for their dedicated efforts throughout the experimental procedures. ESC was supported by the Chung-Ang University Graduate Research Scholarship in 2024.
Author information
Authors and Affiliations
Contributions
O-HK: Investigation, Data curation, Validation, Writing—original draft, Writing—review and editing.C-HS: Conceptualization, Methodology, Validation, Writing—original draft, Writing—review and editing.M-WC: Methodology, Resources.J-YH: Resources, Project administration.JJC: Conceptualization, Supervision.D-KS: Conceptualization, Data curation, Validation, Writing—review and editing.J-YC: Methodology, Resources.ESC: Resources, Writing—review and editing.HJL: Conceptualization, Supervision, Writing—review and editing.S-KK: Conceptualization, Methodology, Data curation, Formal analysis, Writing—original draft.
Corresponding authors
Ethics declarations
Ethic approval
All animal care and experimental procedures have been approved by the Institutional Animal Care and Use Committee of Daegu Haany University (Approval No. DHU2023-049).
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kim, OH., Shin, CH., Cho, MW. et al. Transcranial vibrotactile stimulation enhances hippocampal cholinergic signaling and memory through frequency-dependent mechanotransduction. Sci Rep (2026). https://doi.org/10.1038/s41598-026-49377-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-49377-3