Abstract
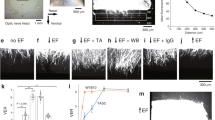
Electric field (EF) stimulation is an emerging neuromodulatory strategy for promoting the repair and functional recovery of degenerated neural networks in neurodegenerative conditions such as glaucoma. EF stimulation therapeutic potential is thought to arise, in part, from modulation of calcium-dependent signaling pathways that regulate neuronal survival and plasticity. However, despite extensive use of EF stimulation in retinal research and clinical studies, it remains unclear how EF waveform frequency and shape govern population-level intracellular calcium dynamics in retinal ganglion cells (RGCs), limiting the rational design of stimulation protocols. Here we address this gap by combining large-scale ex-vivo calcium imaging of Thy1-GCaMP6f mouse retinas with controlled EF stimulation spanning a wide frequency range and a uniform, non-contact stimulation geometry. This approach enables direct measurement of intracellular calcium responses across thousands of individual RGCs under stimulation conditions relevant to non-invasive and translational paradigms. We further develop a morphologically detailed RGC model in NEURON incorporating reaction-diffusion calcium dynamics and admittance-based extracellular stimulation to mechanistically interpret the frequency-dependent responses observed under sinusoidal EF stimulation. Using this integrated experimental–computational framework, we reveal how electric field stimulation modulates population-level calcium signaling in retinal ganglion cells, enabling simultaneous characterization of spatial response patterns and ensemble-averaged activity across thousands of cells. At this scale, RGC calcium responses are constrained to a distinct frequency regime: low frequencies (below 5 Hz) evoke oscillatory transients, intermediate frequencies (10–100 Hz) produce sustained calcium elevation across the population, and high-frequency stimulation (>3 kHz) leads to a sharp attenuation of calcium responses. Among all tested waveforms, a 1:4 asymmetric charge-balanced stimulus at 50 Hz most effectively and consistently elevated intracellular calcium across the RGC population. The computational model reproduces the experimentally observed frequency dependence for sinusoidal stimulation and reveals that the behavior of these different frequency regimes emerges from the interplay between calcium influx, calcium-activated potassium feedback, calcium extrusion kinetics, and soma geometry. Beyond these findings, this work delivers, to our knowledge, the first large-scale dataset of single-cell calcium responses from RGC populations exposed to diverse EF waveforms and frequencies. This dataset enables future data-driven and hybrid modeling approaches that require rich mappings between extracellular stimulation parameters and intracellular calcium dynamics, and establishes a foundation for systematic, physiology-informed optimization of EF stimulation strategies targeting retinal neurodegenerative disease.
Similar content being viewed by others
Funding
KKG and GL were supported by grants from NEI/NIH (R01EY035375) and NSF (2121164). KKG was also supported by a grant from the Research to Prevent Blindness Foundation Disney Award for Amblyopia. OS was supported by a grant from the NSF (2121164). This work was also supported by an unrestricted grant to the Department of Ophthalmology from Research to Prevent Blindness and the NEI (P30EY029220). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. No funding sponsors were involved in the study design, collection, analysis, interpretation of data, writing of the report, or decision to submit the article for publication.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sharafi, O., Silliman, T., Mokhtari Dowlatabad, H. et al. Population-scale analysis of frequency-dependent calcium dynamics in retinal ganglion cells under electric field stimulation. Sci Rep (2026). https://doi.org/10.1038/s41598-026-49531-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-49531-x