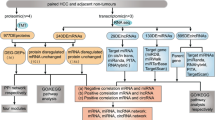
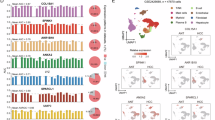
Abstract
Hepatocellular carcinoma represents one of the most common and fatal malignancies worldwide, with early diagnosis being critical for enhancing patient survival rates. Traditional tumor biomarkers have limitations, as serum biomarker levels remain normal in certain patients, thus constraining diagnostic accuracy. Previous researches have elucidated that tRNA-derived small RNAs (tsRNAs) exert a critical function as regulators in the pathogenesis of malignant tumors. Nevertheless, the clinical relevance of circulating tsRNAs in HCC remains largely unclear. Consequently, this research aimed to examine the possible application of a newly identified tsRNA (tRF-34-P4R8YP9LON4VHM) for auxiliary diagnosis, disease surveillance, and prognostic evaluation of HCC. tRF-34-P4R8YP9LON4VHM was detected through high-throughput sequencing, and its serum concentration was quantified using qRT-PCR. Methodological evaluation demonstrated that qRT-PCR for detecting serum tRF-34-P4R8YP9LON4VHM exhibited excellent stability, reproducibility, and specificity. Serum level of this biomarker were markedly upregulated in HCC patients compared to hepatitis patients and the healthy, and it decreased significantly following surgical resection. Moreover, elevated serum level was strongly associated with advanced TNM stage, lymph node metastasis, and tumor differentiation. Our data suggest that serum tRF-34-P4R8YP9LON4VHM is a promising auxiliary diagnostic biomarker for HCC and may further function as a valuable indicator for disease monitoring and prognostic stratification.
Similar content being viewed by others
Acknowledgements
We appreciate all the patients who participated in this study and all those who contributed to it.
Funding
This work was financially supported by the Nantong City Natural Science Foundation (JC2024013) and the Jiangsu Province Graduate Innovation Training Program (S202510304160).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The study protocol was approved by the Clinical Research Ethics Committee of the First Affiliated Hospital of Nantong University.
Consent for publication
The informed consent obtained from study participants.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Guo, Y., Huang, M., Lu, Y. et al. tRF-34-P4R8YP9LON4VHM may serve as a promising potential diagnostic biomarker for hepatocellular carcinoma. Sci Rep (2026). https://doi.org/10.1038/s41598-026-49634-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-49634-5