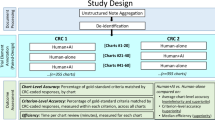
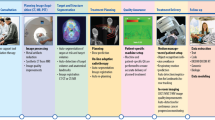
Abstract
Clinical trial enrollment in oncology remains limited by increasingly complex eligibility criteria, biomarker stratification, and fragmented clinical data, contributing to prolonged recruitment timelines and low participation rates. This review examines contemporary pre-screening and screening approaches, spanning manual workflows, health-system–embedded digital tools, and emerging artificial intelligence–enabled methods. We assess the relative strengths and limitations of large language model–based strategies, including retrieval-augmented and domain-adapted approaches, in addressing scalability, accuracy, and equity challenges. Hybrid frameworks that integrate automated screening with clinician oversight appear most effective in improving trial matching efficiency, representativeness, and timely access to investigational therapies across diverse oncology populations.
Similar content being viewed by others
Data Availability
No datasets were generated or analyzed during the current study.
References
Subbiah, V. et al. The best management for most patients with incurable cancer is on a clinical trial. Ann. Oncol. 36, 240–243 (2025).
Fogel, D. B. Factors associated with clinical trials that fail and opportunities for improving the likelihood of success: a review. Contemp. Clin. Trials Commun. 11, 156–164 (2018).
IQVIA institute for human data science. rethinking clinical trial country prioritization: enabling agility through global diversification, July 2024. Available from www.iqviainstitute.org.
Sedrak, M. inaS. et al. Physician perceptions of the use of social media for recruitment of patients in cancer clinical trials. JAMA Netw. Open 2, e1911528–e1911528 (2019).
Unger, J. M. et al. The role of clinical trial participation in cancer research: barriers, evidence, and strategies. Am. Soc. Clin. Oncol. Educ. Book 35, 185–198 (2016).
NCCN Guideline. Available Online: https://www.nccn.org/guidelines/category_1 (Accessed on July 19th 2024).
Beck, J. T. et al. Artificial intelligence tool for optimizing eligibility screening for clinical trials in a large community cancer center. JCO Clin. Cancer Inform. 4, 50–59 (2020).
ClinicalTrial.gov. Available Online: clinicaltrials.gov. (Accessed on July 19th 2024).
Schwaederle M., et al. Molecular tumor board: the University of California-San Diego Moores Cancer Center experience. Oncologist 19(6):631-636 (2024).
FDA (2024). Diversity Action Plans to Improve Enrollment of Participants from Underrepresented Populations in Clinical Studies. Available Online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/diversity-action-plans-improve-enrollment-participants-underrepresented-populations-clinical-studies (Accessed on July 19th 2024).
Diversity action plans to improve enrollment of participants from underrepresented populations in clinical studies: guidance for industry. https://www.fda.gov/media/179593/download Accessed Aug 24, 2024.
Beck et al. Artificial intelligence tool for optimizing eligibility screening for clinical trials in a large community cancer center. JCO Clin. Cancer Inform. 4, 50–59 (2020).
Ismail, A. bdalah, Talha Al-Zoubi, I. ssam, El Naqa & Saeed, H. ina The role of artificial intelligence in hastening time to recruitment in clinical trials. BJR Open 5, 20220023 (2023).
Von Itzstein, M. S. et al. Application of information technology to clinical trial evaluation and enrollment: a review. JAMA Oncol. 7(10), 1559–1566 (2021).
Kurnaz, S. et al. AI-enabled trial matching: Transforming patient recruitment using real-world data. J. Clin. Oncol. 42(16_suppl), e13501 (2024).
Kurnaz, S. elin et al. SYNERGY-AI: Artificial intelligence-based precision oncology clinical trial matching and registry. JGO 5, 22–22 (2019).
SYNERGY-AI registry. Available Online: https://clinicaltrials.gov/study/NCT03452774 (Accessed on July 19th 2024).
Wornow, Michael, et al. Zero-shot clinical trial patient matching with LLMs. NEJM AI, https://doi.org/10.1056/aics2400360 (December 2024).
K. K. Y. Ng, I. Matsuba, and P. C. Zhang. RAG in health care: a novel framework for improving communication and decision-making by addressing LLM limitations. NEJM AI, https://doi.org/10.1056/AIra2400380 (December 2024).
Kurnaz, S. elin et al. Effect of a novel artificial intelligence (AI) –enabled multi-trial matching system on patient matching using real-world data. JCO 42, e13501–e13501 (2024).
Loaiza-Bonilla A., et al. Driving knowledge to action: building a better future with artificial intelligence-enabled multidisciplinary oncology. Am. Soc. Clin. Oncol. Educ. Book. 45, e100048 (2025).
Loaiza-Bonilla, A. et al. Transforming oncology clinical trial matching through multi-agent AI and an oncology-specific knowledge graph: a prospective evaluation in 3800 patients. JCO 43, 1554–1554 (2025).
Leyfman, Y. et al. Performance evaluation of an AI-powered system for clinical trial eligibility using mCODE data standards. JCO 43, e13621–e13621 (2025).
Loaiza-Bonilla, A., Penberthy Scott. Harnessing Moravec’s Paradox in health care: a new era of collaborative intelligence. NEJM AI. 2, https://doi.org/10.1056/AIp2500005 2025.
Subbiah, V., Burris, H. A. 3rd & Kurzrock, R. Revolutionizing cancer drug development: harnessing the potential of basket trials. Cancer 130, 186–200 (2024).
Subbiah, V. et al. Dabrafenib plus trametinib in BRAFV600E-mutated rare cancers: the phase 2 ROAR trial. Nat. Med. 29, 1103–1112 (2023).
D’Angelo, et al. Afamitresgene autoleucel for advanced synovial sarcoma and myxoid round cell liposarcoma (SPEARHEAD-1): an international, open-label, phase 2 trial. Lancet 403, 1460–1471 (2024).
Author information
Authors and Affiliations
Contributions
The authors have competing interests as defined by Nature Portfolio, or other interests that might be perceived to influence the results and/or discussion reported in this paper: D.H.: affiliated with the European Alliance for Personalized Medicine and Sam Higginbottom University of Agriculture, Technology and Sciences. Declaration: no competing financial or non-financial interests have been declared. J.P.: employed by Exelixis, a biopharmaceutical company involved in oncology therapeutics. Declaration: May have financial interests related to the development or commercialization of oncology treatments. A.L.B.: affiliated with Massive Bio and St. Luke’s University Health Network/Lewis Katz School of Medicine at Temple University. Declaration: may have financial interests related to the development of AI-driven solutions or other proprietary technologies in oncology. Consulting fees from Guardant Health, AstraZeneca, Ipsen, Medscape, stock in BioLineRx. Patents filed: "Neurosymbolic architectures with dynamic knowledge graph feedback for real time clinical trial matching". "Systems and Methods for Real Time Individualized Drug Utilization Optimization Using Causal Machine Learning, Dynamic Knowledge Graphs, and Digital Twin Patient Modeling". "Systems and Methods for Dynamic Patient Journey Mapping Using Multimodal Data Vectors, Neurosymbolic Knowledge Graphs, and Digital Twin Simulation for Real Time Trial Matching". "MEMORY-EFFICIENT POSITIVE GEOMETRY LAYERS FOR PREDICTIVE MODELS" C.S.: affiliated with N-Power Medicine, which focuses on digital health solutions. Declaration: May have non-financial interests linked to advancing digital or AI-based clinical trial matching tools. U.M.: affiliated with the University Federico II of Naples. Declaration: No competing financial or non-financial interests have been declared. F.P.L.: affiliated with Center Jean Perrin and Université d’Auvergne. Declaration: no competing financial or non-financial interests have been declared. H.M.A.R.: affiliated with Qatar Cancer Society.Declaration: no competing financial or non-financial interests have been declared. P.H.: affiliated with IHU RespirERA, FHU OncoAge, and related clinical research institutions. Declaration: no competing financial or non-financial interests have been declared. S.K.: affiliated with HITLAB at Columbia University, a center focused on healthcare innovation and technology. Declaration: no competing financial or non-financial interests have been declared. D.S.: employed by Johnson and Johnson, a major multinational healthcare company. Declaration: may have financial interests related to products or technologies in oncology that could be perceived as relevant to this work. V.S.: affiliated with the Sarah Cannon Research Institute, which is involved in clinical research and trial operations. Declaration: may have financial interests associated with clinical trial management or related services in oncology. Note: the above statements are intended to provide transparency regarding potential competing interests. Each author confirms that they have reviewed these declarations and, where applicable, will update the journal with any additional relevant details or clarifications.
Corresponding author
Ethics declarations
Competing interests
D.H. (Denis Horgan):Affiliated with the European Alliance for Personalized Medicine and Sam Higginbottom University of Agriculture, Technology and Sciences.Declaration: No competing financial or non-financial interests have been declared.J.P. (Joe Paulson):Employed by Exelixis, a biopharmaceutical company involved in oncology therapeutics.Declaration: May have financial interests related to the development or commercialization of oncology treatments.A.L.B. (Arturo Loaiza-Bonilla):Affiliated with Massive Bio and St. Luke’s University Health Network / Lewis Katz School of Medicine at Temple University.Declaration: May have financial interests related to the development of AI-driven solutions or other proprietary technologies in oncology. Consulting fees from Guardant Health, AstraZeneca, Ipsen, Medscape, stock in BioLineRx. Patents filed: “Neurosymbolic architectures with dynamic knowledge graph feedback for real time clinical trial matching". “Systems and Methods for Real Time Individualized Drug Utilization Optimization Using Causal Machine Learning, Dynamic Knowledge Graphs, and Digital Twin Patient Modeling". “Systems and Methods for Dynamic Patient Journey Mapping Using Multimodal Data Vectors, Neurosymbolic Knowledge Graphs, and Digital Twin Simulation for Real Time Trial Matching". “MEMORY-EFFICIENT POSITIVE GEOMETRY LAYERS FOR PREDICTIVE MODELS"C.S. (Christer Svedman):Affiliated with N-Power Medicine, which focuses on digital health solutions.Declaration: May have non-financial interests linked to advancing digital or AI-based clinical trial matching tools.U.M. (Umberto Malapelle):Affiliated with the University Federico II of Naples.Declaration: No competing financial or non-financial interests have been declared.F.P.L. (Frédérique Penault Lorca):Affiliated with Center Jean Perrin and Université d’Auvergne.Declaration: No competing financial or non-financial interests have been declared.H.M.A.R. (Hadi Mohamad Abu Rahsheed):Affiliated with Qatar Cancer Society.Declaration: No competing financial or non-financial interests have been declared.P.H. (Paul Hofman):Affiliated with IHU RespirERA, FHU OncoAge, and related clinical research institutions.Declaration: No competing financial or non-financial interests have been declared.S.K. (Stan Kachnowsk):Affiliated with HITLAB at Columbia University, a center focused on healthcare innovation and technology.Declaration: No competing financial or non-financial interests have been declared.D.S. (Daniel Schneider):Employed by Johnson and Johnson, a major multinational healthcare company.Declaration: May have financial interests related to products or technologies in oncology that could be perceived as relevant to this work.V.S. (Vivek Subbiah):Affiliated with the Sarah Cannon Research Institute, which is involved in clinical research and trial operations.Declaration: May have financial interests associated with clinical trial management or related services in oncology.Note: The above statements are intended to provide transparency regarding potential competing interests. Each author confirms that they have reviewed these declarations and, where applicable, will update the journal with any additional relevant details or clarifications.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Horgan, D., Paulson, J., Loaiza-Bonilla, A. et al. A unified framework for pre-screening and screening tools in oncology clinical trials. npj Precis. Onc. (2026). https://doi.org/10.1038/s41698-026-01306-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41698-026-01306-3