Abstract
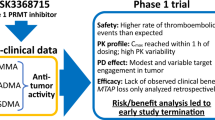
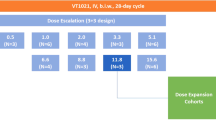
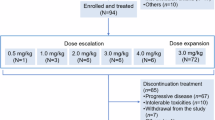
BBP-398 is a selective allosteric SHP2 inhibitor designed to inhibit mitogen-activated protein kinase (MAPK) pathway–driven tumors. We performed the first-in-human phase 1 trial described herein to assess the safety, tolerability, pharmacokinetics, and preliminary efficacy of BBP-398 in patients with advanced solid tumors harboring MAPK pathway mutations. Once-daily BBP-398 was administered at 350-550 mg in the dose-escalation phase (1a; n = 35) followed by a dose-expansion phase (1b; n = 37). The study endpoints were dose-limiting toxicities, treatment-emergent adverse events, pharmacokinetics, target engagement, disease control rate, progression-free survival, and overall survival. In phase 1a, 26% of the 23 evaluable patients had stable disease, with a median progression-free survival duration of 1.8 months (range, 1.7-4.1 months). In phase 1b, 30% of the 27 evaluable patients had stable disease (31% at 350 mg, 27% at 450 mg), with median progression-free survival of 2.2 months and 1.9 months at 350 mg and 450 mg, respectively. We halted dose escalation at 550 mg owing to an increased rate of thrombocytopenia and edema. At daily doses of up to 450 mg, BBP-398 exhibited an acceptable safety profile and produced disease stabilization in nearly 30% of heavily pretreated patients.
Similar content being viewed by others
Data availability
Anonymized individual participant data on completed studies and applicable supporting clinical study documents are available upon request in a secured access environment. Requests for access to data can be submitted to the corresponding author. Access will be provided contingent upon the approval of a research proposal and the execution of a data sharing agreement.
References
Cox, A. D., Fesik, S. W., Kimmelman, A. C., Luo, J. & Der, C. J. Drugging the undruggable RAS: Mission Possible?. Nat. Rev. Drug Discov. 13, 828–851 (2014).
Bunda, S. et al. Inhibition of SHP2-mediated dephosphorylation of Ras suppresses oncogenesis. Nat. Commun. 6, 8859 (2015).
Dhillon, A. S., Hagan, S., Rath, O. & Kolch, W. MAP kinase signalling pathways in cancer. Oncogene 26, 3279–3290 (2007).
Gschwind, A., Fischer, O. M. & Ullrich, A. The discovery of receptor tyrosine kinases: targets for cancer therapy. Nat. Rev. Cancer 4, 361–370 (2004).
Barford, D. & Neel, B. G. Revealing mechanisms for SH2 domain mediated regulation of the protein tyrosine phosphatase SHP-2. Structure 6, 249–254 (1998).
Mullard, A. Phosphatases start shedding their stigma of undruggability. Nat. Rev. Drug Discov. 17, 847–849 (2018).
Kerr, D. L., Haderk, F. & Bivona, T. G. Allosteric SHP2 inhibitors in cancer: Targeting the intersection of RAS, resistance, and the immune microenvironment. Curr. Opin. Chem. Biol. 62, 1–12 (2021).
Song, Z. et al. Tyrosine phosphatase SHP2 inhibitors in tumor-targeted therapies. Acta Pharm. Sin. B 11, 13–29 (2021).
Ran, H., Tsutsumi, R., Araki, T. & Neel, B. G. Sticking It to Cancer with Molecular Glue for SHP2. Cancer Cell 30, 194–196 (2016).
Chen, Y.-N. P. et al. Allosteric inhibition of SHP2 phosphatase inhibits cancers driven by receptor tyrosine kinases. Nature 535, 148–152 (2016).
Brana, I. et al. Initial results from a dose finding study of TNO155, a SHP2 inhibitor, in adults with advanced solid tumors. J. Clin. Oncol. 39, 3005 (2021).
Ou, S. I. et al. A12 The SHP2 Inhibitor RMC-4630 in Patients with KRAS-Mutant Non-Small Cell Lung Cancer: Preliminary Evaluation of a First-in-Man Phase 1 Clinical Trial. J. Thorac. Oncol. 15, S15–S16 (2020).
Stice, J. P. et al. Abstract P207: BBP-398, a potent, small molecule inhibitor of SHP2, enhances the response of established NSCLC xenografts to KRASG12C and mutEGFR inhibitors. Mol. Cancer Ther. 20, P207 (2021).
Liu, C. et al. Combinations with Allosteric SHP2 Inhibitor TNO155 to Block Receptor Tyrosine Kinase Signaling. Clin. Cancer Res. 27, 342–354 (2021).
Drilon, A. E. et al. A first-in-human, phase 1 study of the SHP2 inhibitor PF-07284892 as monotherapy and in combination with different targeted therapies in oncogene-driven, treatment-resistant solid tumors. J. Clin. Oncol. 41, 3020 (2023).
McKean, M. et al. Preliminary results from FLAGSHP-1: A Phase I dose escalation study of ERAS-601, a potent SHP2 inhibitor, in patients with previously treated advanced or metastatic solid tumors. Eur. J. Cancer 174, S34 (2022).
Schreck, K. C., Grossman, S. A. & Pratilas, C. A. BRAF Mutations and the Utility of RAF and MEK Inhibitors in Primary Brain Tumors. Cancers 11, 1262 (2019).
Özgü, E. et al. Therapeutic vulnerabilities and pan-cancer landscape of BRAF class III mutations in epithelial solid tumors. BJC Rep. 2, (2024).
Wang, Y. et al. SHP2 blockade enhances anti-tumor immunity via tumor cell intrinsic and extrinsic mechanisms. Sci. Rep. 11, 1399 (2021).
Zhao, M. et al. SHP2 inhibition triggers anti-tumor immunity and synergizes with PD-1 blockade. Acta Pharm. Sin. B 9, 304–315 (2019).
Lopes, C. D. H., Braganca Xavier, C., Torrado, C., Veneziani, A. C. & Megid, T. B. C. A Comprehensive Exploration of Agents Targeting Tumor Microenvironment: Challenges and Future Perspectives. J. Immunother. Precis. Oncol. 7, 283–299 (2024).
Acknowledgements
The authors express their sincere gratitude to the patients and their families for their participation in this study and to all investigators involved, particularly Dr. Aditya Sarvaria, for their invaluable efforts in enrolling patients onto the protocol. We also thank the Editing Services, Research Medical Library from The University of Texas MD Anderson Cancer Center for their for their support revising the final version of this manuscript.
Author information
Authors and Affiliations
Contributions
D.V.V. and D.S.H. conceptualized the work: E.L. performed statistical data analyses. All authors revised the provided results. C.B.X. wrote the initial draft. All authors reviewed the final manuscript.
Corresponding author
Ethics declarations
Competing interests
G.F. has received royalties from Wolters Kluwer; been an advisor for AbbVie, Jubilant, BostonGene, Teon, Merck, Sanofi, BridgeBio, and Beijing Avistone (2024); received a speaker honorarium from Clinical Care Options; received travel funds from Sarah Cannon Research Institute, Amgen, Synthorx/Sanofi, GSK, and Cyteir; and received research funding from Abbisko, ABL Bio, Abbvie, ADC Therapeutics, Accutar, Agenus, Aileron, Alterome, Amgen, Arcus, ARMO/Eli Lilly, Artios, Astellas, AstraZeneca, Bayer, BeiGene, Beijing Avistone, Bioatla, Bioinvent, Biomea Fusion, Bicycle, Black Diamond, Boehringer Ingelheim, Boundless, Centessa, Conjupro, Cyteir, Cytomx, D3 Bio, Daiichi, Deciphera, Dynamicure, Eikon, Eli Lilly, Epizyme, Erasca, Exelixis, Freenome, Fujifilm, GSK, Harbour BioMed, Hutchison MediPharma, IGM Biosciences, IDEAYA, Ikena, Immuneering, Immunitas, ImmunoGen/MacroGenics, Incyte, Jacobio, Jazz, Jounce, Jubilant, Kineta, Kumquat, Kura, Loxo/Bayer, Medilink, Merck, Metabomed, Mirati, ModeX, Molecular Templates, Nammi, Navire/BridgeBio, NGM Bio, NiKang, Novartis, Nuvalent, Nuvectis, Oncorus, Oncusp, OnKure, Phanes, Poseida, Prelude, PureTech, Pyramid, Pyxis, Quanta, RasCal, Regeneron, Relay, Rgenix, Ribon, Roche, Samumed, Sapience, Sarah Cannon Development Innovations, Seagen, Silicon/Stingthera, Simcha, Sirnaomics, Synthorx/Sanofi, Tachyon, Takeda, Tallac, Tango, Tarus, Tarveda, Teneobio, Tesaro, TORL, Turning Point, Xencor, and Zhuhai Yufan. D.V.V. has been employed by Navire Pharma. J.M. has been an advisor for Astra Zeneca, Janssen, Abbvie, Jazz, Sanofi, Bristol Myers Squibb, Takeda, and Daiichi Sankyo; been a consultant for Regeneron and Bristol Myers Squibb; been a Data Safety and Monitoring Committee member for Bioatla; and received research funding from NCI/ARPA-H and Astra Zeneca. S.S. has been a consultant and/or served on the speaker bureau for Eisai and BMS. A.R.K. has received grants and/or personal fees from Pfizer, AstraZeneca, Bristol Myers Squibb, EMD Serono, Exelixis, Genentech, Gilead Sciences, Immunomedics, Novartis, Seattle Genetics/Astellas, Amgen, Astellas Medivation, AVEO, Eisai, Genentech/Roche, Janssen, Merck, Myovant Sciences, Sanofi, Bayer, Arvinas, Mirati Therapeutics, and POINT Biopharma and has stock and other ownership interests in ECOM Medical. L.W. has been employed by Navire Pharma. F.R. has received consulting fees from Eikon Therapeutics. E.L. has received consulting fees from Navire Pharma for performing the data analyses and summaries for the present study, which may be considered a potential conflict of interest with respect to the subject matter of this manuscript. A.I.S. has been a consultant or advisor for Incyte, Amgen, Novartis, Mirati Therapeutics, Jazz Pharmaceuticals, Takeda, Janssen Research & Development, Mersana, Gritstone bio, Daiichi Sankyo/AstraZeneca, Regeneron, Eli Lilly, Black Diamond Therapeutics, Sanofi, ArriVent Biopharma, Synthekine, GSK, Crispr Therapeutics, and Revolution Medicines and has received research funding from LAM Therapeutics, Roche, AstraZeneca, Boehringer Ingelheim, Astellas Pharma, MedImmune, Novartis, Incyte, AbbVie, Ignyta, Takeda, Macrogenics, CytomX Therapeutics, Astex Pharmaceuticals, Bristol-Myers Squibb, Loxo, Gritstone bio, Plexxikon, Amgen, Daiichi Sankyo, ADC Therapeutics, Janssen Oncology, Rubius, Synthekine, Mersana, Blueprint Medicines, Regeneron, Alkermes, Revolution Medicines, Medikine, Black Diamond Therapeutics, BluPrint Oncology, Nalo Therapeutics, Scorpion Therapeutics, ArriVent Biopharma, Prelude Therapeutics, and Eli Lilly. D.S. has been an employee and shareholder of Texas Oncology, a shareholder of NEXT Oncology, received honoraria from Syneos, received consulting fees from Guidepoint, received advisory board payments from Revolution Medicines and Nimbus Therapeutics, and received ongoing or past institutional research funding for studies from Abbvie, Acrivon Therapeutics, ADC Therapeutics, Aprea Therapeutics, Ascentage Pharma Group, Astellas, Avenzo Therapeutics, Biomea Fusion, Boehringer Ingelheim, BJ Bioscience, BioNTech, Bristol Myers Squibb, Compugen, Day One Biopharma, Dicerna/Novo Nordisk, Dren Bio, Exelixis, Fate Therapeutics, Gilead Sciences, GSK, Haihe Pharmaceutical, Iconovir Bio, Ideaya Biosciences, Immuneering, Impact Therapeutics, Incendia, Kura Oncology, MediLink Therapeutics, Mirati Therapeutics, ModeX Therapeutics, Monopteros Therapeutics, Navire Pharma, Nimbus Therapeutics, NGM Biopharmaceuticals, OBI Pharma, OncoResponse, Pfizer, Revolution Medicines, Step Pharmaceuticals, Symphogen, Tachyon Therapeutics, Teon Therapeutics, Tyligand Bioscience, Vincerx Pharma, Vividion Therapeutics, ZielBio, and Zymeworks. I.G.-L. has been a consultant or advisor for SOTIO, AbbVie, Revolution Medicines, Eli Lilly, and Quanta Therapeutics and received research funding from EcoR1, Guidepoint, Novartis, Bayer, Bristol Myers Squibb, Pfizer, MedImmune, Eli Lilly, Incyte, GSK, Tolero Pharmaceuticals, BridgeBio Pharma, Jacobio, Repare Therapeutics, Sumitomo Dainippon Pharma Oncology, Revolution Medicines, Yingli Pharma, Quanta Therapeutics, 280 BIO, ABM Therapeutics, and Tango. D.S.H. has been a consultant or advisor for AbbVie, Acuta, Alpha Insights, Amgen, Axiom, BeiGene, Boxer Capital, COR2ed, EcoR1, Erasca, GLG, Guidepoint, ImmunoGen, Kestrel Therapeutics, Medscape, Mirati Therapeutics, Pfizer, Revolution Medicines, T-Knife, and WebMD; received research funding from AbbVie, Adaptimmune, Adlai-Nortye, Amgen, Astellas, AstraZeneca, Bayer, BeiGene USA, Bristol Myers Squibb, Eisai, Eli Lilly, Endeavor, Erasca, Exelixis, F. Hoffmann-La Roche, Genentech, ImmunoGen, Merck, Mirati, NCI-CTEP, Novartis, Pfizer, Revolution Medicines, STCube, TCR2, and VM Oncology; received for travel, accommodations, and expenses from the American Association for Cancer Research, ImmunoGen, Medscape, and Telperian; and has had other ownership interests in Molecular Match, OncoResponse, and Telperian. The other authors (C.B.X., M.K., and Y.P.) do not have a competing interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Falchook, G., Braganca Xavier, C., Van Veenhuyzen, D. et al. A first-in-human phase 1 study of the SHP2 inhibitor BBP-398 in patients with advanced solid tumors. npj Precis. Onc. (2026). https://doi.org/10.1038/s41698-026-01340-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41698-026-01340-1