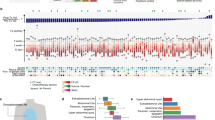
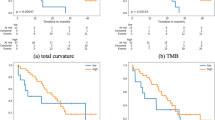
Abstract
Although neoadjuvant chemotherapy (NACT) is commonly used for advanced ovarian cancer, patient outcomes vary substantially. We developed a graph convolutional network (GCN) that integrates patient-specific baseline clinical variables and computed tomography–derived radiomic features while modeling inter-patient relationships to improve outcome prediction beyond standard models. The GCN operates without reliance on high-performance computing resources and predicts long-term overall survival (OS) while stratifying short-term surgical outcomes (R0 resection). The GCN was compared with the CA-125 ELIMination rate constant K (KELIM) score and three Cox-based comparator models. Model performance was evaluated using the concordance index (C-index) for OS, area under the receiver operating characteristic curve for 3-year OS, Kaplan–Meier survival analysis, and R0 resection stratification. The GCN demonstrated strong OS prognosis performance (C-index = 0.73, 0.72, and 0.70 across the training and two external test datasets), stratified surgical outcomes, and identified 16.30% of patients with low KELIM scores but favorable survival.
Similar content being viewed by others
Data availability
The datasets used and analyzed in this study are available from the corresponding author upon reasonable request.
Code availability
The code related to this study has been released on a GitHub repository (https://github.com/jessiezhang1021/GCN-OC).
References
Cancer stat facts: ovarian cancer. https://seer.cancer.gov/statfacts/html/ovary.html (2025).
Siegel, R. L., Giaquinto, A. N. & Jemal, A. Cancer statistics, 2024. CA Cancer J. Clin. 74, 12–49 (2024).
Gaillard, S. et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: ASCO guideline update. J. Clin. Oncol. 43, 868–891 (2025).
Bercow, A. et al. Utilization of primary cytoreductive surgery for advanced-stage ovarian cancer. JAMA Netw. Open 7, e2439893 (2024).
Vergote, I. et al. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N. Engl. J. Med. 363, 943–953 (2010).
Coada, C. A. et al. Optimal number of neoadjuvant chemotherapy cycles prior to interval debulking surgery in advanced epithelial ovarian cancer: a systematic review and meta-analysis of progression-free survival and overall survival. J. Gynecol. Oncol. 34, e82 (2023).
Cho, J. H., Kim, S. & Song, Y. S. Neoadjuvant chemotherapy in advanced ovarian cancer: optimal patient selection and response evaluation. Chin. Clin. Oncol. 7, 58 (2018).
Cioffi, R. et al. Neoadjuvant chemotherapy in high-risk ovarian cancer patients: role of age. Tumori 105, 168–173 (2019).
Tajik, P. et al. The FIGO stage IVA versus IVB of ovarian cancer: prognostic value and predictive value for neoadjuvant chemotherapy. Int. J. Gynecol. Cancer 28, 453–458 (2018).
Fagotti, A. et al. Randomized trial of primary debulking surgery versus neoadjuvant chemotherapy for advanced epithelial ovarian cancer (SCORPION-NCT01461850). Int. J. Gynecol. Cancer 30, 1657–1664 (2020).
Bryant, A. et al. Impact of residual disease as a prognostic factor for survival in women with advanced epithelial ovarian cancer after primary surgery. Cochrane Database Syst. Rev. 9, CD015048 (2022).
You, B. et al. CA-125 ELIMination rate constant K (KELIM) is a marker of chemosensitivity in patients with ovarian cancer: results from the phase II CHIVA trial. Clin. Cancer Res. 26, 4625–4632 (2020).
Kessous, R. et al. CA-125 reduction during neoadjuvant chemotherapy is associated with success of cytoreductive surgery and outcome of patients with advanced high-grade ovarian cancer. Acta Obstet. Gynecol. Scand. 99, 933–940 (2020).
Liontos, M. et al. Neutrophil-to-lymphocyte ratio and chemotherapy response score as prognostic markers in ovarian cancer patients treated with neoadjuvant chemotherapy. J. Ovarian Res. 14, 148 (2021).
Tuntinarawat, P., Tangmanomana, R. & Kittisiam, T. Association between alteration of neutrophil to lymphocyte ratio, platelet to lymphocyte ratio, cancer antigen-125 and surgical outcomes in advanced stage ovarian cancer patient who received neoadjuvant chemotherapy. Gynecol. Oncol. Rep. 52, 101347 (2024).
Wood, N. et al. Association between CT-based body composition assessment and patient outcomes during neoadjuvant chemotherapy for epithelial ovarian cancer. Gynecol. Oncol. 169, 55–63 (2023).
Mao, N. et al. A multimodal and fully automated system for prediction of pathological complete response to neoadjuvant chemotherapy in breast cancer. Sci. Adv. 11, eadr1576 (2025).
Schutte, K. et al. An artificial intelligence model predicts the survival of solid tumour patients from imaging and clinical data. Eur. J. Cancer 174, 90–98 (2022).
Chen, S. et al. Deep learning-based multi-model prediction for disease-free survival status of patients with clear cell renal cell carcinoma after surgery: a multicenter cohort study. Int. J. Surg. 110, 2970–2977 (2024).
Crispin-Ortuzar, M. et al. Integrated radiogenomics models predict response to neoadjuvant chemotherapy in high grade serous ovarian cancer. Nat. Commun. 14, 6756 (2023).
Suidan, R. S. et al. A multicenter assessment of the ability of preoperative computed tomography scan and CA-125 to predict gross residual disease at primary debulking for advanced epithelial ovarian cancer. Gynecol. Oncol. 145, 27–31 (2017).
Yin, R. et al. Predicting neoadjuvant chemotherapy response and high-grade serous ovarian cancer from CT images in ovarian cancer with multitask deep learning: a multicenter study. Acad. Radiol. 30, S192–S201 (2023).
AlSomairi, A., Himayda, S., Altelmesani, A., Lee, Y. J. & Lee, J. Y. Prognostic value of HE4 in advanced-stage, high-grade serous ovarian cancer: analysis of HE4 kinetics during NACT, predicting surgical outcome and recurrence in comparison to CA125. Gynecol. Oncol. 181, 155–161 (2024).
Dinsdale, N. K. et al. Challenges for machine learning in clinical translation of big data imaging studies. Neuron 110, 3866–3881 (2022).
Tobore, I. et al. Deep learning intervention for health care challenges: some biomedical domain considerations. JMIR Mhealth Uhealth 7, e11966 (2019).
Xue, J., Yu, J., Mao, Q. & Gu, X. Advancements and future directions of artificial intelligence in tumor imaging: A comprehensive review of techniques and applications. EngMedicine 2, 100086 (2025).
Pandey, M. et al. The transformational role of GPU computing and deep learning in drug discovery. Nat. Mach. Intell. 4, 211–221 (2022).
Gawehn, E., Hiss, J. A., Brown, J. B. & Schneider, G. Advancing drug discovery via GPU-based deep learning. Expert Opin. Drug Discov. 13, 579–582 (2018).
Bouvarel, B. et al. Clinical impact of CA-125 ELIMination rate constant K (KELIM) on surgical strategy in advanced serous ovarian cancer patients. Int. J. Gynecol. Cancer 34, 574–580 (2024).
Keyl, J. et al. Decoding pan-cancer treatment outcomes using multimodal real-world data and explainable artificial intelligence. Nat. Cancer 6, 307–322 (2025).
Ding, S. et al. HGMSurvNet: a two-stage hypergraph learning network for multimodal cancer survival prediction. Med. Image Anal. 104, 103661 (2025).
Baheti, B. et al. Multimodal explainable artificial intelligence for prognostic stratification of glioblastoma patients. Mod. Pathol. 38, 100797 (2025).
Tian, R. et al. Multimodal fusion model for prognostic prediction and radiotherapy response assessment in head and neck squamous cell carcinoma. NPJ Digit. Med. 8, 302 (2025).
Wang, L. et al. Role of artificial intelligence in medical image analysis. Chin. Med. J. 138, 2879–2894 (2025).
Guo, W. et al. Patients with macroscopic lymph node metastasis expect poor prognosis after neoadjuvant chemotherapy in advanced ovarian cancer: a retrospective cohort study based on a single gynecological team. BMC Cancer 25, 832 (2025).
Zannoni, G. F. et al. Chemotherapy response score (CRS): a comprehensive review of its prognostic and predictive value in high-grade serous carcinoma (HGSC). Gynecol. Oncol. 194, 1–10 (2025).
Li, C. et al. CA-125 elimination rate constant K (KELIM) as a promising predictor of complete cytoreduction after neoadjuvant chemotherapy in advanced ovarian cancer patients: a retrospective study from two Chinese hospitals. BMC Cancer 24, 609 (2024).
Trewin-Nybraten, C. B., Leithe, S., Paulsen, T., Langseth, H. & Fortner, R. T. Ovarian cancer survival by residual disease following cytoreductive surgery: a nationwide study in Norway. Br. J. Cancer 132, 1158–1166 (2025).
Piedimonte, S. et al. Validation of the KELIM score as a predictor of response to neoadjuvant treatment in patients with advanced high grade serous ovarian cancer. Gynecol. Oncol. 167, 417–422 (2022).
Narasimhulu, D. M. et al. Using an evidence-based triage algorithm to reduce 90-day mortality after primary debulking surgery for advanced epithelial ovarian cancer. Gynecol. Oncol. 155, 58–62 (2019).
Kumar, A. et al. Models to predict outcomes after primary debulking surgery: independent validation of models to predict suboptimal cytoreduction and gross residual disease. Gynecol. Oncol. 154, 72–76 (2019).
Postl, M. et al. The predictive value of the fibrinogen-albumin-ratio index on surgical outcomes in patients with advanced high-grade serous ovarian cancer. Cancers https://doi.org/10.3390/cancers16193295 (2024).
Bregar, A. et al. CT prediction of surgical outcome in patients with advanced epithelial ovarian carcinoma undergoing neoadjuvant chemotherapy. Gynecol. Oncol. 152, 568–573 (2019).
Wright, A. A. et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: Society of Gynecologic Oncology and American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. 34, 3460–3473 (2016).
Colombo, N. et al. ESMO-ESGO consensus conference recommendations on ovarian cancer: pathology and molecular biology, early and advanced stages, borderline tumours and recurrent disease. Ann. Oncol. 30, 672–705 (2019).
Colombo, N. et al. Consensus statements and treatment algorithm to guide clinicians in the selection of maintenance therapy for patients with newly diagnosed, advanced ovarian carcinoma: Results of a Delphi study. Gynecol. Oncol. 175, 182–189 (2023).
Ledermann, J. A. et al. ESGO-ESMO-ESP consensus conference recommendations on ovarian cancer: pathology and molecular biology and early, advanced and recurrent disease. Ann. Oncol. 35, 248–266 (2024).
Fathi Kazerooni, A. et al. Multiparametric MRI along with machine learning predicts prognosis and treatment response in pediatric low-grade glioma. Nat. Commun. 16, 340 (2025).
Li, G. et al. An MRI radiomics approach to predict survival and tumour-infiltrating macrophages in gliomas. Brain 145, 1151–1161 (2022).
Huang, Y. & Chung, A. C. S. Disease prediction with edge-variational graph convolutional networks. Med. Image Anal. 77, 102375 (2022).
Nguyen, T. J. et al. Effect of immediate reconstruction on postmastectomy surgical site infection. Ann. Surg. 256, 326–333 (2012).
Wang, H. et al. Development and internal validation of a nomogram to predict mortality during the ICU stay of thoracic fracture patients without neurological compromise: an analysis of the MIMIC-III clinical database. Front. Public Health 9, 818439 (2021).
van Griethuysen, J. J. M. et al. Computational radiomics system to decode the radiographic phenotype. Cancer Res. 77, e104–e107 (2017).
Abel, M. K. et al. Neoadjuvant chemotherapy, case volume, and mortality in advanced ovarian cancer. JAMA Netw. Open 8, e2523434 (2025).
Aletti, G. D., Dowdy, S. C., Podratz, K. C. & Cliby, W. A. Relationship among surgical complexity, short-term morbidity, and overall survival in primary surgery for advanced ovarian cancer. Am. J. Obstet. Gynecol. 197, 676. e671–676. e677 (2007).
Feng, Y., You, H., Zhang, Z., Ji, R. & Gao, Y. Hypergraph Neural Networks. Proceedings of the AAAI Conference on Artificial Intelligence. 33, 3558–3565 (2019).
Biomarker Kinetics. CA-125 KELIM™ calculation website. Available at: https://www.biomarker-kinetics.org/CA-125-neo (accessed 16 May 2025).
Acknowledgements
This work was supported by the National Natural Science Foundation of China [82072885 and 92259104]; Noncommunicable Chronic Diseases-National Science and Technology Major Project [2025ZD0545600 and 2025ZD0545601]; Xingliao Talent Program of Liaoning Province [XLYC2403102] and the Science and Technology Plan Joint Plan of Liaoning Province [2023JH2/101700193].
Author information
Authors and Affiliations
Contributions
Designing the study: S.Z., J.S., and X.Z. Methodology: S.Z., Y.L., J.S., and X.Z. Data curation: S.Z., Z.L., X.L., Y.L., M.L., J.W., and J.Z. Writing—Original Draft: S.Z. Writing—review & editing: J.S., X.Z., and Y.L. Funding acquisition: J.S. and X.Z. Resources: G.W., Z.Y., Y.Z., J.S., and X.Z. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, S., Liu, Y., Liu, Z. et al. Multimodal data-based graph convolutional networks for predicting outcomes in ovarian cancer receiving neoadjuvant chemotherapy. npj Precis. Onc. (2026). https://doi.org/10.1038/s41698-026-01346-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41698-026-01346-9