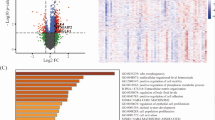
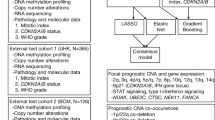
Abstract
Oxaliplatin-based chemotherapy is a standard treatment for metastatic colorectal cancer (mCRC), yet accurate biomarkers to identify responders remain lacking. In this study, we developed and validated a genomic copy number alteration (CNA)-based biomarker to predict clinical response to oxaliplatin-based chemotherapy. A total of 297 samples were collected, and shallow sequencing was employed to extract CNA features. The resulting model named “CNA fingerprint” is an XGBoost model trained using 7 CNA features. The model was validated across three independent test cohorts from two centers, achieving area under the receiver operating characteristic curve (AUC) of 0.87, 0.87, and 0.85, respectively. The primary predictor was the number of DNA segments with high absolute copy numbers. Our findings suggest that the CNA fingerprint could be used as biomarker for oxaliplatin-based chemotherapy response prediction in mCRC. Further prospective clinical trials are warranted to evaluate CNA fingerprint’s performance in clinical applications.
Similar content being viewed by others
References
Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Ca-a Cancer J. Clin. 71, 209–249 (2021).
SEER. Cancer Stat Facts: Colorectal Cancer, https://seer.cancer.gov/statfacts/html/colorect.html (2024).
Kahi, C. J. et al. Colonoscopy surveillance after colorectal cancer resection: recommendations of the US multi-society task force on colorectal cancer. Gastroenterology 150, 758–768 (2016).
Alese, O. B. et al. Update on emerging therapies for advanced colorectal cancer. Am. Soc. Clin. Oncol. Educ. Book, e389574, https://doi.org/10.1200/edbk_389574 (2023).
Kawai, S. et al. Comparison of irinotecan and oxaliplatin as the first-line therapies for metastatic colorectal cancer: a meta-analysis. BMC Cancer 21, 116 (2021).
Bahrami, A. et al. Genetic variants as potential predictive biomarkers in advanced colorectal cancer patients treated with oxaliplatin-based chemotherapy. J. Cell. Physiol. 233, 2193–2201 (2018).
Chen, L. J. et al. Machine learning predicts oxaliplatin benefit in early colon cancer. J. Clin. Oncol. 42, 1520–1530 (2024).
Yin, M. et al. ERCC1 and ERCC2 polymorphisms predict clinical outcomes of oxaliplatin-based chemotherapies in gastric and colorectal cancer: a systemic review and meta-analysis. Clin. Cancer Res. 17, 1632–1640 (2011).
Abraham, J. P. et al. Clinical validation of a machine-learning-derived signature predictive of outcomes from first-line oxaliplatin-based chemotherapy in advanced colorectal cancer. Clin. Cancer Res. 27, 1174–1183 (2021).
Russo, V. et al. Artificial intelligence predictive models of response to cytotoxic chemotherapy alone or combined to targeted therapy for metastatic colorectal cancer patients: a systematic review and meta-analysis. Cancers 14, 4012 (2022).
Yi, K. & Ju, Y. S. Patterns and mechanisms of structural variations in human cancer. Exp. Mol. Med. 50, 98 (2018).
Tao, Z. Y. et al. The repertoire of copy number alteration signatures in human cancer. Brief. Bioinform. 24, bbad053 (2023).
Wang, S. X. et al. Copy number signature analysis tool and its application in prostate cancer reveals distinct mutational processes and clinical outcomes. PLoS Genet. 17, e1009557 (2021).
McShane, L. M. et al. Reporting recommendations for tumor marker prognostic studies (REMARK). Natl. Cancer Inst. 97, 1180–1184 (2005).
Haan, J. C. et al. Genomic landscape of metastatic colorectal cancer. Nat. Commun. 5, https://doi.org/10.1038/ncomms6457 (2014).
Malla, S. B. et al. In-depth clinical and biological exploration of dna damage immune response as a biomarker for oxaliplatin use in colorectal cancer. Clin. Cancer Res. 27, 288–300 (2021).
Replogle, J. M. et al. Aneuploidy increases resistance to chemotherapeutics by antagonizing cell division. Proc. Natl. Acad. Sci. USA 117, 30566–30576 (2020).
Shukla, A. et al. Chromosome arm aneuploidies shape tumour evolution and drug response. Nat. Commun. 11, 449 (2020).
Baran, B. et al. Difference between left-sided and right-sided colorectal cancer: a focused review of literature. 11, https://doi.org/10.14740/gr1062w (2018).
Watson, R. G. et al. Amplification of thymidylate synthetase in metastatic colorectal cancer patients pretreated with 5-fluorouracil-based chemotherapy. Eur. J. Cancer 46, 3358–3364 (2010).
Buess, M. et al. STRAP is a strong predictive marker of adjuvant chemotherapy benefit in colorectal cancer. Neoplasia 6, 813–820 (2004).
Fujita, Y. et al. aCGH analysis of predictive biomarkers for response to bevacizumab plus oxaliplatin- or irinotecan-based chemotherapy in patients with metastatic colorectal cancer. Oncologist 24, 327–337 (2019).
Wu, Z. Y. et al. Copy number amplification of DNA damage repair pathways potentiates therapeutic resistance in cancer. Theranostics 10, 3939–3951 (2020).
Ippolito, M. R. et al. Gene copy-number changes and chromosomal instability induced by aneuploidy confer resistance to chemotherapy. Dev. Cell 56, 2440–244 (2021).
Galofré, C. et al. Tetraploidy-associated genetic heterogeneity confers chemo-radiotherapy resistance to colorectal cancer cells. Cancers 12, 1118 (2020).
Jardim, D. L., Rodrigues, C. A., Novis, Y. A. S., Rocha, V. G. & Hoff, P. M. Oxaliplatin-related thrombocytopenia. Ann. Oncol. 23, 1937–1942 (2012).
Arango, D. et al. Molecular mechanisms of action and prediction of response to oxaliplatin in colorectal cancer cells. Br. J. Cancer 91, 1931–1946 (2004).
Bencardino, K. et al. Oxaliplatin immune-induced syndrome occurs with cumulative administration and rechallenge: single institution series and systematic review study. Clin. Colorectal Cancer 15, 213–221 (2016).
Wang, S. & Liu, X. -s The UCSCXenaTools R package: a toolkit for accessing genomics data from UCSC Xena platform, from cancer multi-omics to single-cell RNA-seq. J. Open Source Softw. 4, 1627 (2019).
Chen, S. F. Ultrafast one-pass FASTQ data preprocessing, quality control, and deduplication using fastp. Imeta 2, e107 (2023).
Li, H. Aligning sequence reads, clone sequences and assembly contigs with BWA-MEM, https://doi.org/10.48550/arXiv.1303.3997 (2013).
Scheinin, I. et al. DNA copy number analysis of fresh and formalin-fixed specimens by shallow whole-genome sequencing with identification and exclusion of problematic regions in the genome assembly. Genome Res. 24, 2022–2032 (2014).
Poell, J. B. et al. ACE: absolute copy number estimation from low-coverage whole-genome sequencing data. Bioinformatics 35, 2847–2849 (2019).
Wu, C. X. et al. Single-cell copy number alteration signature analysis reveals masked patterns and potential biomarkers for cancer. Commun. Biol. 8, https://doi.org/10.1038/s42003-025-08994-w (2025).
Hieronymus, H. et al. Tumor copy number alteration burden is a pan-cancer prognostic factor associated with recurrence and death. Elife 7, e37294 (2018).
Drews, R. M. et al. A pan-cancer compendium of chromosomal instability. Nature 606, 976–983 (2022).
Lang., M. et al. mlr3: a modern object-oriented machine learning framework in R. J. Open Source Softw., https://doi.org/10.21105/joss.01903 (2019).
Yao, H. Z. et al. Copy number alteration features in pan-cancer homologous recombination deficiency prediction and biology. Commun. Biol. 6, 527 (2023).
Sztupinszki, Z. et al. Migrating the SNP array-based homologous recombination deficiency measures to next generation sequencing data of breast cancer. npj Breast Cancer 4, 16 (2018).
Kuhn, M. Building predictive models in R using the caret package. J. Stat. Softw. 28, 1–26 (2008).
Robin, X. et al. pROC: an open-source package for R and S plus to analyze and compare ROC curves. BMC Bioinform. 12, 77 (2011).
Acknowledgements
We thank ShanghaiTech University High Performance Computing Public Service Platform for computing services. We thank multi-omics facility, molecular and cell biology core facility of ShanghaiTech University for technical help. This work is supported by National Natural Science Foundation of China (82373149), Shanghai Science and Technology Commission (24J22800700), cross disciplinary Research Fund of Shanghai Ninth People's Hospital, Shanghai JiaoTong University School of Medicine (JYJC202227), open project fund of the National Health Commission’s key laboratory of individualized diagnosis and treatment of nasopharyngeal cancer (2023NPCCK02) and startup funding from ShanghaiTech University.
Author information
Authors and Affiliations
Contributions
J.W., J.W., Z.T., and T.W. contributed equally to this work as joint first authors. X.L., X.L., and X.S.L contributed equally to this work as joint senior authors. X.S.L were responsible for overall study design. J.W., J.W., Z.T., and T.W. accessed and verified the underlying data. J.W., J.W., Z.T., T.W., K.D., J.W., N.W., Z.Y., R.Z., J.S., and X.Z. were responsible for analysis and interpretation of data. J.W., Z.T., T.W., and X.S.L. were responsible for writing, review, and/or revision of the manuscript. X.L., X.L., and X.S.L. were study supervisions and guarantors. All the authors have read, discussed, and unanimously approved the final version of the manuscript. All authors had full access to the data and had the final responsibility for the decision to submit for publication.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Weng, J., Wang, J., Tao, Z. et al. Copy number alteration fingerprint predicts the clinical response of oxaliplatin-based chemotherapy in metastatic colorectal cancer. npj Precis. Onc. (2026). https://doi.org/10.1038/s41698-026-01354-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41698-026-01354-9