Abstract
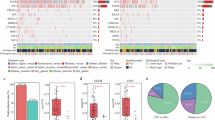
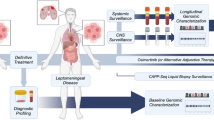
Lung adenocarcinoma (LUAD)-derived leptomeningeal metastases (LM) represent a predominant subtype among all LM cases. Nevertheless, the cerebrospinal fluid (CSF) profile of LUAD-LM patients remains poorly characterized and reliable CSF diagnostic biomarkers for LUAD-LM have yet to be established. Using single-cell RNA sequencing data of CSF cells from six LUAD-LM patients, we drew a systematic transcriptomic atlas of the CSF cellular landscape. Our analysis revealed that LUAD-LM reprograms CSF into an immunosuppressive state, marked by the emergence of pro-tumoral LGMN-SELENOPhigh macrophages and proliferating CSF circulating tumor cells (CSF-CTC). Cell-cell communication analysis showed that CSF-CTC reinforces immunosuppression by co-inhibitory checkpoint axis NECTIN2_TIGIT axis with the CD8+T/NK cells, and via CD47_SIRPA axis with antigen-presenting cells. Furthermore, we identified the single-cell transcriptomic difference between CSF-CTC and tumor cells of parenchymal brain metastases (PBM). Notably, Trophoblast cell surface antigen 2 (TROP2) levels in CSF were significantly elevated in LUAD-LM patients versus both normal controls (NC) and LUAD patients without LM (Non-LM). It showed strong diagnostic accuracy for distinguishing LUAD LM from Non-LM or NC, and PBM did not influence the CSF TROP2 level. Collectively, our findings advance the understanding of LUAD-LM pathogenesis and highlight the potential of CSF TROP2 as a diagnostic biomarker for LM.
Similar content being viewed by others
Data availability
The data that support the findings of this study are openly available in the National Center for Biotechnology information Gene Expression Omnibus (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE276139), reference number GSE276139.
References
Ozcan, G., Singh, M. & Vredenburgh, J. J. Leptomeningeal Metastasis from Non-Small Cell Lung Cancer and Current Landscape of Treatments. Clin. Cancer Res 29, 11–29 (2023).
Wang, Y., Yang, X., Li, N. J. & Xue, J. X. Leptomeningeal metastases in non-small cell lung cancer: Diagnosis and treatment. Lung Cancer 174, 1–13 (2022).
Wu, F. et al. Single-cell profiling of tumor heterogeneity and the microenvironment in advanced non-small cell lung cancer. Nat. Commun. 12, 2540 (2021).
Kim, N. et al. Single-cell RNA sequencing demonstrates the molecular and cellular reprogramming of metastatic lung adenocarcinoma. Nat. Commun. 11, 2285 (2020).
Sun, H. F. et al. Single-cell RNA sequencing reveals cellular and molecular reprograming landscape of gliomas and lung cancer brain metastases. Clin. Transl. Med 12, e1101 (2022).
Wang, Z. et al. Single-cell transcriptomic analyses provide insights into the cellular origins and drivers of brain metastasis from lung adenocarcinoma. Neuro Oncol. 25, 1262–1274 (2023).
Maynard, A. et al. Therapy-Induced Evolution of Human Lung Cancer Revealed by Single-Cell RNA Sequencing. Cell 182, 1232–1251 e1222 (2020).
Yaniv, D., Mattson, B., Talbot, S., Gleber-Netto, F. O. & Amit, M. Targeting the peripheral neural-tumour microenvironment for cancer therapy. Nat. Rev. Drug Discov. 23, 780–796 (2024).
Ruan, H. et al. Circulating tumor cell characterization of lung cancer brain metastases in the cerebrospinal fluid through single-cell transcriptome analysis. Clin. Transl. Med 10, e246 (2020).
Fitzpatrick, A. et al. Assessing CSF ctDNA to Improve Diagnostic Accuracy and Therapeutic Monitoring in Breast Cancer Leptomeningeal Metastasis. Clin. Cancer Res 28, 1180–1191 (2022).
Li, X. et al. Diagnostic value of cerebrospinal fluid human epididymis protein 4 for leptomeningeal metastasis in lung adenocarcinoma. Front Immunol. 15, 1339914 (2024).
Wang, X. et al. CEACAM6 serves as a biomarker for leptomeningeal metastasis in lung adenocarcinoma. Cancer Med 12, 4521–4529 (2023).
Liu, X. et al. Advances in Trop2-targeted therapy: Novel agents and opportunities beyond breast cancer. Pharm. Ther. 239, 108296 (2022).
Zheng, Z. & Dong, X. J. Clinical value of serum trophoblast cell surface protein 2 (TROP2) antibody in non-small-cell lung cancer patients. Biomarkers 21, 739–742 (2016).
Barrett, C. W. et al. Selenoprotein P influences colitis-induced tumorigenesis by mediating stemness and oxidative damage. J. Clin. Invest 125, 2646–2660 (2015).
Lai, X. et al. Ferritinophagy activation states determine the susceptibility to ferroptosis of macrophages in bone marrow and spleen. Int J. Biol. Sci. 21, 4567–4585 (2025).
Liu, Z. et al. Lipid-associated macrophages in the tumor-adipose microenvironment facilitate breast cancer progression. Oncoimmunology 11, 2085432 (2022).
Cheng, S. et al. A pan-cancer single-cell transcriptional atlas of tumor infiltrating myeloid cells. Cell 184, 792–809 e723 (2021).
Urh, K., Zidar, N. & Bostjancic, E. Bioinformatics Analysis of RNA-seq Data Reveals Genes Related to Cancer Stem Cells in Colorectal Cancerogenesis. Int. J. Mol. Sci 23, 13252 (2022).
Maryam, A. & Chin, Y. R. ANLN Enhances Triple-Negative Breast Cancer Stemness Through TWIST1 and BMP2 and Promotes its Spheroid Growth. Front Mol. Biosci. 8, 700973 (2021).
Hsu, C. C. et al. A multi-mode Wnt- and stemness-regulatory module dictated by FOXM1 and ASPM isoform I in gastric cancer. Gastric Cancer 24, 624–639 (2021).
Pan, X. W. et al. Identification of a novel cancer stem cell subpopulation that promotes progression of human fatal renal cell carcinoma by single-cell RNA-seq analysis. Int J. Biol. Sci. 16, 3149–3162 (2020).
Zhu, C. et al. Targeting DEP domain containing 1 in anaplastic thyroid carcinoma: Implications for stemness regulation and malignant phenotype suppression. Heliyon 10, e27150 (2024).
Tilghman, J. et al. HMMR maintains the stemness and tumorigenicity of glioblastoma stem-like cells. Cancer Res 74, 3168–3179 (2014).
Deng, Y. et al. NUF2 Promotes Breast Cancer Development as a New Tumor Stem Cell Indicator. Int. J. Mol. Sci. 24, (2023).
Li, H. et al. Nucleolar and spindle associated protein 1 promotes metastasis of cervical carcinoma cells by activating Wnt/beta-catenin signaling. J. Exp. Clin. Cancer Res 38, 33 (2019).
Sacco, A. et al. SOX2 promotes a cancer stem cell-like phenotype and local spreading in oral squamous cell carcinoma. PLoS One 18, e0293475 (2023).
Zhao, X. et al. Single-cell transcriptomics reveals heterogeneous progression and EGFR activation in pancreatic adenosquamous carcinoma. Int J. Biol. Sci. 17, 2590–2605 (2021).
Walcher, L. et al. Cancer Stem Cells-Origins and Biomarkers: Perspectives for Targeted Personalized Therapies. Front Immunol. 11, 1280 (2020).
Tian, X., Ning, Q., Yu, J. & Tang, S. T-cell immunoglobulin and ITIM domain in cancer immunotherapy: A focus on tumor-infiltrating regulatory T cells. Mol. Immunol. 147, 62–70 (2022).
Alvarez Calderon, F. et al. Targeting of the CD161 inhibitory receptor enhances T-cell-mediated immunity against hematological malignancies. Blood 143, 1124–1138 (2024).
Zhang, W. et al. Advances in Anti-Tumor Treatments Targeting the CD47/SIRPalpha Axis. Front. Immunol. 11, 18 (2020).
Joller, N., Anderson, A. C. & Kuchroo, V. K. LAG-3, TIM-3, and TIGIT: Distinct functions in immune regulation. Immunity 57, 206–222 (2024).
Liu, X. et al. Deciphering the tumor immune microenvironment of imatinib-resistance in advanced gastrointestinal stromal tumors at single-cell resolution. Cell Death Dis. 15, 190 (2024).
Tekguc, M., Wing, J. B., Osaki, M., Long, J., Sakaguchi, S. Treg-expressed CTLA-4 depletes CD80/CD86 by trogocytosis, releasing free PD-L1 on antigen-presenting cells. Proc. Natl. Acad. Sci. USA 118, (2021).
Halliday, N. et al. CD86 Is a Selective CD28 Ligand Supporting FoxP3+ Regulatory T Cell Homeostasis in the Presence of High Levels of CTLA-4. Front. Immunol. 11, 600000 (2020).
Tan, Y. et al. Malignant cell-mediated Treg immune suppression via ICOSLG-ICOS axis in tumor microenvironment relates to nasopharyngeal carcinoma prognosis. Int. Immunopharmacol. 167, 115743 (2025).
Wang, X. et al. NQO1 Triggers Neutrophil Recruitment and NET Formation to Drive Lung Metastasis of Invasive Breast Cancer. Cancer Res. 84, 3538–3555 (2024).
Jin, R. et al. Desmoglein-2 modulates tumor progression and osimertinib drug resistance through the EGFR/Src/PAK1 pathway in lung adenocarcinoma. Cancer Lett. 483, 46–58 (2020).
Li, S., Cheng, Y., Gao, C., Yuan, Q. & Lu, X. SEMA3C promotes thyroid cancer via the Wnt/beta-catenin pathway. Exp. Cell Res. 444, 114378 (2025).
Yu, H. et al. Integrated single-cell analyses decode the developmental landscape of the human fetal spine. iScience 25, 104679 (2022).
Yang, Y., Zhang, B., Yang, Y., Peng, B. & Ye, R. FOXM1 accelerates wound healing in diabetic foot ulcer by inducing M2 macrophage polarization through a mechanism involving SEMA3C/NRP2/Hedgehog signaling. Diab Res Clin. Pr. 184, 109121 (2022).
Lv, X. et al. VEGFA and APOE regulate distinct functional states of mast cells in hepatocellular carcinoma: A single-cell transcriptome analysis. Int J. Biol. Macromol. 321, 146131 (2025).
Yang, K., Yang, T., Yu, J., Li, F. & Zhao, X. Integrated transcriptional analysis reveals macrophage heterogeneity and macrophage-tumor cell interactions in the progression of pancreatic ductal adenocarcinoma. BMC Cancer 23, 199 (2023).
Motevasseli, M. et al. Distinct tumor-TAM interactions in IDH-stratified glioma microenvironments unveiled by single-cell and spatial transcriptomics. Acta Neuropathol. Commun. 12, 133 (2024).
Mathew, D. et al. Combined JAK inhibition and PD-1 immunotherapy for non-small cell lung cancer patients. Science 384, eadf1329 (2024).
Zhou, F. et al. The changing treatment landscape of EGFR-mutant non-small-cell lung cancer. Nat. Rev. Clin. Oncol. 22, 95–116 (2025).
Bakiri, L. et al. Liver cancer development driven by the AP-1/c-Jun~Fra-2 dimer through c-Myc. Proc. Natl. Acad. Sci. USA 121, e2404188121 (2024).
Bao, Z. et al. SNAIL Induces EMT and Lung Metastasis of Tumours Secreting CXCL2 to Promote the Invasion of M2-Type Immunosuppressed Macrophages in Colorectal Cancer. Int J. Biol. Sci. 18, 2867–2881 (2022).
Tolaney, S. M. et al. A Phase I Study of DLYE5953A, an Anti-LY6E Antibody Covalently Linked to Monomethyl Auristatin E, in Patients with Refractory Solid Tumors. Clin. Cancer Res 26, 5588–5597 (2020).
Liu, X. et al. Trop2-targeted therapies in solid tumors: advances and future directions. Theranostics 14, 3674–3692 (2024).
Zhu, J. et al. Delineating the dynamic evolution from preneoplasia to invasive lung adenocarcinoma by integrating single-cell RNA sequencing and spatial transcriptomics. Exp. Mol. Med 54, 2060–2076 (2022).
Zou, Y. et al. The Single-Cell Landscape of Intratumoral Heterogeneity and The Immunosuppressive Microenvironment in Liver and Brain Metastases of Breast Cancer. Adv. Sci. (Weinh.) 10, e2203699 (2023).
Rousseau, A., Parisi, C. & Barlesi, F. Anti-TIGIT therapies for solid tumors: a systematic review. ESMO Open 8, 101184 (2023).
Guan, X. et al. Anti-TIGIT antibody improves PD-L1 blockade through myeloid and T(reg) cells. Nature 627, 646–655 (2024).
Remsik, J. & Boire, A. The path to leptomeningeal metastasis. Nat. Rev. Cancer 24, 448–460 (2024).
Liu, W., Powell, C. A. & Wang, Q. Tumor microenvironment in lung cancer-derived brain metastasis. Chin. Med J. (Engl.) 135, 1781–1791 (2022).
Nelson, B. E. & Meric-Bernstam, F. Leveraging TROP2 Antibody-Drug Conjugates in Solid Tumors. Annu Rev. Med 75, 31–48 (2024).
Stepan, L. P. et al. Expression of Trop2 cell surface glycoprotein in normal and tumor tissues: potential implications as a cancer therapeutic target. J. Histochem Cytochem 59, 701–710 (2011).
Lin, J. C. et al. TROP2 is epigenetically inactivated and modulates IGF-1R signalling in lung adenocarcinoma. EMBO Mol. Med 4, 472–485 (2012).
Hu, Y., Zhu, Y., Qi, D., Tang, C. & Zhang, W. Trop2-targeted therapy in breast cancer. Biomark. Res. 12, 82 (2024).
Hou, J. et al. TROP2 promotes the proliferation and metastasis of glioblastoma cells by activating the JAK2/STAT3 signaling pathway. Oncol. Rep. 41, 753–764 (2019).
Wang, X. et al. Chemotherapy agents-induced immunoresistance in lung cancer cells could be reversed by trop-2 inhibition in vitro and in vivo by interaction with MAPK signaling pathway. Cancer Biol. Ther. 14, 1123–1132 (2013).
Bessede, A. et al. TROP2 Is Associated with Primary Resistance to Immune Checkpoint Inhibition in Patients with Advanced Non-Small Cell Lung Cancer. Clin. Cancer Res 30, 779–785 (2024).
Chamberlain, M. et al. Leptomeningeal metastases: a RANO proposal for response criteria. Neuro Oncol. 19, 484–492 (2017).
Smalley, I. et al. Single-Cell Characterization of the Immune Microenvironment of Melanoma Brain and Leptomeningeal Metastases. Clin. Cancer Res. 27, 4109–4125 (2021).
He, L. J. et al. TROP2 is highly expressed in cutaneous squamous cell carcinomas and a subset of adnexal carcinomas: A potential therapeutic target with TROP2-directed antibody drug conjugates. Hum. Pathol. 160, 105853 (2025).
Tas, F., Karabulut, S. & Duranyildiz, D. Clinical significance of serum epithelial cell adhesion molecule (EPCAM) levels in patients with lung cancer. Mol. Cell Biochem. 396, 307–312 (2014).
Chi, Y. et al. Cancer cells deploy lipocalin-2 to collect limiting iron in leptomeningeal metastasis. Science 369, 276–282 (2020).
Ruan, H. et al. Single-cell RNA sequencing reveals the characteristics of cerebrospinal fluid tumour environment in breast cancer and lung cancer leptomeningeal metastases. Clin. Transl. Med. 12, e885 (2022).
Gonzalez, H. et al. Cellular architecture of human brain metastases. Cell 185, 729–745 e720 (2022).
Wu, Y. et al. Spatiotemporal Immune Landscape of Colorectal Cancer Liver Metastasis at Single-Cell Level. Cancer Discov. 12, 134–153 (2022).
Acknowledgements
This work was supported by the National Natural Science Foundation of China, grant number 82102489 (Haoyu Ruan), 82202610 (Yuexinzi Jin), 82502175 (Ting Wang), 82272429 (Ming Guan), 82103328 (Yuqing Wu), and the Shanghai Sailing Program (23YF1457700, Zhe Wang).
Author information
Authors and Affiliations
Contributions
H.R., M.G., Q.O., and Y.W. contributed to the conceptualization and design of the study. H.R., Z.W., J.L. (Jie Luo), Y.J., and T.W. carried out the primary coding, data analysis, and visualization. H.R. and Z.W. drafted the manuscript and assisted with the final revision of the manuscript. Y.M., J.L. (Jingping Liu), and H.J. collected the samples. All authors reviewed, provided feedback, and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, Z., Luo, J., Jin, Y. et al. Cellular landscape and diagnostic value of TROP2 in cerebrospinal fluid of lung adenocarcinoma leptomeningeal metastases. npj Precis. Onc. (2026). https://doi.org/10.1038/s41698-026-01379-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41698-026-01379-0