Abstract
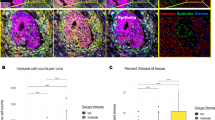
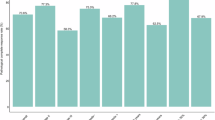
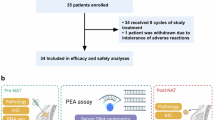
This is a single-arm, phase II clinical trial aimed to evaluate mFOLFOX6 plus camrelizumab and apatinib as a neoadjuvant regimen for locally advanced colon cancer. This trial was registered on ClinicalTrials.gov (NCT04625803), with the registration date of November 30, 2020. The primary endpoint was the pathological response rate (TRG 2–4, Dworak criteria). Time-of-flight, imaging mass cytometry and spatial proteomic analysis was applied to explore efficacy-related predictive biomarkers. Of 12 enrolled patients, 11 underwent surgery. The TRG 2–4 rate was 81.8% overall (87.5% in the MSS subgroup). Five patients (45.5%) achieved pathological complete response (pCR). With a median follow-up of 36.60 months, only one patient relapsed. An increase in the frequency of CD8 + T-cells and a decreased frequency of tissue-resident memory 1 (Trm-1) and regulatory T (Treg) cells are associated with a favorable response to neoadjuvant therapy. Spatiotemporal multiomics analysis highlighted tissue-resident macrophages as potential predictive biomarkers and warrants further verification.
Similar content being viewed by others
Data availability
Data are not publicly available due to privacy requirements but are available upon reasonable request from the corresponding author.
References
Siegel, R. L., Giaquinto, A. N. & Jemal, A. Cancer statistics, 2024. CA Cancer J. Clin. 74, 12–49 (2024).
André, T. et al. Improved overall survival with oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment in stage II or III colon cancer in the MOSAIC trial. J. Clin. Oncol. 27, 3109–3116 (2009).
André, T. et al. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N. Engl. J. Med. 350, 2343–2351 (2004).
Surgical resection with or without preoperative chemotherapy in oesophageal cancer: a randomised controlled trial. Lancet. 359, 1727–1733 (2002).
Cunningham, D. et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N. Engl. J. Med. 355, 11–20 (2006).
Alieva, M., van Rheenen, J. & Broekman, M. L. D. Potential impact of invasive surgical procedures on primary tumor growth and metastasis. Clin. Exp. Metastasis 35, 319–331 (2018).
Peng, C. & Kircher, S. M. Neoadjuvant chemotherapy in colon cancer: more than just an optical illusion. J. Clin. Oncol. 42, 2949–2954 (2024).
Morton, D. et al. Preoperative chemotherapy for operable colon cancer: mature results of an international randomized controlled trial. J. Clin. Oncol. 41, 1541–1552 (2023).
Jensen, L. H. et al. Phase III randomized clinical trial comparing the efficacy of neoadjuvant chemotherapy and standard treatment in patients with locally advanced colon cancer: The NeoCol trial. J. Clin. Oncol. 41, LBA3503 (2023).
Hu, H. et al. Neoadjuvant chemotherapy with oxaliplatin and fluoropyrimidine versus upfront surgery for locally advanced colon cancer: the randomized, Phase III OPTICAL Trial. J. Clin. Oncol. JCO2301889 (2024).
Gosavi, R. et al. Neoadjuvant chemotherapy in locally advanced colon cancer: a systematic review and meta-analysis. Int J. Colorectal Dis. 36, 2063–2070 (2021).
Davey, M. G. et al. Evaluating the oncological safety of neoadjuvant chemotherapy in locally advanced colon carcinoma: a systematic review and meta-analysis of randomised clinical trials and propensity-matched studies. Int J. Colorectal Dis. 38, 193 (2023).
Liang, Z. et al. The role of neoadjuvant chemotherapy in patients with locally advanced colon cancer: A systematic review and meta-analysis. Front Oncol. 12, 1024345 (2022).
André, T. et al. Pembrolizumab in microsatellite-instability-high advanced colorectal cancer. N. Engl. J. Med. 383, 2207–2218 (2020).
Chalabi, M. et al. LBA7 Neoadjuvant immune checkpoint inhibition in locally advanced MMR-deficient colon cancer: The NICHE-2 study. Ann. Oncol. 33, S1389 (2022).
Hu, H. et al. Neoadjuvant PD-1 blockade with toripalimab, with or without celecoxib, in mismatch repair-deficient or microsatellite instability-high, locally advanced, colorectal cancer (PICC): a single-centre, parallel-group, non-comparative, randomised, phase 2 trial. Lancet Gastroenterol. Hepatol. 7, 38–48 (2022).
Xu, J. et al. Camrelizumab in combination with apatinib in patients with advanced hepatocellular carcinoma (RESCUE): a nonrandomized, open-label, Phase II Trial. Clin. Cancer Res. 27, 1003–1011 (2021).
Choueiri, T. K. et al. Lenvatinib plus pembrolizumab versus sunitinib as first-line treatment of patients with advanced renal cell carcinoma (CLEAR): extended follow-up from the phase 3, randomised, open-label study. Lancet Oncol. 24, 228–238 (2023).
Guo, Y. et al. Phase 1b/2 trial of fruquintinib plus sintilimab in treating advanced solid tumours: The dose-escalation and metastatic colorectal cancer cohort in the dose-expansion phases. Eur. J. Cancer 181, 26–37 (2023).
Fukuoka, S. et al. Regorafenib plus nivolumab in patients with advanced gastric or colorectal cancer: an open-label, dose-escalation, and dose-expansion phase Ib trial (REGONIVO, EPOC1603). J. Clin. Oncol. 38, 2053–2061 (2020).
Xu, J. et al. Anti-PD-1 Antibody SHR-1210 Combined with Apatinib for Advanced Hepatocellular Carcinoma, Gastric, or Esophagogastric Junction Cancer: An Open-label, Dose Escalation and Expansion Study. Clin. Cancer Res. 25, 515–523 (2019).
Li, C. et al. Pathologic response of phase III study: perioperative camrelizumab plus rivoceranib and chemotherapy versus chemotherapy for locally advanced gastric cancer (DRAGON IV/CAP 05). J. Clin. Oncol. JCO2400795. (2024).
Cai, X. et al. Apatinib enhanced anti-PD-1 therapy for colon cancer in mice via promoting PD-L1 expression. Int Immunopharmacol. 88, 106858 (2020).
Scott, L. J. Apatinib: a review in advanced gastric cancer and other advanced cancers. Drugs 78, 747–758 (2018).
Li, Q. et al. Low-dose anti-angiogenic therapy sensitizes breast cancer to PD-1 Blockade. Clin. Cancer Res 26, 1712–1724 (2020).
Bortolomeazzi, M. et al. Immunogenomics of colorectal cancer response to checkpoint blockade: analysis of the KEYNOTE 177 trial and validation cohorts. Gastroenterology 161, 1179–1193 (2021).
Bi, K. et al. Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma. Cancer Cell 39, 649–61.e5 (2021).
Zhang, Y. et al. Single-cell analyses reveal key immune cell subsets associated with response to PD-L1 blockade in triple-negative breast cancer. Cancer Cell 39, 1578–1593.e8 (2021).
Galon, J. et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Sci. (N. Y., NY) 313, 1960–1964 (2006).
Christofides, A. et al. The complex role of tumor-infiltrating macrophages. Nat. Immunol. 23, 1148–1156 (2022).
Sica, A. et al. Macrophage polarization in tumour progression. Semin Cancer Biol. 18, 349–355 (2008).
Li, L. et al. Spatially resolved proteomics via tissue expansion. Nat. Commun. 13, 7242 (2022).
Le, D. T. et al. PD-1 blockade in tumors with mismatch-repair deficiency. N. Engl. J. Med 372, 2509–2520 (2015).
Giannakis, M. et al. Genomic correlates of immune-cell infiltrates in colorectal carcinoma. Cell Rep. 17, 1206 (2016).
Chen, D. S. & Mellman, I. Oncology meets immunology: the cancer-immunity cycle. Immunity 39, 1–10 (2013).
Hegde, P. S., Karanikas, V. & Evers, S. The where, the when, and the how of immune monitoring for cancer immunotherapies in the era of checkpoint inhibition. Clin. Cancer Res. 22, 1865–1874 (2016).
Gebhardt, T., Palendira, U., Tscharke, D. C. & Bedoui, S. Tissue-resident memory T cells in tissue homeostasis, persistent infection, and cancer surveillance. Immunol. Rev. 283, 54–76 (2018).
Jiang, F. et al. PD-1 and TIGIT coexpressing CD8 + CD103 + tissue-resident memory cells in endometrial cancer as potential targets for immunotherapy. Int. Immunopharmacol. 127, 111381 (2024).
Romagnoli, G. et al. CD8+CD103+PD1+TIM3+ T cells in glioblastoma microenvironment correlate with prognosis. Immunology 171, 198–211 (2024).
Luoma, A. M. et al. Molecular Pathways of Colon Inflammation Induced by Cancer Immunotherapy. Cell 182, 655–71.e22 (2020).
Chalabi, M. et al. Neoadjuvant immunotherapy leads to pathological responses in MMR-proficient and MMR-deficient early-stage colon cancers. Nat. Med. 26, 566–576 (2020).
Avallone, A. et al. 491P Neoadjuvant nivolumab in early stage colorectal cancer. Ann. Oncol. 31, S449 (2020).
Hissong, E. et al. Neoadjuvant botensilimab (BOT) plus balstilimab (BAL) in resectable mismatch repair proficient (pMMR) and deficient (dMMR) colorectal cancer (CRC): NEST clinical trial update. J. Clin. Oncol. 43, 207 (2025).
Ambrosini, M. et al. Preoperative botensilimab (BOT) with or without balstilimab (BAL) for patients with resectable locally advanced pMMR or dMMR colon cancer: Results from the UNICORN trial by GONO. J. Clin. Oncol. 43, 158 (2025).
Kasi, P. M. et al. Neoadjuvant botensilimab plus balstilimab response pattern in locally advanced mismatch repair proficient colorectal cancer. Oncogene 42, 3252–3259 (2023).
Tesniere, A. et al. Immunogenic death of colon cancer cells treated with oxaliplatin. Oncogene 29, 482–491 (2010).
Park, S. J. et al. Cisplatin and oxaliplatin induce similar immunogenic changes in preclinical models of head and neck cancer. Oral. Oncol. 95, 127–135 (2019).
Guan, Y. et al. FOLFOX Chemotherapy Ameliorates CD8 T Lymphocyte Exhaustion and Enhances Checkpoint Blockade Efficacy in Colorectal Cancer. Front Oncol. 10, 586 (2020).
Kudo, M. Combination cancer immunotherapy with molecular targeted agents/anti-CTLA-4 antibody for hepatocellular carcinoma. Liver Cancer 8, 1–11 (2019).
Lazarov, T., Juarez-Carreño, S., Cox, N. & Geissmann, F. Physiology and diseases of tissue-resident macrophages. Nature 618, 698–707 (2023).
Etzerodt, A. et al. Tissue-resident macrophages in omentum promote metastatic spread of ovarian cancer. J. Exp. Med. 217.(2020).
Casanova-Acebes, M. et al. Tissue-resident macrophages provide a pro-tumorigenic niche to early NSCLC cells. Nature 595, 578–584 (2021).
Zhang, Q. W. et al. Prognostic significance of tumor-associated macrophages in solid tumor: a meta-analysis of the literature. PLoS ONE 7, e50946 (2012).
Gentles, A. J. et al. The prognostic landscape of genes and infiltrating immune cells across human cancers. Nat. Med. 21, 938–945 (2015).
DeNardo, D. G. & Ruffell, B. Macrophages as regulators of tumour immunity and immunotherapy. Nat. Rev. Immunol. 19, 369–382 (2019).
Nalio Ramos, R. et al. Tissue-resident FOLR2(+) macrophages associate with CD8(+) T cell infiltration in human breast cancer. Cell 185, 1189–207.e25 (2022).
Zhao, J., Andreev, I. & Silva, H. M. Resident tissue macrophages: Key coordinators of tissue homeostasis beyond immunity. Sci. Immunol. 9, eadd1967 (2024).
Bao, X. et al. An immunometabolism subtyping system identifies S100A9(+) macrophage as an immune therapeutic target in colorectal cancer based on multiomics analysis. Cell Rep. Med 4, 100987 (2023).
Boissonnas, A. et al. CD8+ tumor-infiltrating T cells are trapped in the tumor-dendritic cell network. Neoplasia 15, 85–94 (2013).
Broz, M. L. et al. Dissecting the tumor myeloid compartment reveals rare activating antigen-presenting cells critical for T cell immunity. Cancer Cell 26, 638–652 (2014).
Peranzoni, E. et al. Macrophages impede CD8 T cells from reaching tumor cells and limit the efficacy of anti-PD-1 treatment. Proc. Natl. Acad. Sci. USA 115, E4041–E4050 (2018).
Bao, X. et al. A multiomics analysis-assisted deep learning model identifies a macrophage-oriented module as a potential therapeutic target in colorectal cancer. Cell Rep. Med. 5, 101399 (2024).
Kersten, K. et al. Spatiotemporal co-dependency between macrophages and exhausted CD8(+) T cells in cancer. Cancer Cell 40, 624–38.e9 (2022).
Diskin, B. et al. PD-L1 engagement on T cells promotes self-tolerance and suppression of neighboring macrophages and effector T cells in cancer. Nat. Immunol. 21, 442–454 (2020).
Cotechini T., Atallah A., Grossman A. Tissue-Resident and Recruited Macrophages in Primary Tumor and Metastatic Microenvironments: Potential Targets in Cancer Therapy. Cells. 10.(2021).
Wang, C. et al. MAPT promoter CpG island hypermethylation is associated with poor prognosis in patients with stage II colorectal cancer. Cancer Manag Res 11, 7337–7343 (2019).
Pusztai, L. et al. Evaluation of microtubule-associated protein-Tau expression as a prognostic and predictive marker in the NSABP-B 28 randomized clinical trial. J. Clin. Oncol. 27, 4287–4292 (2009).
Mosaad, Y. M. Clinical Role of Human Leukocyte Antigen in Health and Disease. Scand. J. Immunol. 82, 283–306 (2015).
Iafolla M. A. J. et al. Predicting Toxicity and Response to Pembrolizumab Through Germline Genomic HLA Class 1 Analysis. JNCI Cancer Spectr. 5 (2021).
Correale P. et al. Distinctive germline expression of class I human leukocyte antigen (HLA) alleles and DRB1 heterozygosis predict the outcome of patients with non-small cell lung cancer receiving PD-1/PD-L1 immune checkpoint blockade. J Immunother Cancer. 8.(2020).
Yang, Y. et al. Updated Overall Survival Data and Predictive Biomarkers of Sintilimab Plus Pemetrexed and Platinum as First-Line Treatment for Locally Advanced or Metastatic Nonsquamous NSCLC in the Phase 3 ORIENT-11 Study. J. Thorac. Oncol. 16, 2109–2120 (2021).
Mei, J. et al. HLA class II molecule HLA-DRA identifies immuno-hot tumors and predicts the therapeutic response to anti-PD-1 immunotherapy in NSCLC. BMC Cancer 22, 738 (2022).
Senosain, M. F. et al. HLA-DR cancer cells expression correlates with T cell infiltration and is enriched in lung adenocarcinoma with indolent behavior. Sci. Rep. 11, 14424 (2021).
Gallois, C. et al. Prognostic Impact of Early Treatment and Oxaliplatin Discontinuation in Patients With Stage III Colon Cancer: An ACCENT/IDEA Pooled Analysis of 11 Adjuvant Trials. J. Clin. Oncol. 41, 803–815 (2023).
Antoniotti, C. et al. Upfront FOLFOXIRI plus bevacizumab with or without atezolizumab in the treatment of patients with metastatic colorectal cancer (AtezoTRIBE): a multicentre, open-label, randomised, controlled, phase 2 trial. Lancet Oncol. 23, 876–887 (2022).
Lenz H. J. et al. Modified FOLFOX6 plus bevacizumab with and without nivolumab for first-line treatment of metastatic colorectal cancer: phase 2 results from the CheckMate 9X8 randomized clinical trial. J Immunother Cancer. 12.(2024).
Hu, H. et al. Perioperative chemotherapy with mFOLFOX6 or CAPOX for patients with locally advanced colon cancer (OPTICAL): A multicenter, randomized, phase 3 trial. J. Clin. Oncol. 40, 3500 (2022).
Foxtrot Collaborative, G. Feasibility of preoperative chemotherapy for locally advanced, operable colon cancer: the pilot phase of a randomised controlled trial. Lancet Oncol. 13, 1152–1160 (2012).
Li, J. et al. Safety and pharmacokinetics of novel selective vascular endothelial growth factor receptor-2 inhibitor YN968D1 in patients with advanced malignancies. BMC Cancer 10, 529 (2010).
Tian, S. et al. YN968D1 is a novel and selective inhibitor of vascular endothelial growth factor receptor-2 tyrosine kinase with potent activity in vitro and in vivo. Cancer Sci. 102, 1374–1380 (2011).
Dworak, O., Keilholz, L. & Hoffmann, A. Pathological features of rectal cancer after preoperative radiochemotherapy. Int J. Colorectal Dis. 12, 19–23 (1997).
Liu, L. et al. Single-cell analysis reveals HBV-specific PD-1(+)CD8(+) TRM cells in tumor borders are associated with HBV-related hepatic damage and fibrosis in HCC patients. J. Exp. Clin. Cancer Res. 42, 152 (2023).
Fan, J. et al. NKG2D discriminates diverse ligands through selectively mechano-regulated ligand conformational changes. EMBO J. 41, e107739 (2022).
Sun, X. et al. A pathologically friendly strategy for determining the organ-specific spatial tumor microenvironment topology in lung adenocarcinoma through the integration of snRandom-seq and imaging mass cytometry. Adv. Sci. (Weinh.) 11, e2308892 (2024).
Du, J. et al. Selective oxidative protection leads to tissue topological changes orchestrated by macrophage during ulcerative colitis. Nat. Commun. 14, 3675 (2023).
Windhager, J. et al. An end-to-end workflow for multiplexed image processing and analysis. Nat. Protoc. 18, 3565–3613 (2023).
Meier, F. et al. diaPASEF: parallel accumulation-serial fragmentation combined with data-independent acquisition. Nat. Methods 17, 1229–1236 (2020).
Kong, A. T., Leprevost, F. V., Avtonomov, D. M., Mellacheruvu, D. & Nesvizhskii, A. I. MSFragger: ultrafast and comprehensive peptide identification in mass spectrometry-based proteomics. Nat. Methods 14, 513–520 (2017).
Yu, F. et al. Fast quantitative analysis of timsTOF PASEF data with MSFragger and IonQuant. Mol. Cell Proteom. 19, 1575–1585 (2020).
Demichev, V., Messner, C. B., Vernardis, S. I., Lilley, K. S. & Ralser, M. DIA-NN: neural networks and interference correction enable deep proteome coverage in high throughput. Nat. Methods 17, 41–44 (2020).
Hänzelmann, S., Castelo, R. & Guinney, J. GSVA: gene set variation analysis for microarray and RNA-seq data. BMC Bioinforma. 14, 7 (2013).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Yu, G., Wang, L. G., Han, Y. & He, Q. Y. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16, 284–287 (2012).
Hopewell, S. et al. CONSORT 2025 statement: updated guideline for reporting randomised trials. BMJ 389, e081123 (2025).
Acknowledgements
We appreciate our gratitude to Prof. Jun Li for her valuable work in pathological analysis and to Prof. Bin Qi for his role in radiological tumor evaluation. This work was supported by the National Natural Science Foundation of China (82203186, 82373428), The “Pioneer” and “Leading Goose” R&D Program of Zhejiang (2025C02073), Noncommunicable Chronic Diseases-National Science and Technology Major Project (2023ZD0501600), and Beijing Xisike Clinical Oncology Research Foundation (Y-NESTLE2022QN-0345). The funder played no role in study design, data collection, analysis, and interpretation of data, or the writing of this manuscript.
Author information
Authors and Affiliations
Contributions
Zhou Tong, Weijia Fang-literature search, study design, writing; Xuanwen Bao, Liaoliao Gao- data analysis, data interpretation; Danyang Wang, Wenbin Chen - patients enrollment; Xiaomeng Dai, Hangyu Zhang, Lulu Liu -figures, data collection; Yang Gao, Xudong Zhu- patient management and treatment; Yi Zheng, Qihan Fu- data analyses; Peng Zhao, Fanlong Liu- study design, revised the manuscript; Xiangming Xu, Weijia Fang - study design, supervised the research.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tong, Z., Gao, L., Wang, D. et al. Neoadjuvant mFOLFOX6 plus camrelizumab and apatinib in colon cancer and biomarker research via spatiotemporal-omics profiling. npj Precis. Onc. (2026). https://doi.org/10.1038/s41698-026-01387-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41698-026-01387-0