Abstract
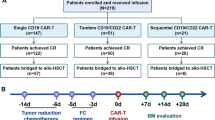
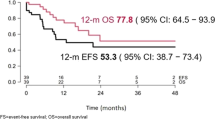
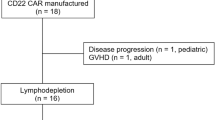
Patients progressing after CD19-targeted immunotherapy in r/r B-ALL experience poor outcomes. CD22-targeted therapies, including CD22 CAR-T cells and Inotuzumab Ozogamicin, show promise as alternatives, although data in those patients is limited. This study retrospectively analyzed 43 r/r B-ALL patients who had previously received CD19-targeted therapy at two centers in China. Among these patients, 27.9% received blinatumomab, 58.1% received CD19 CAR-T cells, and 14% received both. After CD19-targeted therapy, 34.9% of patients experienced CD19-negative relapse, while the remaining patients maintained CD19 expression. Subsequent treatments included CD22 CAR-T cells (55.8%) and InO (44.2%). The median age was 39 (24–56) years, with an overall CR/CRi rate of 54% and 35.1% achieving MRD negativity. Among the 22 patients achieving CR/CRi, 13 (59.1%) experienced relapse. The median relapse-free survival (RFS) was 236 days (95% CI: 132–unreached), and the median OS has not been reached. Multivariate analysis showed similar remission rates and survival for CD22 CAR-T and Inotuzumab Ozogamicin therapies. Patients with extramedullary disease had worse remission rates, and those previously resistant to CD19-targeted therapy had shorter RFS. CD22-targeted therapies offer a potential option for patients progressing after CD19-targeted immunotherapy, but high relapse rates highlight the need for better strategies for lasting remission.
Similar content being viewed by others
Data availability
The data sets used and analyzed during the current study are available from the corresponding author on reasonable request.
References
Topp, M. S. et al. Phase II trial of the anti-CD19 bispecific T cell-engager blinatumomab shows hematologic and molecular remissions in patients with relapsed or refractory B-precursor acute lymphoblastic leukemia. J. Clin. Oncol. 32, 4134–4140 (2014).
Kantarjian, H. et al. Blinatumomab versus chemotherapy for advanced acute lymphoblastic leukemia. N. Engl. J. Med. 376, 836–847 (2017).
Maude, S. L. et al. Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. N. Engl. J. Med. 378, 439–448 (2018).
Shah, B. D. et al. KTE-X19 for relapsed or refractory adult B-cell acute lymphoblastic leukaemia: phase 2 results of the single-arm, open-label, multicentre ZUMA-3 study. Lancet 398, 491–502 (2021).
Roddie, C. et al. Durable responses and low toxicity after fast off-rate CD19 chimeric antigen receptor-T therapy in adults with relapsed or refractory B-cell acute lymphoblastic leukemia. J. Clin. Oncol. 39, 3352–3363 (2021).
Roddie, C. et al. Obecabtagene autoleucel in adults with B-cell acute lymphoblastic leukemia. N. Engl. J. Med. 391, 2219–2230 (2024).
Wudhikarn, K. et al. Interventions and outcomes of adult patients with B-ALL progressing after CD19 chimeric antigen receptor T-cell therapy. Blood 138, 531–543 (2021).
Shah, N. N. et al. Long-term follow-up of CD19-CAR T-cell therapy in children and young adults with B-ALL. J. Clin. Oncol. 39, 1650–1659 (2021).
Park, J. H. et al. Long-term follow-up of CD19 CAR therapy in acute lymphoblastic leukemia. N. Engl. J. Med. 378, 449–459 (2018).
Laetsch, T. W. et al. Three-year update of tisagenlecleucel in pediatric and young adult patients with relapsed/refractory acute lymphoblastic leukemia in the ELIANA trial. J. Clin. Oncol. 41, 1664–1669 (2023).
Laetsch, T. W. et al. Patient-reported quality of life after tisagenlecleucel infusion in children and young adults with relapsed or refractory B-cell acute lymphoblastic leukaemia: a global, single-arm, phase 2 trial. Lancet Oncol. 20, 1710–1718 (2019).
Lee, D. W. et al. T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet 385, 517–528 (2015).
Sotillo, E. et al. Convergence of acquired mutations and alternative splicing of CD19 enables resistance to CART-19 immunotherapy. Cancer Discov. 5, 1282–1295 (2015).
Song, F. et al. Safety and efficacy of autologous and allogeneic humanized CD19-targeted CAR-T cell therapy for patients with relapsed/refractory B-ALL. J. Immunotherapy Cancer 11, https://doi.org/10.1136/jitc-2022-005701 (2023).
Aldoss, I. et al. Correlates of resistance and relapse during blinatumomab therapy for relapsed/refractory acute lymphoblastic leukemia. Am. J. Hematol. 92, 858–865 (2017).
Topp, M. S. et al. Targeted therapy with the T-cell-engaging antibody blinatumomab of chemotherapy-refractory minimal residual disease in B-lineage acute lymphoblastic leukemia patients results in high response rate and prolonged leukemia-free survival. J. Clin. Oncol. 29, 2493–2498 (2011).
Raponi, S. et al. Flow cytometric study of potential target antigens (CD19, CD20, CD22, CD33) for antibody-based immunotherapy in acute lymphoblastic leukemia: analysis of 552 cases. Leuk. lymphoma 52, 1098–1107 (2011).
Kantarjian, H. M. et al. Inotuzumab ozogamicin versus standard therapy for acute lymphoblastic leukemia. N. Engl. J. Med. 375, 740–753 (2016).
Fry, T. J. et al. CD22-targeted CAR T cells induce remission in B-ALL that is naive or resistant to CD19-targeted CAR immunotherapy. Nat. Med. 24, 20–28 (2018).
Pan, J. et al. CD22 CAR T-cell therapy in refractory or relapsed B acute lymphoblastic leukemia. Leukemia 33, 2854–2866 (2019).
Aldoss, I. et al. TP53 mutations are associated with CD19- relapse and inferior outcomes after blinatumomab in adults with ALL. Blood Adv. 9, 2159–2172 (2025).
Lamble, A. J. et al. Preinfusion factors impacting relapse immunophenotype following CD19 CAR T cells. Blood Adv. 7, 575–585 (2023).
Gauthier, J. et al. Factors associated with outcomes after a second CD19-targeted CAR T-cell infusion for refractory B-cell malignancies. Blood 137, 323–335 (2021).
Aldea, M. et al. Overcoming resistance to tumor-targeted and immune-targeted therapies. Cancer Discov. 11, 874–899 (2021).
Frey, N. V. et al. Optimizing chimeric antigen receptor T-cell therapy for adults with acute lymphoblastic leukemia. J. Clin. Oncol. 38, 415–422 (2020).
Pennesi, E. et al. Inotuzumab ozogamicin as single agent in pediatric patients with relapsed and refractory acute lymphoblastic leukemia: results from a phase II trial. Leukemia 36, 1516–1524 (2022).
Kantarjian, H. M. et al. Inotuzumab ozogamicin for relapsed/refractory acute lymphoblastic leukemia in the INO-VATE trial: CD22 pharmacodynamics, efficacy, and safety by baseline CD22. Clin. Cancer Res. 27, 2742–2754 (2021).
Bhojwani, D. et al. Inotuzumab ozogamicin in pediatric patients with relapsed/refractory acute lymphoblastic leukemia. Leukemia 33, 884–892 (2019).
Zhao, Y. et al. Genomic determinants of response and resistance to inotuzumab ozogamicin in B-cell ALL. Blood, https://doi.org/10.1182/blood.2024023930 (2024).
Ma, F. et al. Evidence of long-lasting anti-CD19 activity of engrafted CD19 chimeric antigen receptor-modified T cells in a phase I study targeting pediatrics with acute lymphoblastic leukemia. Hematol. Oncol. 37, 601–608 (2019).
Therneau, T. M., Crowson, C. S. & Atkinson, E. J. Adjusted Survival Curves. survival package vignette, CRAN. https://cran.r-project.org/web/packages/survival/vignettes/adjcurve.pdf (2015).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant numbers 82370223 and 82425002).
Author information
Authors and Affiliations
Contributions
GW, XZ, HH, and YH designed the study. FS analyzed the data and drafted the manuscript. JY, MZ, SF, JF, RH, YL, and AHC provided subject data and performed data analysis. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Song, F., Yang, J., Zhang, M. et al. CD22-targeted immunotherapy for B-cell acute lymphoblastic leukemia progressing following CD19-targeted immunotherapy. npj Precis. Onc. (2026). https://doi.org/10.1038/s41698-026-01413-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41698-026-01413-1