Fig. 1: Phases, attrition rate, and investment in ophthalmic drug discovery and development.
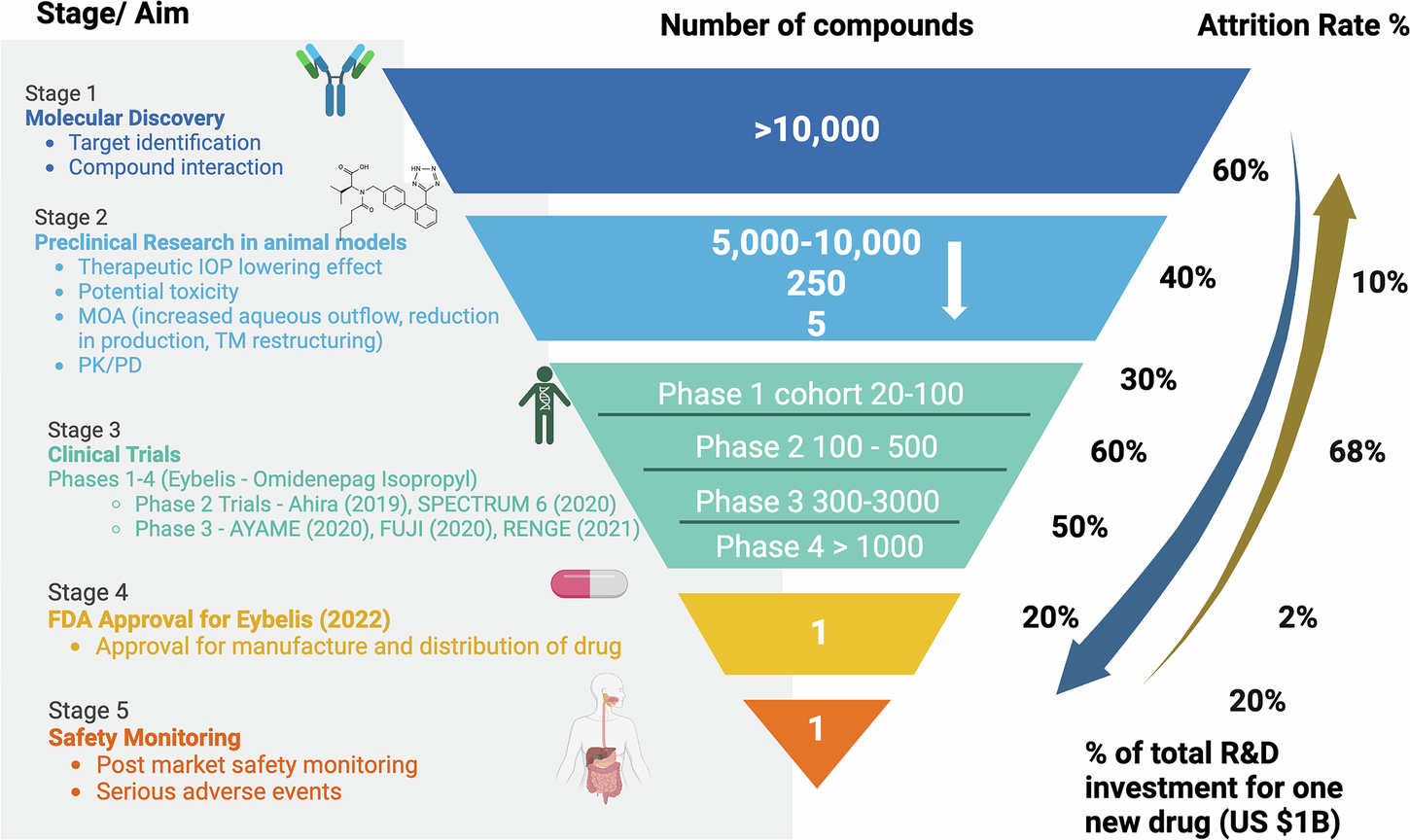
This figure outlines the key stages, attrition rates, and financial investment required in the drug discovery and development process, using ophthalmology as an example. The process begins with target identification and compound screening, followed by preclinical studies assessing efficacy, toxicity, and PK/PD using in silico, in vitro, and in vivo models. Clinical development progresses through Phases I to IV, involving increasing cohort sizes to evaluate safety, dosage, efficacy, and long-term outcomes. Each stage faces significant attrition, with fewer than 1 in 10,000 compounds reaching approval. The average cost per successful drug has been estimated at $172.7 million (2000–2018). Post-market surveillance continues to assess safety and adverse events after regulatory approval. An ophthalmic case study of omidenepag isopropyl (Eybelis), a topical treatment for IOP reduction in glaucoma and ocular hypertension, exemplifies this pipeline. This structured process ensures that only the most promising and safe treatments reach patients. PK/PD pharmacokinetics/pharmacodynamics, FDA Food and Drug Administration, IOP intraocular pressure. Figure created with BioRender.com.
