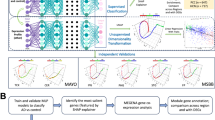
Abstract
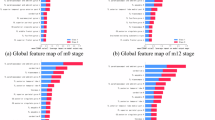
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder increasingly associated with peripheral inflammatory conditions such as chronic periodontitis (CP); however, the molecular mechanisms linking these conditions remain poorly understood. Here, we investigated the therapeutic effects of Huanglian Jieddu Decoction (HLJDD) on CP-induced AD using an integrative machine learning-guided multi-omics approach. Analysis of public single-cell RNA-sequencing data revealed pronounced inflammatory activation in microglia from AD samples. We further established a CP-induced AD rat model and performed hippocampal transcriptomic profiling. Multiple complementary machine learning strategies, including Random Forest-based feature selection, support vector machine-based refinement, network modeling, and interpretable model analysis, were applied to prioritize disease-relevant pathways from high-dimensional transcriptomic data. Across models, components of the cGAS–STING signaling pathway consistently exhibited strong and directional contributions to CP–AD pathology, indicating a central inflammatory axis linking peripheral infection to neurodegeneration. Guided by these data-driven insights, in vivo and in vitro experiments demonstrated that HLJDD suppressed cGAS–STING activation, attenuated neuroinflammation, and improved cognitive function in CP-induced AD models. Collectively, this study highlights the value of machine learning-assisted transcriptomic interpretation for mechanistic prioritization and identifies HLJDD as a multitarget therapeutic strategy for CP-induced AD.
Similar content being viewed by others
Data availability
The single-cell RNA sequencing data analyzed in this study were retrieved from the GEO database under accession number GSE157827. Bulk RNA sequencing data for periodontitis were obtained from GEO under accession number GSE23586. The rat hippocampal RNA sequencing data generated in this study have been deposited in the NCBI Sequence Read Archive (SRA) under accession number PRJNA1394501. All other data supporting the findings of this study are available from the corresponding authors upon reasonable request.
Code availability
All custom code used for data preprocessing, statistical analysis, and machine learning modeling in this study was written in R (v4.1.0). Analyses were performed using established R packages including Seurat (v4.3.0), ggplot2 (v3.4.0), clusterProfiler, randomForest, Boruta, e1071, caret, neuralnet, bnlearn, ropls, and UpSetR. The code is available from the corresponding authors upon reasonable request.
References
Scheltens, P. et al. Alzheimer’s disease. Lancet 388, 505–517 (2016).
Wang, J., Gu, B. J., Masters, C. L. & Wang, Y.-J. A systemic view of Alzheimer’s disease - insights from amyloid-β metabolism beyond the brain. Nat. Rev. Neurol 13, 612–623 (2017).
Toden, S. et al. Noninvasive characterization of Alzheimer’s disease by circulating, cell-free messenger RNA next-generation sequencing. Sci. Adv. 6, eabb1654 (2020).
Im, D. & Kim, H. I. Kinetic modulation of amyloid-β (1-42) fibrillation and alleviated cytotoxicity with rationally designed point mutants. Alzheimer’s Dement. 19, e062215 (2023).
Arévalo-Caro, C. et al. APOE4, Alzheimer’s and periodontal disease: a scoping review. Ageing Res. Rev 105, 102649 (2025).
Madej, M. et al. Structural and functional insights into oligopeptide acquisition by the RagAB transporter from Porphyromonas gingivalis. Nat. Microbiol. 5, 1016–1025 (2020).
Chen, H. et al. Age- and sex-related differences of periodontal bone resorption, cognitive function, and immune state in APP/PS1 murine model of Alzheimer’s disease. J. Neuroinflamm. 20, 153 (2023).
Qian, X. et al. Intestinal homeostasis disrupted by periodontitis exacerbates Alzheimer’s disease in APP/PS1 mice. J. Neuroinflamm. 21, 263 (2024).
Dominy, S. S. et al. Porphyromonas gingivalis in Alzheimer’s disease brains: evidence for disease causation and treatment with small-molecule inhibitors. Sci. Adv. 5, eaau3333 (2019).
Cheng, X. et al. Exogenous monocyte myeloid-derived suppressor cells ameliorate immune imbalance, neuroinflammation and cognitive impairment in 5xFAD mice infected with Porphyromonas gingivalis. J Neuroinflamm. 20, 55 (2023).
Lei, S. et al. Porphyromonas gingivalis bacteremia increases the permeability of the blood–brain barrier via the Mfsd2a/caveolin-1 mediated transcytosis pathway. Int. J. Oral Sci. 15, 3 (2023).
Li, Z., Wang, H. & Yin, Y. Peripheral inflammation is a potential etiological factor in Alzheimer’s disease. Rev. Neurosci. 35, 99–120 (2024).
Lunar Silva, I. & Cascales, E. Molecular strategies underlying Porphyromonas gingivalis virulence. J. Mol. Biol. 433, 166836 (2021).
Ishida, N. et al. Periodontitis induced by bacterial infection exacerbates features of Alzheimer’s disease in transgenic mice. NPJ Aging Mech. Dis. 3, 15 (2017).
Hu, Y. et al. Periodontitis induced by P. gingivalis-LPS is associated with neuroinflammation and learning and memory impairment in Sprague-Dawley rats. Front. Neurosci. 14, 658 (2020).
Ma, X., Shin, Y.-J., Yoo, J.-W., Park, H.-S. & Kim, D.-H. Extracellular vesicles derived from Porphyromonas gingivalis induce trigeminal nerve-mediated cognitive impairment. J. Adv. Res. 54, 293–303 (2023).
Hu, Y. et al. Activated STAT3 signaling pathway by ligature-induced periodontitis could contribute to neuroinflammation and cognitive impairment in rats. J. Neuroinflamm. 18, 80 (2021).
Zhang, J. et al. Porphyromonas gingivalis lipopolysaccharide induces cognitive dysfunction, mediated by neuronal inflammation via activation of the TLR4 signaling pathway in C57BL/6 mice. J. Neuroinflamm. 15, 37 (2018).
Chen, Q., Sun, L. & Chen, Z. J. Regulation and function of the cGAS-STING pathway of cytosolic DNA sensing. Nat. Immunol. 17, 1142–1149 (2016).
Sun, L., Wu, J., Du, F., Chen, X. & Chen, Z. J. Cyclic GMP-AMP synthase is a cytosolic DNA sensor that activates the type I interferon pathway. Science 339, 786–791 (2013).
Hou, Y. et al. NAD+ supplementation reduces neuroinflammation and cell senescence in a transgenic mouse model of Alzheimer’s disease via cGAS–STING. Proc. Natl. Acad. Sci. USA. 118, e2011226118 (2021).
Sharma, M., Rajendrarao, S., Shahani, N., Ramírez-Jarquín, U. N. & Subramaniam, S. Cyclic GMP-AMP synthase promotes the inflammatory and autophagy responses in Huntington disease. Proc. Natl. Acad. Sci. USA 117, 15989–15999 (2020).
Cordova, A. F., Ritchie, C., Böhnert, V. & Li, L. Human SLC46A2 is the dominant cGAMP importer in extracellular cGAMP-sensing macrophages and monocytes. ACS Cent. Sci. 7, 1073–1088 (2021).
He, S. et al. Microglial cGAS deletion preserves intercellular communication and alleviates amyloid-β-induced pathogenesis of Alzheimer’s disease. Adv Sci 12, e2410910 (2025).
Quan, S. et al. The neuroimmune nexus: unraveling the role of the mtDNA–cGAS–STING signal pathway in Alzheimer’s disease. Mol. Neurodegener. 20, 25 (2025).
Fazal, F. et al. cGAS-STING signaling in Alzheimer’s disease: microglial mechanisms and therapeutic opportunities. Mol. Aspects Med. 107, 101444 (2026).
Bi, R. et al. Porphyromonas gingivalis induces an inflammatory response via the cGAS–STING signaling pathway in a periodontitis mouse model. Front. Microbiol. 14, 1183415 (2023).
Wang, T. et al. IGF2 promotes alveolar bone regeneration in murine periodontitis via inhibiting cGAS/STING-mediated M1 macrophage polarization. Int. Immunopharmacol. 132, 111984 (2024).
Zhang, Y. et al. Deciphering the pharmacological mechanism of the Chinese formula Huanglian-Jie-Du decoction in the treatment of ischemic stroke using a systems biology-based strategy. Acta Pharmacol. Sin. 36, 724–733 (2015).
Shang, J. et al. Systems pharmacology, proteomics and in vivo studies identification of mechanisms of cerebral ischemia injury amelioration by Huanglian Jiedu decoction. J. Ethnopharmacol. 293, 115244 (2022).
Gu, X. et al. Huanglian Jiedu decoction remodels the periphery microenvironment to inhibit Alzheimer’s disease progression based on the “brain–gut” axis through multiple integrated omics. Alzheimer’s Res. Ther. 13, 44 (2021).
Qi, Y.-Y. et al. Involvement of Huanglian Jiedu decoction on microglia with abnormal sphingolipid metabolism in Alzheimer’s disease. Drug Des. Dev. Ther. 16, 931–950 (2022).
Zhuang, G.-D. et al. Huang-Lian-Jie-Du decoction alleviates diabetic encephalopathy by regulating inflammation and pyroptosis via suppression of AGEs/RAGE/NF-κB pathways. J. Ethnopharmacol. 337, 118787 (2025).
Zhang, F. et al. Effects of Huanglian Jiedu decoration in rat gingivitis. Evid. Based Complement. Alternat. Med. 2018, 8249013 (2018).
Zhang, R. et al. Berberine promotes osteogenic differentiation of mesenchymal stem cells with therapeutic potential in periodontal regeneration. Eur. J. Pharmacol. 851, 144–150 (2019).
Sun, J.-Y. et al. Baicalin inhibits toll-like receptor 2/4 expression and downstream signaling in rat experimental periodontitis. Int. Immunopharmacol. 36, 86–93 (2016).
Tian, S. et al. The application of in silico drug-likeness predictions in pharmaceutical research. Adv. Drug Deliv. Rev. 86, 2–10 (2015).
Hong, Y. et al. The integration of machine learning into traditional Chinese medicine. J. Pharm. Anal. 15, 101157 (2025).
Gao, Y. et al. Dual inhibitors of histone deacetylases and other cancer-related targets: a pharmacological perspective. Biochem. Pharmacol. 182, 114224 (2020).
Hu, L. et al. Dual-channel hypergraph convolutional network for predicting herb-disease associations. Brief. Bioinf. 25, bbae067 (2024).
Fu, Y. et al. Deep learning-based network pharmacology for exploring the mechanism of licorice for the treatment of COVID-19. Sci. Rep. 13, 5844 (2023).
Kamer, A. R. et al. Periodontal disease associates with higher brain amyloid load in normal elderly. Neurobiol. Aging 36, 627–633 (2015).
Kaye, E. K. et al. Tooth loss and periodontal disease predict poor cognitive function in older men. J. Am. Geriatr. Soc. 58, 713–718 (2010).
Kong, L. et al. Periodontitis-induced neuroinflammation triggers IFITM3-aβ axis to cause Alzheimer’s disease-like pathology and cognitive decline. Alzheimer’s Res. Ther. 17, 166 (2025).
Nie, R. et al. Porphyromonas gingivalis infection induces amyloid-β accumulation in monocytes/macrophages. J. Alzheimer’s Dis. 72, 479–494 (2019).
Chacón, T. & Hernández-Hincapié, H. Relationship periodontitis and Alzheimer’s disease: relevant aspects from an epigenetic view. J. Alzheimer’s Dis. 109, 499–525 (2026).
Deng, L. et al. STING-dependent cytosolic DNA sensing promotes radiation-induced type I interferon-dependent antitumor immunity in immunogenic tumors. Immunity 41, 843–852 (2014).
Naguib, S. et al. The R136S mutation in the APOE3 gene confers resilience against tau pathology via inhibition of the cGAS–STING–IFN pathway. Immunity 58, 1931–1947.e9 (2025).
Chung, S. Blockade of STING activation alleviates microglial dysfunction and a broad spectrum of Alzheimer’s disease pathologies. Exp. Mol. Med. 56, 1936–1951 (2024).
Yang, N.-S. -Y. et al. mtDNA–cGAS–STING axis-dependent NLRP3 inflammasome activation contributes to postoperative cognitive dysfunction induced by sevoflurane in mice. Int. J. Biol. Sci. 20, 1927–1946 (2024).
Carling, G. K. et al. Alzheimer’s disease-linked risk alleles elevate microglial cGAS-associated senescence and neurodegeneration in a tauopathy model. Neuron 112, 3877–3896.e8 (2024).
Tian, L. et al. Nanoimmunomodulation of the aβ-STING feedback machinery in microglia for Alzheimer’s disease treatment. Proc. Natl. Acad. Sci. USA. 122, e2427257122 (2025).
Gu, X. et al. Effects of Huang-Lian-Jie-Du decoction on oxidative stress and AMPK-SIRT1 pathway in Alzheimer’s disease rat. Evid.-based Complement. Altern. Med. 2020, 6212907 (2020).
Wang, P. et al. Intermodule coupling analysis of Huang-Lian-Jie-Du decoction on stroke. Front. Pharmacol. 10, 1288 (2019).
Zhang, Z.-T. et al. Rapid screening of neuroprotective components from Huang-Lian-Jie-Du decoction by living cell biospecific extraction coupled with HPLC-Q-orbitrap-HRMS/MS analysis. J. Chromatogr. B 1176, 122764 (2021).
Xu, D., Lv, Y., Wang, J., Yang, M. & Kong, L. Deciphering the mechanism of Huang-Lian-Jie-Du-decoction on the treatment of sepsis by formula decomposition and metabolomics: enhancement of cholinergic pathways and inhibition of HMGB-1/TLR4/NF-κB signaling. Pharmacol. Res. 121, 94–113 (2017).
Lu, J., Wang, J.-S. & Kong, L.-Y. Anti-inflammatory effects of Huang-Lian-Jie-Du decoction, its two fractions and four typical compounds. J. Ethnopharmacol. 134, 911–918 (2011).
Lee, I.-S. et al. Human neural stem cells alleviate Alzheimer-like pathology in a mouse model. Mol. Neurodegener. 10, 38 (2015).
Leuzy, A. et al. Pittsburgh compound B imaging and cerebrospinal fluid amyloid-β in a multicentre European memory clinic study. Brain: J. Neurol. 139, 2540–2553 (2016).
Chen, H. et al. Applications of artificial intelligence in the research of molecular mechanisms of traditional Chinese medicine formulas. Chin. J. Nat. Med. 23, 1329–1341 (2025).
Ma, J. et al. Machine learning-assisted analysis of serum metabolomics and network pharmacology reveals the effective compound from herbal formula against alcoholic liver injury. Chin. Med. 20, 48–71 (2025).
Kuang, J. et al. Machine learning analysis reveals tumor heterogeneity and stromal-immune niches in breast cancer. npj Digit. Med. 8, 565–579 (2025).
Zheng, X. et al. Bioactive components of Jiedu Sangen decoction against colorectal cancer: a novel and comprehensive research strategy for natural drug development. Phytomedicine 142, 156795 (2025).
Ali, S., Tian, X., Chen, H. & Zhou, J. A new era of artificial intelligence (AI): transforming drug discovery and development. J. Med. Chem. 68, 23643–23652 (2025).
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (No. 82174358) and the 2024 Joint Innovation Foundation of Chengdu University of Traditional Chinese Medicine (Young Leading Talent Program) (No. WXLH20240302).
Author information
Authors and Affiliations
Contributions
All the authors read and approved the manuscript. Wenbin Wu, Guohua Zhao, Yuzhen Xu, and Jie Li designed and supervised the studies. Jie Li and Mingqi Chen performed the experiments with the help of Pan Ren, Furong Zhong, Guangming Sun, Yue Zhu, Yiran Fan, Jinxin Chen, Manru Xu, Mengyuan Qiao, and Ganggang Li. Jie Li and Guangming Sun wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, J., Chen, M., Ren, P. et al. Machine learning–guided Huanglian Jiedu decoction targets STING in periodontitis-induced Alzheimer’s Disease. npj Digit. Med. (2026). https://doi.org/10.1038/s41746-026-02468-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41746-026-02468-x