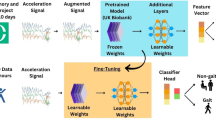
Abstract
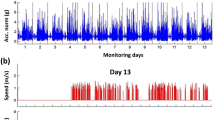
Physical activity and mobility are critical for healthy aging and predict diverse health outcomes. While wrist-worn accelerometers are widely used to monitor physical activity, estimating gait metrics from wrist data remains challenging. We extend ElderNet, a self-supervised deep-learning model previously validated for walking-bout detection, to estimate gait metrics from wrist accelerometry. Validation involved 819 older adults (Rush-Memory-and-Aging-Project) and 85 individuals with gait impairments (Mobilise-D), from six medical centers. In Mobilise-D, ElderNet achieved an absolute error of 8.82 cm/s and an intra-class correlation of 0.87 for gait speed, outperforming state-of-the-art methods (p < 0.001) and models using a lower-back sensor. ElderNet outperformed (percentage error; p < 0.01) competing approaches in estimating cadence and stride length, and better (p < 0.01) classified mobility disability (AUC = 0.80) than conventional gait or physical activity metrics. These results demonstrate the potential of ElderNet a scalable tool for gait assessment using wrist-worn devices in aging and clinical populations.
Similar content being viewed by others
Data availability
The dataset from the Mobilise-D technical validation study (Dataset 3) can be found on Zenodo: https://doi.org/10.5281/zenodo.13899386. All other data and related algorithms (i.e., Dataset 2) included in these analyses are available via the Rush Alzheimer’s Disease Center Research Resource Sharing Hub, which can be found at www.radc.rush.edu. It has descriptions of the studies and available data. Any qualified investigator can create an account and submit requests for de-identified data.
Code availability
The underlying code for this study is available on Github and can be accessed via this link: https://github.com/yonbrand/gait-quality/tree/main.
References
Reijnierse, E. M. et al. Towards a core-set of mobility measures in ageing research: the need to define mobility and its constructs. BMC Geriatr. 23, 220 (2023).
Sabia, S. et al. Association between questionnaire- and accelerometer-assessed physical activity: the role of sociodemographic factors. Am. J. Epidemiol. 179, 781–790 (2014).
Buchman, A. S. et al. Physical activity, common brain pathologies, and cognition in community-dwelling older adults. Neurology 92, E811–E822 (2019).
Zou, Q. et al. Longitudinal association between physical activity, blood lipids, and risk of dyslipidemia among Chinese adults: findings from the China Health and Nutrition Surveys in 2009 and 2015. Nutrients 15, 341 (2023).
Buchman, A. S. Total daily physical activity and longevity in old age. Arch. Intern Med. 172, 444–450 (2012).
Doherty, A. et al. Large scale population assessment of physical activity using wrist worn accelerometers: the UK Biobank study. PLoS ONE 12, e0169649 (2017).
Zhao, A., Cui, E., Leroux, A., Lindquist, M. A. & Crainiceanu, C. M. Evaluating the prediction performance of objective physical activity measures for incident Parkinson’s disease in the UK Biobank. J. Neurol. 270, 5913–5923 (2023).
Leroux, A. et al. Organizing and analyzing the activity data in NHANES. Stat. Biosci. 11, 262–287 (2019).
Willetts, M., Hollowell, S., Aslett, L., Holmes, C. & Doherty, A. Statistical machine learning of sleep and physical activity phenotypes from sensor data in 96,220 UK Biobank participants. Sci. Rep. 8, 7961 (2018).
Lin, W. et al. Can gait characteristics be represented by physical activity measured with wrist-worn accelerometers?. Sensors 23, 8542 (2023).
Studenski, S. Gait speed and survival in older adults. JAMA 305, 50–58 (2011).
Dumurgier, J. et al. Gait speed and decline in gait speed as predictors of incident. Dement. J. Gerontol. Ser. A 72, 655–661 (2017).
Chan, L. L. Y., Espinoza Cerda, M. T., Brodie, M. A., Lord, S. R. & Taylor, M. E. Daily-life walking speed, running duration and bedtime from wrist-worn sensors predict incident dementia: a watch walk – UK biobank study. Int. Psychogeriatr. 100031. https://doi.org/10.1016/J.INPSYC.2024.100031 (2025).
Weiss, A. et al. Toward automated, at-home assessment of mobility among patients with Parkinson disease, using a body-worn accelerometer. Neurorehabilit. Neural Repair 25, 810–818 (2011).
Zadka, A. et al. A wearable sensor and machine learning estimate step length in older adults and patients with neurological disorders. npj Digit. Med. 7, 142 (2024).
Chan, L. L. Y., Choi, T. C. M., Lord, S. R. & Brodie, M. A. Development and large-scale validation of the Watch Walk wrist-worn digital gait biomarkers. Sci. Rep. 12, 16211 (2022).
Hillel, I. et al. Is every-day walking in older adults more analogous to dual-task walking or to usual walking? Elucidating the gaps between gait performance in the lab and during 24/7 monitoring. Eur. Rev. Aging Phys. Act. 16, 6 (2019).
Warmerdam, E. et al. Long-term unsupervised mobility assessment in movement disorders. Lancet Neurol. 19, 462–470 (2020).
Hausdorff, J. M. et al. Everyday stepping quantity and quality among older adult fallers with and without mild cognitive impairment: Initial evidence for new motor markers of cognitive deficits?. J. Gerontol. A Biol. Sci. Med. Sci. 73, 1078–1082 (2018).
Kluge, F. et al. Real-world gait detection using a wrist-worn inertial sensor: validation study. JMIR Form. Res 8, e50035 (2024).
Brand, Y. E. et al. Self-supervised learning of wrist-worn daily living accelerometer data improves the automated detection of gait in older adults. Sci. Rep 14, 20854 (2024).
Bonci, T., Keogh, A., Del Din, S., Scott, K. & Mazzà, C. An objective methodology for the selection of a device for continuous mobility assessment. Sensors 20, 6509 (2020).
Weston, A. R. et al. Turning speed as a more responsive metric of age-related decline in mobility: a comparative study with gait speed. Clin. Biomech. 113, 106196 (2024).
Kirk, C. et al. Mobilise-D insights to estimate real-world walking speed in multiple conditions with a wearable device. Sci. Rep. 14, 1754 (2024).
Adams, J. L. et al. Using a smartwatch and smartphone to assess early Parkinson’s disease in the WATCH-PD study over 12 months. npj Parkinson’s. Dis. 10, 112 (2024).
Yuan, H. et al. Self-supervised learning for human activity recognition using 700,000 person-days of wearable data. npj Digit. Med. 7, 91 (2024).
Acquah, A. et al. Daily steps are a predictor of, but perhaps not a modifiable risk factor for Parkinson’s disease: findings from the UK Biobank. medRxiv. https://doi.org/10.1101/2024.08.13.24311539 (2024).
Small, S. R. et al. Development and validation of a machine learning wrist-worn step detection algorithm with deployment in the UK Biobank. medRxiv. https://doi.org/10.1101/2023.02.20.23285750 (2023).
Shreves, A. H. et al. Amount and intensity of daily total physical activity, step count and risk of incident cancer in the UK Biobank. Br. J. Sports Med. 59, 839–847, https://doi.org/10.1136/BJSPORTS-2024-109360 (2025).
Barker, J. et al. Physical activity of UK adults with chronic disease: cross-sectional analysis of accelerometer-measured physical activity in 96 706 UK Biobank participants. Int J. Epidemiol. 48, 1167–1174 (2019).
Stamatakis, E. et al. Association of wearable device-measured vigorous intermittent lifestyle physical activity with mortality. Nat. Med. 28, 2521–2529 (2022).
Bloomberg, M., Brocklebank, L., Doherty, A., Hamer, M. & Steptoe, A. Associations of accelerometer-measured physical activity, sedentary behaviour, and sleep with next-day cognitive performance in older adults: a micro-longitudinal study. Int. J. Behav. Nutr. Phys. Activity 21, 133 (2024).
Brand, Y. E. et al. Gait detection from a wrist-worn sensor using machine learning methods: a daily living study in older adults and people with Parkinson’s disease. Sensors 22, 7094 (2022).
Soltani, A., Dejnabadi, H., Savary, M. & Aminian, K. Real-world gait speed estimation using wrist sensor: a personalized approach. IEEE J. Biomed. Health Inf. 24, 658–668 (2020).
Urbanek, J. K. et al. Free-living gait cadence measured by wearable accelerometer: a promising alternative to traditional measures of mobility for assessing fall risk. J. Gerontol. Ser. A 78, 802–810 (2023).
Karas, M. et al. Adaptive empirical pattern transformation (ADEPT) with application to walking stride segmentation. Biostatistics 22, 331–347 (2021).
Bennett, D. A. et al. Religious orders study and rush memory and aging project. J. Alzheimer’s. Dis. 64, S161–S189 (2018).
Bennett, D. A. et al. Overview and findings from the rush Memory and Aging Project. Curr. Alzheimer Res. 9, 646–663 (2012).
Mazzà, C. et al. Technical validation of real-world monitoring of gait: a multicentric observational study. BMJ Open 11, e050785 (2021).
Salis, F. et al. A method for gait events detection based on low spatial resolution pressure insoles data. J. Biomech. 127, 110687 (2021).
Salis, F. et al. A multi-sensor wearable system for the assessment of diseased gait in real-world conditions. Front. Bioeng. Biotechnol. 11, 1143248 (2023).
Buchman, A. S., Boyle, P. A., Leurgans, S. E., Evans, D. A. & Bennett, D. A. Pulmonary function, muscle strength, and incident mobility disability in elders. Proc. Am. Thorac. Soc. 6, 581–587 (2009).
Buchman, A. S., Boyle, P. A., Leurgans, S. E., Barnes, L. L. & Bennett, D. A. Cognitive function is associated with the development of mobility impairments in community-dwelling elders. Am. J. Geriatr. Psychiatry 19, 571–580 (2011).
Shema-Shiratzky, S. et al. A wearable sensor identifies alterations in community ambulation in multiple sclerosis: contributors to real-world gait quality and physical activity. J. Neurol. 267, 1912–1921 (2020).
Soltani, A. et al. Real-world gait speed estimation, frailty and handgrip strength: a cohort-based study. Sci. Rep. 11, 18966 (2021).
Tamura, S. et al. Minimal clinically important difference of the short physical performance battery and comfortable walking speed in old-old adults with acute cardiovascular disease: a multicenter, prospective, observational study. Disabil. Rehabil. 45, 1079–1086 (2023).
Bohannon, R. W. & Glenney, S. S. Minimal clinically important difference for change in comfortable gait speed of adults with pathology: a systematic review. J. Eval. Clin. Pr. 20, 295–300 (2014).
Verghese, J., Holtzer, R., Lipton, R. B. & Wang, C. Quantitative gait markers and incident fall risk in older adults. J. Gerontol. Ser. A 64A, 896–901 (2009).
Lord, S., Galna, B. & Rochester, L. Moving forward on gait measurement: toward a more refined approach. Mov. Disord. 28, 1534–1543 (2013).
Asher, E. E. et al. Combining 24-hour continuous monitoring of time-locked heart rate, physical activity and gait in older adults: preliminary findings. Sensors 25, 1945 (2025).
Schalkamp, A. K., Peall, K. J., Harrison, N. A. & Sandor, C. Wearable movement-tracking data identify Parkinson’s disease years before clinical diagnosis. Nat. Med. 29, 2048–2056 (2023).
Mirelman, A., Rochester, L., Simuni, T. & Hausdoff, J. M. Digital mobility measures to predict Parkinson’s disease. Lancet Neurol. 22, 1098–1100 (2023).
Scott, K. et al. Design and validation of a multi-task, multi-context protocol for real-world gait simulation. J. Neuroeng. Rehabil. 19, 141 (2022).
Moe-Nilssen, R. & Helbostad, J. L. Estimation of gait cycle characteristics by trunk accelerometry. J. Biomech 37, 121–126 (2004).
Akiba, T., Sano, S., Yanase, T., Ohta, T. & Koyama, M. Optuna: a next-generation hyperparameter optimization framework. in Proc. ACM SIGKDD International Conference on Knowledge Discovery and Data Mining, 2623–2631 https://doi.org/10.1145/3292500.3330701 (2019).
Hill, E. J. et al. Quantitative mobility measures complement the MDS-UPDRS for characterization of Parkinson’s disease heterogeneity. Parkinsonism Relat. Disord. 84, 105–111 (2021).
Buchman, A. S. et al. Associations between quantitative mobility measures derived from components of conventional mobility testing and Parkinsonian gait in older adults. PLoS ONE 9, e86262 (2014).
van Hees, V. T. et al. Estimation of daily energy expenditure in pregnant and non-pregnant women using a wrist-worn tri-axial accelerometer. PLoS ONE 6, e22922 (2011).
Koo, T. K. & Li, M. Y. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J. Chiropr. Med. 15, 155–163 (2016).
Guralnik, J. M., Ferrucci, L., Simonsick, E. M., Salive, M. E. & Wallace, R. B. Lower-extremity function in persons over the age of 70 years as a predictor of subsequent disability. N. Engl. J. Med. 332, 556–561 (1995).
File:Homoj.svg - Wikimedia Commons. https://commons.wikimedia.org/wiki/File:Homoj.svg.
File:Circle-icons-speedometer.svg - Wikimedia Commons. https://commons.wikimedia.org/wiki/File:Circle-icons-speedometer.svg.
File:Dall-e 3 (jan ’24) artificial intelligence icon.png - Wikimedia Commons. https://commons.wikimedia.org/wiki/File:Dall-e_3_(jan_%2724)_artificial_intelligence_icon.png.
File:Silhouette of man with cane.svg - Wikimedia Commons. https://commons.wikimedia.org/wiki/File:Silhouette_of_man_with_cane.svg.
Acknowledgements
The authors gratefully acknowledge the entire Mobilise-D Work Package 2 team for their ongoing collaboration, insightful discussions, and valuable contributions. Special thanks are extended to the study participants for their time, commitment, and enthusiasm, especially during the challenging circumstances of the COVID-19 pandemic. The authors also sincerely thank the contributors to the RUSH Memory and Aging Project, as well as the dedicated staff at the Rush Alzheimer’s Disease Center for their continuous support. This work was supported in part by grants from the NIH (R01AG017917; R01AG056352, R01AG79133, R01AG075728), the Aufzien Family Center for the Prevention and Treatment of Parkinson’s Disease (APPD) at Tel Aviv University, and the Mobilise-D project. The Mobilise-D project received funding from the Innovative Medicines Initiative 2 Joint Undertaking (JU) under grant agreement No. 820820. This JU receives support from the European Union's Horizon 2020 research and innovation program and the European Federation of Pharmaceutical Industries and Associations (EFPIA). S.D.D., A.J.Y., and L.R. were supported by the IDEA-FAST project that has received funding from the Innovative Medicines Initiative 2 Joint Undertaking (JU) under grant agreement No. 853981. S.D.D., A.J.Y., and L.R. were supported by the National Institute for Health Research (NIHR) Newcastle Biomedical Research Centre (BRC) based at The Newcastle upon Tyne Hospital NHS Foundation Trust, Newcastle University, and the Cumbria, Northumberland and Tyne and Wear (CNTW) NHS Foundation Trust. S.D.D., A.J.Y., and L.R. were also supported by the NIHR/Wellcome Trust Clinical Research Facility (CRF) infrastructure at Newcastle upon Tyne Hospitals NHS Foundation Trust. S.D.D. was supported by the UK Research and Innovation (UKRI) Engineering and Physical Sciences Research Council (EPSRC) (Grant Ref: EP/X031012/1 and Grant Ref: EP/X036146/1). All opinions are those of the authors and not the funders. The content in this publication reflects the authors’ view, and neither IMI nor the European Union, EFPIA, NHS, NIHR, or any associated partners are responsible for any use that may be made of the information contained herein.
Author information
Authors and Affiliations
Contributions
Participant recruitment and clinical oversight: W.M., C.B, J.M.H., A.J.Y., and L.R. Algorithm development: Y.E.B., O.P., and J.M.H. Data analysis, statistical analysis: Y.E.B. Figures and tables preparation: Y.E.B. Data interpretation: Y.E.B., J.M.H. and O.P. Drafting of the initial manuscript: Y.E.B., J.M.H., and O.P. Intellectual contribution: Y.E.B., F.K., L.P., C.B., A.C., W.M., B.V., A.J.Y., L.R., S.D.D., A.M., A.S.B., J.M.H., and O.P. All authors have provided critical intellectual input during the revision of the manuscript. All authors have reviewed the manuscript and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
S.D.D. reports consultancy activity with Hoffmann-La Roche Ltd. outside of this study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Brand, Y.E., Buchman, A.S., Kluge, F. et al. Continuous assessment of daily-living gait using self-supervised learning of wrist-worn accelerometer data. npj Digit. Med. (2026). https://doi.org/10.1038/s41746-026-02528-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41746-026-02528-2