Abstract
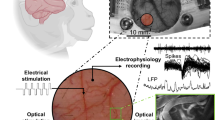
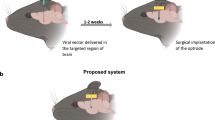
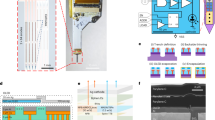
Optical neurotechnologies use light to interface with neurons and can monitor and manipulate neural activity with high spatial-temporal precision over large cortical areas. There has been considerable progress in miniaturizing microscopes for head-mounted configurations, but existing devices are bulky and their application in humans will require a more non-invasive, fully implantable form factor. Here we report an ultrathin, miniaturized subdural complementary metal–oxide–semiconductor (CMOS) optical device for bidirectional optical stimulation and recording. We use a custom CMOS application-specific integrated circuit that is capable of both fluorescence imaging and optogenetic stimulation, creating a probe with a total thickness of less than 200 µm, which is thin enough to lie entirely within the subdural space of the primate brain. We show that the device can be used for imaging and optical stimulation in a mouse model and can be used to decode reach movement speed in a non-human primate.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All imaging data are available at https://github.com/klshepard/scope. All other relevant data are available from the corresponding authors upon reasonable request.
Code availability
Scripts used for image processing, and the decoder model are available at https://github.com/klshepard/scope.
References
Maynard, E. M., Nordhausen, C. T. & Normann, R. A. The Utah Intracortical Electrode Array: a recording structure for potential brain–computer interfaces. Electroencephalogr. Clin. Neurophysiol. 102, 228–239 (1997).
Emiliani, V., Cohen, A. E., Deisseroth, K. & Häusser, M. All-optical interrogation of neural circuits. J. Neurosci. 35, 13917–13926 (2015).
Chen, T.-W. et al. Ultrasensitive fluorescent proteins for imaging neuronal activity. Nature 499, 295–300 (2013).
Jin, L. et al. Single action potentials and subthreshold electrical events imaged in neurons with a fluorescent protein voltage probe. Neuron 75, 779–785 (2012).
Boyden, E. S., Zhang, F., Bamberg, E., Nagel, G. & Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 8, 1263–1268 (2005).
Kleinfeld, D. et al. Can one concurrently record electrical spikes from every neuron in a mammalian brain? Neuron 103, 1005–1015 (2019).
Moreaux, L. C. et al. Integrated neurophotonics: toward dense volumetric interrogation of brain circuit activity—at depth and in real time. Neuron 108, 66–92 (2020).
Ghosh, K. K. et al. Miniaturized integration of a fluorescence microscope. Nat. Methods 8, 871–878 (2011).
Skocek, O. et al. High-speed volumetric imaging of neuronal activity in freely moving rodents. Nat. Methods 15, 429–432 (2018).
Liberti, W. A., Perkins, L. N., Leman, D. P. & Gardner, T. J. An open source, wireless capable miniature microscope system. J. Neural Eng. 14, 045001 (2017).
Rynes, M. L. et al. Miniaturized head-mounted microscope for whole-cortex mesoscale imaging in freely behaving mice. Nat. Methods 18, 417–425 (2021).
Zong, W. et al. Miniature two-photon microscopy for enlarged field-of-view, multi-plane and long-term brain imaging. Nat. Methods 18, 46–49 (2021).
Xue, Y., Davison Ian, G., Boas David, A. & Tian, L. Single-shot 3D wide-field fluorescence imaging with a computational miniature mesoscope. Sci. Adv. 6, eabb7508 (2020).
Yanny, K. et al. Miniscope3D: optimized single-shot miniature 3D fluorescence microscopy. Light. Sci. Appl. 9, 171 (2020).
Adams Jesse, K. et al. Single-frame 3D fluorescence microscopy with ultraminiature lensless FlatScope. Sci. Adv. 3, e1701548 (2017).
Adams, J. K. et al. In vivo lensless microscopy via a phase mask generating diffraction patterns with high-contrast contours. Nat. Biomed. Eng. https://doi.org/10.1038/s41551-022-00851-z (2022).
Choi, J. et al. A 512-pixel, 51-kHz-frame-rate, dual-shank, lens-less, filter-less single-photon avalanche diode CMOS neural imaging probe. IEEE J. Solid-State Circuits 54, 2957–2968 (2019).
Moazeni, S. et al. A mechanically flexible, implantable neural interface for computational imaging and optogenetic stimulation over 5.4×5.4mm2 FOV. IEEE Trans. Biomed. Circuits Syst. https://doi.org/10.1109/TBCAS.2021.3138334 (2021).
Bruschini, C., Homulle, H., Antolovic, I. M., Burri, S. & Charbon, E. Single-photon avalanche diode imagers in biophotonics: review and outlook. Light. Sci. Appl. 8, 87 (2019).
Kawahito, S. Column-parallel ADCs for CMOS image sensors and their FoM-based evaluations. IEICE Trans. Electron. E101-C, 444–456 (2018).
Yokota, T. et al. A conformable imager for biometric authentication and vital sign measurement. Nat. Electron. 3, 113–121 (2020).
Kim, T. H. et al. Long-term optical access to an estimated one million neurons in the live mouse cortex. Cell Rep. 17, 3385–3394 (2016).
Hayakawa, C. K. et al. Perturbation Monte Carlo methods to solve inverse photon migration problems in heterogeneous tissues. Opt. Lett. 26, 1335–1337 (2001).
Johansson, J. Spectroscopic method for determination of the absorption coefficient in brain tissue. J. Biomed. Opt. 15, 057005 (2010).
Wang, L & Wu, H.-I. in Biomedical Optics. 343–346 (John Wiley & Sons, 2009).
Marshel James, H. et al. Cortical layer-specific critical dynamics triggering perception. Science 365, eaaw5202 (2019).
Richard, C., Renaudin, A., Aimez, V. & Charette, P. G. An integrated hybrid interference and absorption filter for fluorescence detection in lab-on-a-chip devices. Lab Chip 9, 1371–1376 (2009).
Boominathan, V., Robinson, J. T., Waller, L. & Veeraraghavan, A. Recent advances in lensless imaging. Optica 9, 1–16 (2022).
Taal, A. J., Lee, C., Choi, J., Hellenkamp, B. & Shepard, K. L. Toward implantable devices for angle-sensitive, lens-less, multifluorescent, single-photon lifetime imaging in the brain using Fabry–Perot and absorptive color filters. Light. Sci. Appl. 11, 24 (2022).
Antipa, N. et al. DiffuserCam: lensless single-exposure 3D imaging. Optica 5, 1–9 (2018).
Boominathan, V., Adams, J. K., Robinson, J. T. & Veeraraghavan, A. PhlatCam: designed phase-mask based thin lensless camera. IEEE Trans. Pattern Anal. Mach. Intell. 42, 1618–1629 (2020).
Uguz, I. & Shepard, K. L. Spatially controlled, bipolar, cortical stimulation with high-capacitance, mechanically flexible subdural surface microelectrode arrays. Sci. Adv. 8, eabq6354 (2022).
Arenkiel, B. R. et al. In vivo light-induced activation of neural circuitry in transgenic mice expressing channelrhodopsin-2. Neuron 54, 205–218 (2007).
Racicot, I. et al. High resolution, wide field optical imaging of macaque visual cortex with a curved detector. J. Neural Eng. 19, 066022 (2022).
Ebina, T. et al. Two-photon imaging of neuronal activity in motor cortex of marmosets during upper-limb movement tasks. Nat. Commun. 9, 1879 (2018).
Mathis, A. et al. DeepLabCut: markerless pose estimation of user-defined body parts with deep learning. Nat. Neurosci. 21, 1281–1289 (2018).
Cisek, P., Crammond, D. J. & Kalaska, J. F. Neural activity in primary motor and dorsal premotor cortex in reaching tasks with the contralateral versus ipsilateral arm. J. Neurophysiol. 89, 922–942 (2003).
Tchoe, Y. et al. Human brain mapping with multithousand-channel PtNRGrids resolves spatiotemporal dynamics. Sci. Transl. Med. 14, eabj1441 (2022).
Chiang, C.-H. et al. Development of a neural interface for high-definition, long-term recording in rodents and nonhuman primates. Sci. Transl. Med. 12, eaay4682 (2020).
Hong, L., Li, H., Yang, H. & Sengupta, K. Fully integrated fluorescence biosensors on-chip employing multi-functional nanoplasmonic optical structures in CMOS. IEEE J. Solid-State Circuits 52, 2388–2406 (2017).
Shemesh, O. A. et al. Precision calcium imaging of dense neural populations via a cell-body-targeted calcium indicator. Neuron 107, 470–486.e411 (2020).
Yu, N. & Capasso, F. Flat optics with designer metasurfaces. Nat. Mater. 13, 139–150 (2014).
Lecoq, J. et al. Removing independent noise in systems neuroscience data using DeepInterpolation. Nat. Methods 18, 1401–1408 (2021).
Simon, C.-J., Sahel, J.-A., Duebel, J., Herlitze, S. & Dalkara, D. Opsins for vision restoration. Biochem. Biophys. Res. Commun. 527, 325–330 (2020).
Qin, H., Lu, J., Jin, W., Chen, X. & Fu, L. Multichannel fiber photometry for mapping axonal terminal activity in a restricted brain region in freely moving mice. Neurophotonics 6, 035011 (2019).
Andersen, R. A., Musallam, S. & Pesaran, B. Selecting the signals for a brain–machine interface. Curr. Opin. Neurobiol. 14, 720–726 (2004).
Silversmith, D. B. et al. Plug-and-play control of a brain–computer interface through neural map stabilization. Nat. Biotechnol. 39, 326–335 (2021).
Yazdan-Shahmorad, A. et al. A large-scale interface for optogenetic stimulation and recording in nonhuman primates. Neuron 89, 927–939 (2016).
Acknowledgements
This work was supported in part by DARPA under contract N66001-17-C-4012 (K.L.S. and V.A.P.), by the National Science Foundation under grant 1706207 (K.L.S.) and by the NIH under grant NS-103518 (B.P.). We gratefully acknowledge TSMC for chip fabrication and their support in the use of experimental SPAD devices.
Author information
Authors and Affiliations
Contributions
E.H.P. and K.L.S. conceptualized the study. E.H.P., S.M., Y.G. and A.B. designed the circuits and performed the die thinning. E.H.P., H.Y. and S.M. designed and performed the optical packaging. V.B., J.T.R. and A.V. wrote the computational imaging algorithm. E.H.P., H.Y., I.U. and V.A.-P. performed the in vivo mouse experiments. E.H.P., H.Y., A.P., A.D. and K.E.W. performed the in vivo NHP experiments and data processing. B.P. and J.S.C. performed the surgical procedures in NHP. V.A.P., K.L.S. and B.P. acquired the funding. E.H.P., B.P. and K.L.S. wrote and edited the paper. K.L.S. provided overall supervision and guidance.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Electronics thanks Ying Fang, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Sections 1–14, Figs. 1–32, Tables 1–6, Videos 1–3 and Refs. 1–76.
Supplementary Video 1 (download MP4 )
In vivo mouse experiment—electrical stimulation and optical recording.
Supplementary Video 2 (download MP4 )
In vivo mouse experiment—optical stimulus and optical recording.
Supplementary Video 3 (download MP4 )
In vivo NHP experiment—behavioural stimulus and optical recording.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pollmann, E.H., Yin, H., Uguz, I. et al. A subdural CMOS optical device for bidirectional neural interfacing. Nat Electron 7, 829–841 (2024). https://doi.org/10.1038/s41928-024-01209-w
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41928-024-01209-w
This article is cited by
-
Chronically implantable μLED arrays for optogenetic cortical surface stimulation in mice
Nature Communications (2026)
-
Multifunctional bioelectronics for brain–body circuits
Nature Reviews Bioengineering (2025)
-
An implantable CMOS deep-brain fluorescence imager with single-neuron resolution
Nature Electronics (2025)
-
A wireless subdural-contained brain–computer interface with 65,536 electrodes and 1,024 channels
Nature Electronics (2025)