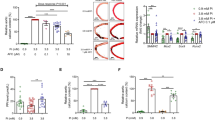
Abstract
The liver plays an important role in cardiovascular disease by amplifying systemic inflammation, while the underlying mechanisms remain to be defined. Soluble epoxide hydrolase (sEH) is a pro-inflammatory enzyme, and pharmacological inhibition of sEH was shown to protect against various inflammatory diseases. In this study, we have identified a novel role of the liver, through expression of sEH, in the pathogenesis of abdominal aortic aneurysm (AAA). sEH expression and activity were markedly higher in mouse liver compared with aorta and further increased in the context of AAA. Pharmacological inhibition or hepatocyte-specific disruption of sEH prevented AAA formation in two animal models of AAA (angiotensin II infusion and aortic calcium chloride application in male mice), concomitant with reduced expression of complement C3 and serum amyloid A, liver-derived inflammatory factors causally linked to AAA formation. Interestingly, data from co-incubation of liver ex vivo with aorta identified galectin-3 secreted from the aneurysm-prone aorta that activates sEH in the liver. We also determined 12,13-dihydroxyoctadecenoic acid (DiHOME) and various circulating pro-inflammatory cytokines as a downstream mechanism potentially associated with hepatic sEH in the context of AAA. These novel findings provide direct evidence that bidirectional crosstalk between aorta and liver contributes to AAA via hepatic sEH.

Similar content being viewed by others
Data availability
Datasets analyzed in this study are available from the corresponding authors upon reasonable request. Specific data underlying each Figure is noted in Figure Legends and Supplementary Data file. The LC-MS/MS proteomics data have been deposited to the ProteomeXchange Consortium via the Proteomics Identification database (PRIDE) partner repository with the dataset identifier PXD073197.
References
Abdulameer, H., Al Taii, H., Al-Kindi, S. G. & Milner, R. Epidemiology of fatal ruptured aortic aneurysms in the United States (1999-2016). J. Vasc. Surg. 69, 378–384 e372 (2019).
Howard, D. P. et al. Population-based study of incidence and outcome of acute aortic dissection and premorbid risk factor control: 10-year results from the Oxford Vascular Study. Circulation 127, 2031–2037 (2013).
Wise, E. S., Hocking, K. M. & Brophy, C. M. Prediction of in-hospital mortality after ruptured abdominal aortic aneurysm repair using an artificial neural network. J. Vasc. Surg. 62, 8–15 (2015).
Mantovani, A. & Garlanda, C. Humoral innate immunity and acute-phase proteins. N. Engl. J. Med. 388, 1725–1726 (2023).
Pagano, M. B. et al. Complement-dependent neutrophil recruitment is critical for the development of elastase-induced abdominal aortic aneurysm. Circulation 119, 1805–1813 (2009).
Hinterseher, I. et al. Role of complement cascade in abdominal aortic aneurysms. Arterioscler Thromb. Vasc. Biol. 31, 1653–1660 (2011).
Zhou, H. F. et al. Fibrinogen-specific antibody induces abdominal aortic aneurysm in mice through complement lectin pathway activation. Proc. Natl. Acad. Sci. USA 110, E4335–E4344 (2013).
Allen-Redpath, K. et al. Phospholipid membranes drive abdominal aortic aneurysm development through stimulating coagulation factor activity. Proc. Natl. Acad. Sci. USA 116, 8038–8047 (2019).
Webb, N. R. et al. Deficiency of endogenous acute-phase serum amyloid A protects apoE-/- mice from angiotensin II-induced abdominal aortic aneurysm formation. Arterioscler Thromb. Vasc. Biol. 35, 1156–1165 (2015).
Nagareddy, P. R., Noothi, S. K., Flynn, M. C. & Murphy, A. J. It’s reticulated: the liver at the heart of atherosclerosis. J. Endocrinol. 238, R1–R11 (2018).
Vaughan, C. J., Gotto, A. M. Jr. & Basson, C. T. The evolving role of statins in the management of atherosclerosis. J. Am. Coll. Cardiol. 35, 1–10 (2000).
Kazi, D. S. et al. Cost-effectiveness of PCSK9 inhibitor therapy in patients with heterozygous familial hypercholesterolemia or atherosclerotic cardiovascular disease. JAMA 316, 743–753 (2016).
Imig, J. D. et al. An orally active epoxide hydrolase inhibitor lowers blood pressure and provides renal protection in salt-sensitive hypertension. Hypertension 46, 975–981 (2005).
Kim, S. A. et al. Soluble epoxide hydrolase inhibitor, TPPU, attenuates progression of atherosclerotic lesions and vascular smooth muscle cell phenotypic switching. Vasc. Pharm. 145, 107086 (2022).
Zhang, L. N. et al. Inhibition of soluble epoxide hydrolase attenuated atherosclerosis, abdominal aortic aneurysm formation, and dyslipidemia. Arterioscler Thromb. Vasc. Biol. 29, 1265–1270 (2009).
Enayetallah, A. E., French, R. A., Thibodeau, M. S. & Grant, D. F. Distribution of soluble epoxide hydrolase and of cytochrome P450 2C8, 2C9, and 2J2 in human tissues. J. Histochem. Cytochem. 52, 447–454 (2004).
Gill, S. S. & Hammock, B. D. Distribution and properties of a mammalian soluble epoxide hydrase. Biochem. Pharm. 29, 389–395 (1980).
Decker, M., Arand, M. & Cronin, A. Mammalian epoxide hydrolases in xenobiotic metabolism and signalling. Arch. Toxicol. 83, 297–318 (2009).
Tanaka, H. et al. Transcriptional regulation of the human soluble epoxide hydrolase gene EPHX2. Biochim. Biophys. Acta 1779, 17–27 (2008).
Vasconez, A. E. et al. The histone demethylase Jarid1b mediates angiotensin II-induced endothelial dysfunction by controlling the 3’UTR of soluble epoxide hydrolase. Acta Physiol. 225, e13168 (2019).
Wang, W. et al. Soluble epoxide hydrolase contributes to cell senescence and ER stress in aging mice colon. Int. J. Mol. Sci. 24, 4570 (2023).
Pinot, F., Grant, D. F., Spearow, J. L., Parker, A. G. & Hammock, B. D. Differential regulation of soluble epoxide hydrolase by clofibrate and sexual hormones in the liver and kidneys of mice. Biochem. Pharm. 50, 501–508 (1995).
Smith, K. R. et al. Attenuation of tobacco smoke-induced lung inflammation by treatment with a soluble epoxide hydrolase inhibitor. Proc. Natl. Acad. Sci. USA 102, 2186–2191 (2005).
Ostermann, A. I. et al. Oral treatment of rodents with soluble epoxide hydrolase inhibitor 1-(1-propanoylpiperidin-4-yl)-3-[4-(trifluoromethoxy)phenyl]urea (TPPU): resulting drug levels and modulation of oxylipin pattern. Prostaglandins Other Lipid Mediat 121, 131–137 (2015).
Sobotta, S. et al. Model based targeting of IL-6-induced inflammatory responses in cultured primary hepatocytes to improve application of the JAK inhibitor ruxolitinib. Front. Physiol. 8, 775 (2017).
Liu, X. & Green, R. M. Endoplasmic reticulum stress and liver diseases. Liver Res. 3, 55–64 (2019).
Chiou, A. C., Chiu, B. & Pearce, W. H. Murine aortic aneurysm produced by periarterial application of calcium chloride. J. Surg. Res. 99, 371–376 (2001).
Wang, Y., Krishna, S. & Golledge, J. The calcium chloride-induced rodent model of abdominal aortic aneurysm. Atherosclerosis 226, 29–39 (2013).
Khambu B., Yan S., Huda N., Yin X. M. Role of high-mobility group box-1 in liver pathogenesis. Int. J. Mol. Sci. 20, 5314 (2019).
Wen, Y., Jeong, S., Xia, Q. & Kong, X. Role of osteopontin in liver diseases. Int J. Biol. Sci. 12, 1121–1128 (2016).
Kram, M. Galectin-3 inhibition as a potential therapeutic target in non-alcoholic steatohepatitis liver fibrosis. World J. Hepatol. 15, 201–207 (2023).
Lu, H. Y. et al. Galectin-3 modulates macrophage activation and contributes smooth muscle cells apoptosis in abdominal aortic aneurysm pathogenesis. Int. J. Mol. Sci. 21, 8257 (2020).
Dai, Y. et al. Enhancement of the liver’s neuroprotective role ameliorates traumatic brain injury pathology. Proc. Natl. Acad. Sci. USA 120, e2301360120 (2023).
Wu, Y. et al. Hepatic soluble epoxide hydrolase activity regulates cerebral Aβ metabolism and the pathogenesis of Alzheimer’s disease in mice. Neuron 111, 2847–2862.e10 (2023).
Sawada, H., Lu, H. S., Cassis, L. A. & Daugherty, A. Twenty years of studying AngII (angiotensin II)-induced abdominal aortic pathologies in mice: continuing questions and challenges to provide insight into the human disease. Arterioscler Thromb. Vasc. Biol. 42, 277–288 (2022).
Lu H. Y. et al. Galectin-3 Modulates Macrophage activation and contributes smooth muscle cells apoptosis in abdominal aortic aneurysm pathogenesis. Int. J. Mol. Sci. 21, 8257 (2020).
Fashanu, O. E. et al. Galectin-3 and the incidence of abdominal aortic aneurysm: the atherosclerosis risk in communities (ARIC) study. Am. J. Cardiovasc Dis. 7, 114–121 (2017).
Kohno, T. et al. High-mobility group box 1 protein blockade suppresses development of abdominal aortic aneurysm. J. Cardiol. 59, 299–306 (2012).
Wang, S. K. et al. Osteopontin may be a driver of abdominal aortic aneurysm formation. J. Vasc. Surg. 68, 22S–29S (2018).
Imig, J. D. & Hammock, B. D. Soluble epoxide hydrolase as a therapeutic target for cardiovascular diseases. Nat. Rev. Drug Discov. 8, 794–805 (2009).
Hildreth, K., Kodani, S. D., Hammock, B. D. & Zhao, L. Cytochrome P450-derived linoleic acid metabolites EpOMEs and DiHOMEs: a review of recent studies. J. Nutr. Biochem. 86, 108484 (2020).
Moxon, J. V. et al. Comparison of the serum lipidome in patients with abdominal aortic aneurysm and peripheral artery disease. Circ. Cardiovasc. Genet. 7, 71–79 (2014).
Bergmann, C. B. et al. sEH-derived metabolites of linoleic acid drive pathologic inflammation while impairing key innate immune cell function in burn injury. Proc. Natl. Acad. Sci. USA 119, e2120691119 (2022).
Moghaddam, M. F. et al. Bioactivation of leukotoxins to their toxic diols by epoxide hydrolase. Nat. Med. 3, 562–566 (1997).
Tang, X. et al. Coronary sinus metabolite 12,13-diHOME is a novel biomarker for left atrial remodeling in patients with atrial fibrillation. Circ. Arrhythm. Electrophysiol. 17, e012486 (2024).
Wutzler, A. et al. Variations in the human soluble epoxide hydrolase gene and recurrence of atrial fibrillation after catheter ablation. Int. J. Cardiol. 168, 3647–3651 (2013).
Chi, L. F., Yi, X. Y., Shao, M. J., Lin, J. & Zhou, Q. Interaction between ALOX5AP and CYP3A5 gene variants significantly increases the risk for cerebral infarctions in Chinese. Neuroreport 25, 452–457 (2014).
Yi, X., Liao, D., Fu, X., Zhang, B. & Wang, C. Interaction among CYP2C8, EPHX2, and CYP4A11 gene variants significantly increases the risk for ischemic stroke in Chinese populations. J. Atheroscler. Thromb. 22, 1148–1157 (2015).
Lee, C. R. et al. Genetic variation in soluble epoxide hydrolase (EPHX2) and risk of coronary heart disease: The Atherosclerosis Risk in Communities (ARIC) study. Hum. Mol. Genet. 15, 1640–1649 (2006).
Golledge, J., Thanigaimani, S., Powell, J. T. & Tsao, P. S. Pathogenesis and management of abdominal aortic aneurysm. Eur. Heart J. 44, 2682–2697 (2023).
Chen, D. et al. Pharmacokinetics and pharmacodynamics of AR9281, an inhibitor of soluble epoxide hydrolase, in single- and multiple-dose studies in healthy human subjects. J. Clin. Pharm. 52, 319–328 (2012).
Lazaar, A. L. et al. Pharmacokinetics, pharmacodynamics and adverse event profile of GSK2256294, a novel soluble epoxide hydrolase inhibitor. Br. J. Clin. Pharm. 81, 971–979 (2016).
Hammock, B. D. et al. Movement to the clinic of soluble epoxide hydrolase inhibitor EC5026 as an analgesic for neuropathic pain and for use as a nonaddictive opioid alternative. J. Med. Chem. 64, 1856–1872 (2021).
Horimatsu, T. et al. Niacin protects against abdominal aortic aneurysm formation via GPR109A independent mechanisms: role of NAD+/nicotinamide. Cardiovasc. Res. 116, 2226–2238 (2020).
Greenway, J. et al. Profiling of histone modifications reveals epigenomic dynamics during abdominal aortic aneurysm formation in mouse models. Front. Cardiovasc. Med. 7, 595011 (2020).
Weintraub, N. L. et al. Role of prostaglandin D2 receptors in the pathogenesis of abdominal aortic aneurysm formation. Clin. Sci. 136, 309–321 (2022).
Acknowledgements
This study was funded by grants AG076235 (NIH), 971459 (AHA), 863622 (AHA), R35 ES030443 (NIH/NIEHS RIVER award), and P42 ES004699 (NIH/NIEHS Superfund Research Program).
Author information
Authors and Affiliations
Contributions
H.W.K. and N.L.W. conceived and designed research, D.S.K., T.H., M.O., B.G., H.S., P.V., R.C., N.C., M.M., R.P., S.C., T.W.B., R.H., G.A., H.G.S., M.S., S.H. W.Z., and P.C. performed experiments, D.S.K., T.H., M.O, B.G., P.V., R.C., N.C., N.L.W., and H.W.K. analyzed data, D.J.F, G.C., Y.H., X.L., R.L., A.G., L.Y., D.C.Z., S.H.H., B.D.H., N.L.W., and H.W.K. interpreted results of experiments, N.L.W. and H.W.K. wrote manuscript, D.J.F., R.L., S.H.H., B.D.H., N.L.W., and H.W.K. edit and revised manuscript, N.L.W. and H.W.K. approved final version of manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks María Galán and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Christopher Hine and Dario Ummarino. [A peer review file is available].
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kim, D.S., Horimatsu, T., Ogbi, M. et al. Soluble epoxide hydrolase in the liver orchestrates abdominal aortic aneurysm formation in mice. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09765-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09765-x