Abstract
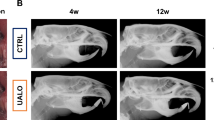
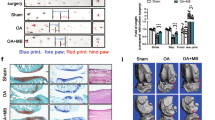
Temporomandibular joint osteoarthritis (TMJOA) is a disease that affects the TMJ and is associated with degenerative changes in the articular cartilage. Fibroblast-like synoviocytes (FLSs) have been found to contribute to osteoarthritis. Here, we aim to investigate the role of METTL3-mediated FLS senescence in the TMJOA process. TMJOA model rats were successfully generated, displaying typical structural and inflammatory alterations, and primary FLSs were isolated from monosodium iodoacetate (MIA)-induced TMJOA rats; these FLSs were accompanied by increased senescence, attenuated mitophagy, and upregulated METTL3. FLSs from TMJOA rats also induced cartilage degradation. Mechanistically, METTL3 silencing can increase PINK1 expression by increasing its RNA stability through m6A modification. In addition, we found that METTL3 silencing could delay cellular senescence and promote mitophagy by upregulating PINK1 in bleomycin (BLM)-induced hFLSs. Senescent FLSs can also accelerate pathological progression and cartilage degradation in Sprague–Dawley (SD) rats. This study revealed that METTL3 silencing could suppress the senescence of FLSs and promote mitophagy by mediating m6A modification to upregulate PINK1 during TMJOA progression, which might provide a theoretical basis for TMJOA therapy.

Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request. The numerical source data underlying graphs in the manuscript can be found in the supplementary data file.
References
Roberts, W. E. & Goodacre, C. J. The Temporomandibular joint: a critical review of life-support functions, development, articular surfaces, biomechanics and degeneration. J. Prosthodont. 29, 772–779 (2020).
Wang, D. et al. Recent advances in animal models, diagnosis, and treatment of temporomandibular joint Osteoarthritis. Tissue Eng. B Rev. 29, 62–77 (2023).
Liu, G. et al. Insights into the Notch signaling pathway in degenerative musculoskeletal disorders: Mechanisms and perspectives. Biomed. Pharmacother. 169, 115884 (2023).
Zhao, X. et al. Symptoms, disc position, occluding pairs, and facial skeletal characteristics of older patients with temporomandibular disorders. J. Int. Med. Res. 49, 300060521990530 (2021).
Zhou, J., Ren, R., Li, Z., Zhu, S. & Jiang, N. Temporomandibular joint osteoarthritis: A review of animal models induced by surgical interventions. Oral. Dis. 29, 2521–2528 (2023).
Nygaard, G. & Firestein, G. S. Restoring synovial homeostasis in rheumatoid arthritis by targeting fibroblast-like synoviocytes. Nat. Rev. Rheumatol. 16, 316–333 (2020).
Tsaltskan, V. & Firestein, G. S. Targeting fibroblast-like synoviocytes in rheumatoid arthritis. Curr. Opin. Pharm. 67, 102304 (2022).
Yang, J. et al. Targeting YAP1-regulated glycolysis in fibroblast-like synoviocytes impairs macrophage infiltration to ameliorate diabetic osteoarthritis progression. Adv. Sci. 11, e2304617 (2024).
Liu, B. et al. Inflammatory fibroblast-like synoviocyte-derived exosomes aggravate osteoarthritis via enhancing macrophage glycolysis. Adv. Sci. 11, e2307338 (2024).
Yang, F. et al. Abnormal mechanical stress induced chondrocyte senescence by YAP loss-mediated METTL3 upregulation. Oral. Dis. 30, 3308–3320 (2024).
Miwa, S., Kashyap, S., Chini, E. & von Zglinicki, T. Mitochondrial dysfunction in cell senescence and aging. J. Clin. Investig. 132 (2022).
Jiang, W. et al. Mechanisms linking mitochondrial mechanotransduction and chondrocyte biology in the pathogenesis of osteoarthritis. Ageing Res. Rev. 67, 101315 (2021).
Bai, Y. et al. RNA methylation, homologous recombination repair and therapeutic resistance. Biomed. Pharmacother. 166, 115409 (2023).
Zhao, X. et al. Alterations of the m(6)A Methylation Induced by TGF-β2 in ARPE-19 Cells. Front Biosci. 28, 148 (2023).
Wei, F., Zhang, J. N., Zhao, Y. Q., Lyu, H. & Chen, F. Expression of m6A RNA Methylation Regulators and Their Clinical Predictive Value in Intrahepatic Cholangiocarcinoma. Front. Biosci. 28, 120 (2023).
Ye, G. et al. ALKBH5 facilitates CYP1B1 mRNA degradation via m6A demethylation to alleviate MSC senescence and osteoarthritis progression. Exp. Mol. Med. 55, 1743–1756 (2023).
Lu, Y. et al. Expression pattern analysis of m6A regulators reveals IGF2BP3 as a key modulator in osteoarthritis synovial macrophages. J. Transl. Med. 21, 339 (2023).
Jiang, X. et al. The role of m6A modification in the biological functions and diseases. Signal Transduct. Target Ther. 6, 74 (2021).
Han, D. et al. The emerging role of fibroblast-like synoviocytes-mediated synovitis in osteoarthritis: An update. J. Cell Mol. Med. 24, 9518–9532 (2020).
Zhou, Y. et al. Senolytics alleviate the degenerative disorders of temporomandibular joint in old age. Aging Cell 20, e13394 (2021).
Saul, D. et al. A new gene set identifies senescent cells and predicts senescence-associated pathways across tissues. Nat. Commun. 13, 4827 (2022).
Coryell, P. R., Diekman, B. O. & Loeser, R. F. Mechanisms and therapeutic implications of cellular senescence in osteoarthritis. Nat. Rev. Rheumatol. 17, 47–57 (2021).
Yagi, M., Endo, K., Komori, K. & Sekiya, I. Comparison of the effects of oxidative and inflammatory stresses on rat chondrocyte senescence. Sci. Rep. 13, 7697 (2023).
Huang, W., Hickson, L. J., Eirin, A., Kirkland, J. L. & Lerman, L. O. Cellular senescence: the good, the bad and the unknown. Nat. Rev. Nephrol. 18, 611–627 (2022).
O’Sullivan, E. A., Wallis, R., Mossa, F. & Bishop, C. L. The paradox of senescent-marker positive cancer cells: challenges and opportunities. NPJ Aging 10, 41 (2024).
Chen, J. et al. DCR2, a cellular senescent molecule, is a novel marker for assessing tubulointerstitial fibrosis in patients with immunoglobulin A nephropathy. Kidney Blood Press Res 44, 1063–1074 (2019).
Li, K. et al. Tyrosine kinase Fyn promotes osteoarthritis by activating the β-catenin pathway. Ann. Rheum. Dis. 77, 935–943 (2018).
Sun, K., Jing, X., Guo, J., Yao, X. & Guo, F. Mitophagy in degenerative joint diseases. Autophagy 17, 2082–2092 (2021).
Bernabei, I., So, A., Busso, N. & Nasi, S. Cartilage calcification in osteoarthritis: mechanisms and clinical relevance. Nat. Rev. Rheumatol. 19, 10–27 (2023).
Wang, Y. et al. Mst1 promotes mitochondrial dysfunction and apoptosis in oxidative stress-induced rheumatoid arthritis synoviocytes. Aging 12, 16211–16223 (2020).
Luo, Y., Zhang, L., Su, N., Liu, L. & Zhao, T. YME1L-mediated mitophagy protects renal tubular cells against cellular senescence under diabetic conditions. Biol. Res. 57, 10 (2024).
Li, J. et al. PINK1/Parkin-mediated mitophagy in neurodegenerative diseases. Ageing Res. Rev. 84, 101817 (2023).
Imberechts, D. et al. DJ-1 is an essential downstream mediator in PINK1/parkin-dependent mitophagy. Brain 145, 4368–4384 (2022).
Yan, C. et al. PHB2 (prohibitin 2) promotes PINK1-PRKN/Parkin-dependent mitophagy by the PARL-PGAM5-PINK1 axis. Autophagy 16, 419–434 (2020).
Lippai, M. & Lőw, P. The role of the selective adaptor p62 and ubiquitin-like proteins in autophagy. Biomed. Res. Int. 2014, 832704 (2014).
Zhang, S. et al. Negative pressure wound therapy improves bone regeneration by promoting osteogenic differentiation via the AMPK-ULK1-autophagy axis. Autophagy 18, 2229–2245 (2022).
Nähse, V. et al. ATPase activity of DFCP1 controls selective autophagy. Nat. Commun. 14, 4051 (2023).
Sirko, A., Wawrzyńska, A., Brzywczy, J. & Sieńko, M. Control of ABA signaling and crosstalk with other hormones by the selective degradation of pathway components. Int. J. Mol. Sci. 22. https://doi.org/10.3390/ijms22094638 (2021).
Luo, S. et al. METTL3-mediated m6A mRNA methylation regulates neutrophil activation through targeting TLR4 signaling. Cell Rep. 42, 112259 (2023).
Kumari, R. et al. mRNA modifications in cardiovascular biology and disease: with a focus on m6A modification. Cardiovasc. Res. 118, 1680–1692 (2022).
Chen, L. et al. METTL3-mediated m6A modification stabilizes TERRA and maintains telomere stability. Nucleic Acids Res. 50, 11619–11634 (2022).
Chen, X. et al. METTL3-mediated m(6)A modification of ATG7 regulates autophagy-GATA4 axis to promote cellular senescence and osteoarthritis progression. Ann. Rheum. Dis. 81, 87–99 (2022).
Chen, B. et al. Metformin suppresses oxidative stress induced by high glucose via activation of the Nrf2/HO-1 signaling pathway in Type 2 diabetic osteoporosis. Life Sci. 312, 121092 (2023).
Jiang, H. et al. Chronic pain causes peripheral and central responses in MIA-induced TMJOA Rats. Cell. Mol. Neurobiol. 42, 1441–1451 (2022).
Feng, S. Y. et al. Increased joint loading induces subchondral bone loss of the temporomandibular joint via the RANTES-CCRs-Akt2 axis. JCI Insight 7. https://doi.org/10.1172/jci.insight.158874 (2022).
Yuan, W. et al. Comparison and applicability of three induction methods of temporomandibular joint osteoarthritis in murine models. J. Oral. Rehabil. 49, 430–441 (2022).
Sukkaew, A. et al. Heterogeneity of clinical isolates of chikungunya virus and its impact on the responses of primary human fibroblast-like synoviocytes. J. Gen. Virol. 99, 525–535 (2018).
Acknowledgements
The present study was supported in part by the National Natural Science Foundation of China (82470953) and the Natural Science Foundation of Sichuan Province (2025ZNSFSC0750).
Author information
Authors and Affiliations
Contributions
Conceptualization: L.X. and Y.M. Data curation: J.L., K.T., and Q.D. Formal analysis: L.X., J.L., and J.G. Funding acquisition: Y.M. and L.X. Investigation: J.L., W.L., X.Y., and S.L. Methodology: L.X. and K.T. Project administration: L. X. and J.L. Visualization: L.X. and K.T. Writing - original draft: L.X. and Y.M. Writing - review & editing: All authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Wacharapol Saengsiwaritt and Ruina Kong for their contribution to the peer review of this work. Primary Handling Editors: Carmen Hueasa and Joao Valente.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tian, K., Du, Q., Guo, J. et al. METTL3-mediated fibroblast-like synoviocytes senescence promotes temporomandibular joint osteoarthritis progression. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09773-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09773-x