Abstract
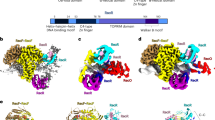
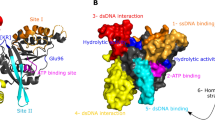
Bacterial RecA protein oligomerizes on single-stranded DNA (ssDNA) in the presence of ATP to form an active filament, RecA*, which is essential for homologous recombination and the activation of bacterial DNA damage response (SOS response). Despite extensive studies on the structure and function of RecA and RecA*, the exact function of the RecA flexible C-terminal tail remains poorly understood. Here, we report the crystal structure of the full-length RecA protein from K. pneumoniae, revealing that the C-terminal tail adopts a β-strand conformation. The negatively charged C-terminal tail is observed to interact with two positively charged conserved residues in the RecA core ATPase domain. We also demonstrate that the C-terminal tail of the E. coli and K. pneumoniae RecA inhibits the formation of RecA filament, DNA binding, and DNA strand exchange during homologous recombination, but promotes LexA self-cleavage as a co-protease and enhances the SOS response induced by mitomycin and ciprofloxacin. Our results provide mechanistic insights into the regulatory function of the RecA C-terminal tail in genome maintenance and DNA damage response.
Similar content being viewed by others
Data availability
The atomic coordinates and structure factors have been deposited in the Research Collaboratory for Structural Bioinformatics Protein Data Bank (RCSB PDB, https://www.rcsb.org/) with the following accession codes: 9UOO. Source data for the graphs are available in the Supplementary Data.
References
Radman, M. SOS repair hypothesis: phenomenology of an inducible DNA repair which is accompanied by mutagenesis. Basic Life Sci. 5a, 355–367 (1975).
Witkin, E. M. RecA protein in the SOS response: milestones and mysteries. Biochimie 73, 133–141 (1991).
Schlacher, K. & Goodman, M. F. Lessons from 50 years of SOS DNA-damage-induced mutagenesis. Nat. Rev. Mol. Cell Biol. 8, 587–594 (2007).
Walker, G. C. Mutagenesis and inducible responses to deoxyribonucleic acid damage in Escherichia coli. Microbiol. Rev. 48, 60–93 (1984).
Fernández De Henestrosa, A. R. et al. Identification of additional genes belonging to the LexA regulon in Escherichia coli. Mol. Microbiol. 35, 1560–1572 (2000).
Recacha, E. et al. Suppression of the SOS response modifies spatiotemporal evolution, post-antibiotic effect, bacterial fitness and biofilm formation in quinolone-resistant Escherichia coli. J. Antimicrob. Chemother. 74, 66–73 (2019).
Maslowska, K. H., Makiela-Dzbenska, K. & Fijalkowska, I. J. The SOS system: a complex and tightly regulated response to DNA damage. Environ. Mol. Mutagen. 60, 368–384 (2019).
Torres, R., Carrasco, B., Ayora, S. & Alonso, J. C. Hallmarks of DNA replication stress responses in Escherichia coli and Bacillus subtilis. FEMS Microbiol. Rev. 49, https://doi.org/10.1093/femsre/fuaf041 (2025).
Del Val, E., Nasser, W., Abaibou, H. & Reverchon, S. RecA and DNA recombination: a review of molecular mechanisms. Biochem. Soc. Trans. 47, 1511–1531 (2019).
Lusetti, S. L. & Cox, M. M. The bacterial RecA protein and the recombinational DNA repair of stalled replication forks. Annu. Rev. Biochem. 71, 71–100 (2002).
Cox, M. M. Motoring along with the bacterial RecA protein. Nat. Rev. Mol. Cell Biol. 8, 127–138 (2007).
Chen, Z., Yang, H. & Pavletich, N. P. Mechanism of homologous recombination from the RecA-ssDNA/dsDNA structures. Nature 453, 489–484 (2008).
Bell, C. E. Structure and mechanism of Escherichia coli RecA ATPase. Mol. Microbiol. 58, 358–366 (2005).
Lee, J. Y., Qi, Z. & Greene, E. C. ATP hydrolysis promotes duplex DNA release by the RecA presynaptic complex. J. Biol. Chem. 291, 22218–22230 (2016).
Lee, J. Y. et al. Sequence imperfections and base triplet recognition by the Rad51/RecA family of recombinases. J. Biol. Chem. 292, 11125–11135 (2017).
Yang, H., Zhou, C., Dhar, A. & Pavletich, N. P. Mechanism of strand exchange from RecA-DNA synaptic and D-loop structures. Nature 586, 801–806 (2020).
Greene, E. C. DNA sequence alignment during homologous recombination. J. Biol. Chem. 291, 11572–11580 (2016).
Little, J. W., Edmiston, S. H., Pacelli, L. Z. & Mount, D. W. Cleavage of the Escherichia coli lexA protein by the recA protease. Proc. Natl. Acad. Sci. USA. 77, 3225–3229 (1980).
Roland, K. L., Smith, M. H., Rupley, J. A. & Little, J. W. In vitro analysis of mutant LexA proteins with an increased rate of specific cleavage. J. Mol. Biol. 228, 395–408 (1992).
Kim, B. & Little, J. W. LexA and lambda Cl repressors as enzymes: specific cleavage in an intermolecular reaction. Cell 73, 1165–1173 (1993).
Luo, Y. et al. Crystal structure of LexA: a conformational switch for regulation of self-cleavage. Cell 106, 585–594 (2001).
Zhang, A. P., Pigli, Y. Z. & Rice, P. A. Structure of the LexA-DNA complex and implications for SOS box measurement. Nature 466, 883–886 (2010).
Nohmi, T., Battista, J. R., Dodson, L. A. & Walker, G. C. RecA-mediated cleavage activates UmuD for mutagenesis: mechanistic relationship between transcriptional derepression and posttranslational activation. Proc. Natl. Acad. Sci. USA. 85, 1816–1820 (1988).
Ferentz, A. E., Walker, G. C. & Wagner, G. Converting a DNA damage checkpoint effector (UmuD2C) into a lesion bypass polymerase (UmuD'2C). Embo J. 20, 4287–4298 (2001).
Fang, J. et al. Conformational dynamics of the Escherichia coli DNA polymerase manager proteins UmuD and UmuD. J. Mol. Biol. 398, 40–53 (2010).
Rusche, J. R., Konigsberg, W. & Howard-Flanders, P. Isolation of altered recA polypeptides and interaction with ATP and DNA. J. Biol. Chem. 260, 949–955 (1985).
Benedict, R. C. & Kowalczykowski, S. C. Increase of the DNA strand assimilation activity of recA protein by removal of the C terminus and structure-function studies of the resulting protein fragment. J. Biol. Chem. 263, 15513–15520 (1988).
Lusetti, S. L. et al. C-terminal deletions of the Escherichia coli RecA protein. Characterization of in vivo and in vitro effects. J. Biol. Chem. 278, 16372–16380 (2003).
Tateishi, S., Horii, T., Ogawa, T. & Ogawa, H. C-terminal truncated Escherichia coli RecA protein RecA5327 has enhanced binding affinities to single- and double-stranded DNAs. J. Mol. Biol. 223, 115–129 (1992).
Yu, X. & Egelman, E. H. Removal of the RecA C-terminus results in a conformational change in the RecA-DNA filament. J. Struct. Biol. 106, 243–254 (1991).
Larminat, F. & Defais, M. Modulation of the SOS response by truncated RecA proteins. Mol. Gen. Genet. 216, 106–112 (1989).
Eggler, A. L., Lusetti, S. L. & Cox, M. M. The C terminus of the Escherichia coli RecA protein modulates the DNA binding competition with single-stranded DNA-binding protein. J. Biol. Chem. 278, 16389–16396 (2003).
Fan, H. F. & Su, S. The regulation mechanism of the C-terminus of RecA proteins during DNA strand-exchange process. Biophys. J. 120, 3166–3179 (2021).
Kim, R. et al. RecA requires two molecules of Mg2+ ions for its optimal strand exchange activity in vitro. Nucleic Acids Res. 46, 2548–2559 (2018).
Kurumizaka, H. et al. A possible role of the C-terminal domain of the RecA protein. A gateway model for double-stranded DNA binding. J. Biol. Chem. 271, 33515–33524 (1996).
VanLoock, M. S. et al. Complexes of RecA with LexA and RecX differentiate between active and inactive RecA nucleoprotein filaments. J. Mol. Biol. 333, 345–354 (2003).
Drees, J. C., Lusetti, S. L. & Cox, M. M. Inhibition of RecA protein by the Escherichia coli RecX protein: modulation by the RecA C terminus and filament functional state. J. Biol. Chem. 279, 52991–52997 (2004).
Cárdenas, P. P. et al. RecX facilitates homologous recombination by modulating RecA activities. PLoS Genet. 8, e1003126 (2012).
Le, S. et al. Bacillus subtilis RecA with DprA-SsbA antagonizes RecX function during natural transformation. Nucleic Acids Res. 45, 8873–8885 (2017).
Schlacher, K. et al. DNA polymerase V and RecA protein, a minimal mutasome. Mol. Cell 17, 561–572 (2005).
Story, R. M., Weber, I. T. & Steitz, T. A. The structure of the E. coli recA protein monomer and polymer. Nature 355, 318–325 (1992).
Xing, X. & Bell, C. E. Crystal structures of Escherichia coli RecA in a compressed helical filament. J. Mol. Biol. 342, 1471–1485 (2004).
Krishna, R. et al. Crystallographic identification of an ordered C-terminal domain and a second nucleotide-binding site in RecA: new insights into allostery. Nucleic Acids Res. 34, 2186–2195 (2006).
Machuca, J. et al. Effect of RecA inactivation on quinolone susceptibility and the evolution of resistance in clinical isolates of Escherichia coli. J. Antimicrob. Chemother. 76, 338–344 (2021).
Recacha, E. et al. Quinolone resistance reversion by targeting the SOS response. mBio 8, https://doi.org/10.1128/mBio.00971-17 (2017).
Barrett, T. C., Mok, W. W. K., Murawski, A. M. & Brynildsen, M. P. Enhanced antibiotic resistance development from fluoroquinolone persisters after a single exposure to antibiotic. Nat. Commun. 10, 1177 (2019).
Dörr, T., Lewis, K. & Vulic, M. SOS response induces persistence to fluoroquinolones in Escherichia coli. Plos Genet. 5, e1000760 (2009). ARTN.
Guerin, E. et al. The SOS response controls integron recombination. Science 324, 1034 (2009).
Aranda, J. et al. Acinetobacter baumannii RecA protein in repair of DNA damage, antimicrobial resistance, general stress response, and virulence. J. Bacteriol. 193, 3740–3747 (2011).
Alam, M. K., Alhhazmi, A., DeCoteau, J. F., Luo, Y. & Geyer, C. R. RecA inhibitors potentiate antibiotic activity and block evolution of antibiotic resistance. Cell Chem. Biol. 23, 381–391 (2016).
Shinohara, T. et al. Loop L1 governs the DNA-binding specificity and order for RecA-catalyzed reactions in homologous recombination and DNA repair. Nucleic Acids Res. 43, 973–986 (2015).
Yu, X. & Egelman, E. H. The LexA repressor binds within the deep helical groove of the activated RecA filament. J. Mol. Biol. 231, 29–40 (1993).
Mustard, J. A. & Little, J. W. Analysis of Escherichia coli RecA interactions with LexA, lambda CI, and UmuD by site-directed mutagenesis of recA. J. Bacteriol. 182, 1659–1670 (2000).
Gao, B. et al. Structural basis for regulation of SOS response in bacteria. Proc. Natl. Acad. Sci. USA. 120, e2217493120 (2023).
Behrmann, M. S. et al. Targeted chromosomal Escherichia coli:dnaB exterior surface residues regulate DNA helicase behavior to maintain genomic stability and organismal fitness. PLoS Genet. 17, e1009886 (2021).
Warren, A. J., Maccubbin, A. E. & Hamilton, J. W. Detection of mitomycin C-DNA adducts in vivo by 32P-postlabeling: time course for formation and removal of adducts and biochemical modulation. Cancer Res. 58, 453–461 (1998).
Shariati, A. et al. The resistance mechanisms of bacteria against ciprofloxacin and new approaches for enhancing the efficacy of this antibiotic. Front. Public Health 10, 1025633 (2022).
Cory, M. B. et al. The LexA-RecA* structure reveals a cryptic lock-and-key mechanism for SOS activation. Nat. Struct. Mol. Biol. 31, 1522–1531 (2024).
Shibata, T. et al. Homology recognition without double-stranded DNA-strand separation in D-loop formation by RecA. Nucleic Acids Res. 52, 2565–2577 (2024).
Lee, C. D. & Wang, T. F. The N-terminal domain of Escherichia coli RecA have multiple functions in promoting homologous recombination. J. Biomed. Sci. 16, 37 (2009).
Cox, J. M., Abbott, S. N., Chitteni-Pattu, S., Inman, R. B. & Cox, M. M. Complementation of one RecA protein point mutation by another. Evidence for trans catalysis of ATP hydrolysis. J. Biol. Chem. 281, 12968–12975 (2006).
Mirshad, J. K. & Kowalczykowski, S. C. Biochemical characterization of a mutant RecA protein altered in DNA-binding loop 1. Biochemistry 42, 5945–5954 (2003).
Hörtnagel, K. et al. Saturation mutagenesis of the E. coli RecA loop L2 homologous DNA pairing region reveals residues essential for recombination and recombinational repair. J. Mol. Biol. 286, 1097–1106 (1999).
Babosan, A. et al. A qnrD-plasmid promotes biofilm formation and class 1 integron gene cassette rearrangements in Escherichia coli. Antibiotics 11, 715 (2022).
Matsui, N. et al. Electrochemical mutagen screening using microbial chip. Biosens. Bioelectron. 21, 1202–1209 (2006).
Chen, S. H., Byrne, R. T., Wood, E. A. & Cox, M. M. Escherichia coli radD(yejH) gene: a novel function involved in radiation resistance and double-strand break repair. Mol. Microbiol. 95, 754–768 (2015).
Brenner, S. L., Zlotnick, A. & Griffith, J. D. RecA protein self-assembly. Multiple discrete aggregation states. J. Mol. Biol. 204, 959–972 (1988).
Ghodke, H. et al. Spatial and temporal organization of RecA in the Escherichia coli DNA-damage response. Elife 8, https://doi.org/10.7554/eLife.42761 (2019).
Story, R. M. & Steitz, T. A. Structure of the recA protein-ADP complex. Nature 355, 374–376 (1992).
Lesterlin, C., Ball, G., Schermelleh, L. & Sherratt, D. J. RecA bundles mediate homology pairing between distant sisters during DNA break repair. Nature 506, 249–253 (2014).
Kabsch, W. XDS. Acta Crystallogr. D. Biol. Crystallogr. 66, 125–132 (2010).
Winn, M. D. et al. Overview of the CCP4 suite and current developments. Acta Crystallogr. D Biol. Crystallogr. 67, 235–242 (2011).
Evans, P. R. & Murshudov, G. N. How good are my data and what is the resolution? Acta Crystallogr. D Biol. Crystallogr. 69, 1204–1214 (2013).
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
Liebschner, D. et al. Macromolecular structure determination using X-rays, neutrons and electrons: recent developments in Phenix. Acta Crystallogr. D Struct. Biol. 75, 861–877 (2019).
Acknowledgements
We thank the beamline staff of BL02U1 at Shanghai Synchrotron Radiation Facility for their help with data collection. We thank the staff members of the Large-scale Protein Preparation System at the National Facility for Protein Science in Shanghai (NFPS), Shanghai Advanced Research Institute, Chinese Academy of Sciences, China, for providing technical support and assistance in data collection and analysis. We thank Yuchen Zhang in the Center of Cryo-Electron Microscopy (CCEM), Zhejiang University for her technical assistance on Transmission Electron Microscopy. Figure 1B (left), 2A, 3A, 4A, 6A, and 7B were created with biorender.com(https://BioRender.com/4neox63, https://BioRender.com/02ng0vb, https://BioRender.com/214bz8b, https://BioRender.com/6p874qp, https://BioRender.com/9pcplor, https://BioRender.com/goa6tob), with licenses. This work was supported by the National Natural Science Foundation of China (32371250 to C.Z.) and the Key Project of Natural Science Foundation of Zhejiang Province (LZ24C050002 to C.Z.).
Author information
Authors and Affiliations
Contributions
C.Z. and L.S. designed research; L.S., X.L., F.W., M.W., Y.L., Y.Z., A.G., L.C., Y.G., M.X., and C.Z. performed experiments; L.S., N.D., Y.F., and C.Z. analyzed data; L.S. and C.Z. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Rubén Torres, Ben Bax and the other anonymous reviewer(s) for their contribution to the peer review of this work. Primary handling editors: Joanna Timmins and Laura Rodríguez.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Su, L., Li, X., Wang, F. et al. Mechanistic insights into the structure and function of the RecA C-terminal tail. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09788-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09788-4