Abstract
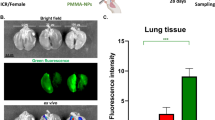
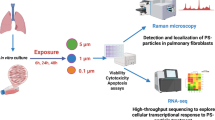
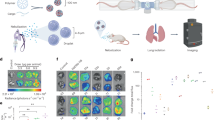
Micro- and nanoplastics (MNPs) are emerging pollutants that can carry harmful substances like benzo(a)pyrene, posing potential health risks. While the harmful effects of nanoplastics on the lungs are known, how they interact with benzo(a)pyrene to affect cellular communication remains unclear. In our study, We explore this interplay using a 16-week mouse model exposed to environmentally relevant doses of polystyrene nanoplastics, benzo(a)pyrene, or a combination of both. We find that only the combined exposure leads to significant lung damage, characterized by severe inflammation and tissue scarring, which are not seen with single exposures. This combined exposure also increases oxidative stress and reduces antioxidant defenses in the lungs. Furthermore, we notice increased levels of inflammation-related molecules and markers of lung tissue damage, confirming a more severe toxic effect. Transcriptomic analysis highlights the involvement of the Relaxin signaling pathway, which influences inflammatory and tissue damage processes through PI3K-AKT and MAPK cascades; Relaxin4 activated PLC-IP3R, opening ER calcium channels and raising cytosolic Ca²⁺, which triggered macrophage extracellular trap (MET) formation. Additionally, a macrophage-MLE-12 co-culture system confirmed that Mix-induced METs are linked to the exacerbation of alveolar inflammation and the progression of pulmonary fibrosis. Our findings reveal novel molecular connections that explain how these pollutants worsen lung health, suggesting that targeting the identified signaling pathways could offer a potential approach to mitigating these harmful effects.

Similar content being viewed by others
Data availability
All data supporting the findings are included in the paper and its supplementary files. All raw data are provided in the supplementary information. Sequencing reads are deposited in NCBI SRA under accession PRJNA1401317. Additional data are available from the corresponding author upon reasonable request.
References
Roland, G., Jenna, R. J. & Kara Lavender, L. Production, use, and fate of all plastics ever made. Sci. Adv. 3, e1700782 (2017).
Gigault, J. et al. Current opinion: What is a nanoplastic? Environ. Pollut. 235, 1030–1034 (2018).
Richard, C. T. et al. Lost at Sea: Where Is All the Plastic? Science 304, 838 (2004).
Saha, S. C. & Saha, G. Effect of microplastics deposition on human lung airways: A review with computational benefits and challenges. Heliyon 10, e24355 (2024).
Merga, L. B. et al. Distribution of microplastic and small macroplastic particles across four fish species and sediment in an African lake. Sci. Total Environ. 741, 140527 (2020).
González-Soto, N. et al. Impacts of dietary exposure to different sized polystyrene microplastics alone and with sorbed benzo[a]pyrene on biomarkers and whole organism responses in mussels Mytilus galloprovincialis. Sci. Total Environ. 684, 548–566 (2019).
Gao, W. et al. From water to sediment: A meta-analysis of microplastic distribution and the impact of dams in reservoir ecosystems. Eco-Environ. Health 4, 1000188 (2025).
Feng, M. et al. Polystyrene nanoplastic exposure induces developmental toxicity by activating the oxidative stress response and base excision repair pathway in Zebrafish (Danio rerio). ACS Omega 7, 32153–32163 (2022).
Waring, R. H., HarriS, R. M. & Mitchell, S. C. Plastic contamination of the food chain: A threat to human health? Maturitas 115, 64–68 (2018).
Enyoh, C. E. et al. Airborne microplastics: a review study on method for analysis, occurrence, movement and risks. Environ. Monit. Assess. 191, 668 (2019).
Joana Correia, P. et al. Environmental exposure to microplastics: An overview on possible human health effects. Sci. Total Environ. 702, 134455 (2020).
Zekang, L. et al. Polystyrene microplastics cause cardiac fibrosis by activating Wnt/β-catenin signaling pathway and promoting cardiomyocyte apoptosis in rats. Environ. Pollut. 265, 115025 (2020).
Xiaoqi, Z. et al. Micro- and nanoplastics: A new cardiovascular risk factor? Environ. Int. 171, 107662 (2023).
Feng, Y. et al. A systematic review of the impacts of exposure to micro- and nano-plastics on human tissue accumulation and health. Eco-Environ. Health 2, 195–207 (2023).
JI, Y. et al. Mucin corona delays intracellular trafficking and alleviates cytotoxicity of nanoplastic-benzopyrene combined contaminant. J. Hazard. Mater. 406, 124306 (2021).
Da Silva Brito, W. A. et al. Sonicated polyethylene terephthalate nano- and micro-plastic-induced inflammation, oxidative stress, and autophagy in vitro. Chemosphere 355, 141813 (2024).
Kumar, R. et al. Micro(nano)plastics pollution and human health: How plastics can induce carcinogenesis to humans? Chemosphere 298, 134267 (2022).
Camacho, M. et al. Organic pollutants in marine plastic debris from Canary Islands beaches. Sci. Total Environ. 662, 22–31 (2019).
Wang, F. et al. Interaction of toxic chemicals with microplastics: A critical review. Water Res. 139, 208–219 (2018).
Barnes, D. K. A. et al. Accumulation and fragmentation of plastic debris in global environments. Philos. Trans. R. Soc. B 364, 1985–1998 (2009).
Liu, J. et al. Polystyrene nanoplastics-enhanced contaminant transport: role of irreversible adsorption in glassy polymeric domain. Environ. Sci. Ecotechnol. 52, 2677–2685 (2018).
Thompson, R. C. et al. Twenty years of microplastic pollution research—what have we learned? Science 386, eadl2746 (2024).
Pittura, L. et al. Microplastics as Vehicles of Environmental PAHs to marine organisms: combined chemical and physical hazards to the Mediterranean Mussels, Mytilus galloprovincialis. Front. Mar. Sci. 5, 15 (2018).
Sun, N. et al. Compound effect and mechanism of oxidative damage induced by nanoplastics and benzo [a] pyrene. J. Hazard. Mater. 460, 132513 (2023).
Zhang, S. Y. et al. Metabolomics analysis reveals that benzo[a]pyrene, a component of PM(2.5), promotes pulmonary injury by modifying lipid metabolism in a phospholipase A2-dependent manner in vivo and in vitro. Redox Biol. 13, 459–469 (2017).
Dris, R. et al. A first overview of textile fibers, including microplastics, in indoor and outdoor environments. Environ. Pollut. 221, 453–458 (2017).
Zhi, L. et al. Immunotoxicity of microplastics: Carrying pathogens and destroying the immune system. TrAC Trends Anal. Chem. 177, 117817 (2024).
Wu, Q. et al. Polystyrene nanoplastics-induced lung apoptosis and ferroptosis via ROS-dependent endoplasmic reticulum stress. Sci. Total Environ. 912, 169260 (2024).
Jin, W. et al. Microplastics exposure causes the senescence of human lung epithelial cells and mouse lungs by inducing ROS signaling. Environ. Int. 185, 108489 (2024).
Lin, Y. et al. Transcriptomics integrated with metabolomics reveals the effect of benzo[a]pyrene exposure on acute lung injury. Ecotoxicol. Environ. Saf. 288, 117323 (2024).
Thannickal, V. J. et al. Mechanisms of pulmonary fibrosis. Annu. Rev. Med. 55, 395–417 (2004).
Soumyakrishnan, S. et al. Role of immune cells in the initiation and progression of pulmonary fibrosis. Crit. Rev. Immunol. 42, 21–41 (2022).
Zhao, L. et al. Prolonged oral ingestion of microplastics induced inflammation in the liver tissues of C57BL/6J mice through polarization of macrophages and increased infiltration of natural killer cells. Ecotoxicol. Environ. Saf. 227, 112882 (2021).
Yang, Q. et al. Oral feeding of nanoplastics affects brain function of mice by inducing macrophage IL-1 signal in the intestine. Cell Rep. 42, 112346 (2023).
Tarasco, M. et al. New insights into benzo[α]pyrene osteotoxicity in zebrafish. Ecotoxicol. Environ. Saf. 226, 112838 (2021).
Jang, H. J. et al. Tobacco-induced hyperglycemia promotes lung cancer progression via cancer cell-macrophage interaction through paracrine IGF2/IR/NPM1-driven PD-L1 expression. Nat. Commun. 15, 4909 (2024).
Sadeghi, M. et al. Neutrophil extracellular trap: A key player in the pathogenesis of autoimmune diseases. Int. Immunopharmacol. 116, 109843 (2023).
Yin, K. et al. Polystyrene microplastics promote liver inflammation by inducing the formation of macrophages extracellular traps. J. Hazard. Mater. 452, 131236 (2023).
Wang, S. et al. Polystyrene microplastics-induced macrophage extracellular traps contributes to liver fibrotic injury by activating ROS/TGF-β/Smad2/3 signaling axis. Environ. Pollut. 324, 121388 (2023).
Zhang, Y. et al. Activation of gut metabolite ACSL4/LPCAT3 by microplastics in drinking water mediates ferroptosis via gut-kidney axis. Commun. Biol. 8, 211 (2025).
Yang, D. et al. Intestinal mucin induces more endocytosis but less transcytosis of nanoparticles across enterocytes by triggering nanoclustering and strengthening the retrograde pathway. ACS Appl. Mater. Interfaces 10, 11443–11456 (2018).
Ren, H. et al. Synergy between nanoplastics and benzo[a]pyrene promotes senescence by aggravating ferroptosis and impairing mitochondria integrity in Caenorhabditis elegans. Sci. Total Environ. 946, 174418 (2024).
Samuel, C. S. & Bennett, R. G. Relaxin as an anti-fibrotic treatment: Perspectives, challenges and future directions. Biochem. Pharmacol. 197, 114884 (2022).
Yang, M., Liu, Q. & Xuan, A. Relaxin-2 Alleviates Hyperoxia-Induced Acute Lung Injury in Neonatal Rats by Inhibiting TLR4/NF-κB. Chem. Biol. Drug Des. 105, e70140 (2025).
Huang, X. et al. Relaxin regulates myofibroblast contractility and protects against lung fibrosis. Am. J. Pathol. 179, 2751–2765 (2011).
Halls, M. L. Constitutive formation of an RXFP1-signalosome: a novel paradigm in GPCR function and regulation. Br. J. Pharm. 165, 1644–1658 (2012).
Kern, A. & Bryant-Greenwood, G. D. Characterization of relaxin receptor (RXFP1) desensitization and internalization in primary human decidual cells and RXFP1-transfected HEK293 cells. Endocrinology 150, 2419–2428 (2009).
Lin, D. et al. FOXO3a induces myocardial fibrosis by upregulating mitophagy. Front. Biosci. (Landmark Ed.) 29, 56 (2024).
Bennett, R. G. Relaxin and its role in the development and treatment of fibrosis. Transl. Res. 154, 1–6 (2009).
Yuan, Y. et al. Relaxin alleviates TGFβ1-induced cardiac fibrosis via inhibition of Stat3-dependent autophagy. Biochem. Biophys. Res. Commun. 493, 1601–1607 (2017).
Fang, C. et al. Fibroblast PI3K/AKT signaling and extracellular matrix homeostasis: mechanisms, targets, and delivery challenges. Front. Cell Dev Biol. 13, 1681875 (2025).
Ahmad, N. et al. Relaxin induces matrix-metalloproteinases-9 and -13 via RXFP1: induction of MMP-9 involves the PI3K, ERK, Akt and PKC-ζ pathways. Mol. Cell. Endocrinol. 363, 46–61 (2012).
Lam et al. Serelaxin as a novel therapeutic opposing fibrosis and contraction in lung diseases. Pharmacol. Ther. 187, 61–70 (2018).
Singh, S., Simpson, R. L. & Bennett, R. G. Relaxin activates peroxisome proliferator-activated receptor γ (PPARγ) through a pathway involving PPARγ coactivator 1α (PGC1α). J. Biol. Chem. 290, 950–959 (2015).
Qi, F. et al. LncRNA TUG1 promotes pulmonary fibrosis progression via up-regulating CDC27 and activating PI3K/Akt/mTOR pathway. Epigenetics 18, 2195305 (2023).
Hu, X. et al. PI3K-Akt-mTOR/PFKFB3 pathway mediated lung fibroblast aerobic glycolysis and collagen synthesis in lipopolysaccharide-induced pulmonary fibrosis. Lab. Investig. 100, 801–811 (2020).
Brinkmann, V. et al. Neutrophil extracellular traps kill bacteria. Science 303, 1532–1535 (2004).
Arias, A. H. et al. Beached microplastics at the Bahia Blanca Estuary (Argentina): Plastic pellets as potential vectors of environmental pollution by POPs. Mar. Pollut. Bull. 187, 114520 (2023).
Schmidt, A. et al. Antioxidant defense in primary murine lung cells following short- and long-term exposure to plastic particles. Antioxidants 12, 227 (2023).
Arora, S. et al. Macrophages: Their role, activation and polarization in pulmonary diseases. Immunobiology 223, 383–396 (2018).
Wang, X. et al. Cytotoxicity and pro-inflammatory effect of polystyrene nano-plastic and micro-plastic on RAW264.7 cells. Toxicology 484, 153391 (2023).
Rasmussen, K. H. & Hawkins, C. A.-O. Role of macrophage extracellular traps in innate immunity and inflammatory disease. Biochem. Soc. Trans. 50, 21–32 (2022).
Zhang, J. et al. Polystyrene microplastics induce pulmonary fibrosis by promoting alveolar epithelial cell ferroptosis through cGAS/STING signaling. Ecotoxicol. Environ. Saf. 277, 116357 (2024).
Brinkmann, V. & Zychlinsky, A. Entering the neutrophil trap. Nat. Rev. Immunol. 21, 615 (2021).
Kummarapurugu, A. B. et al. Neutrophil Elastase triggers the release of macrophage extracellular traps: relevance to cystic fibrosis. Am. J. Respir. Cell Mol. Biol. 66, 76–85 (2022).
Vaarmann, A., Gandhi, S. & Abramov, A. Y. Dopamine induces Ca2+ signaling in astrocytes through reactive oxygen species generated by monoamine oxidase. J. Biol. Chem. 285, 25018–25023 (2010).
Wu, S. et al. Macrophage extracellular traps aggravate iron overload-related liver ischaemia/reperfusion injury. Br. J. Pharmacol. 178, 3783–3796 (2021).
Yang, J. et al. A novel co-culture model for investigation of the effects of LPS-induced macrophage-derived cytokines on brain endothelial cells. PloS One 18, e0288497 (2023).
Dong, C. D. et al. Polystyrene microplastic particles: In vitro pulmonary toxicity assessment. J. Hazard. Mater. 385, 121575 (2020).
Woo, J. H. et al. Polypropylene nanoplastic exposure leads to lung inflammation through p38-mediated NF-κB pathway due to mitochondrial damage. Part Fibre Toxicol. 20, 2 (2023).
Fan, J. et al. Acute exposure to polystyrene nanoparticles promotes liver injury by inducing mitochondrial ROS-dependent necroptosis and augmenting macrophage-hepatocyte crosstalk. Part Fibre Toxicol. 21, 20 (2024).
Hu, M. & Palić, D. Micro- and nano-plastics activation of oxidative and inflammatory adverse outcome pathways. Redox Biol. 37, 101620 (2020).
Goda, C. et al. Loss of FOXM1 in macrophages promotes pulmonary fibrosis by activating p38 MAPK signaling pathway. PLoS Genet. 16, e1008692 (2020).
Fortier, S. M. et al. MAPK phosphatase 1 inhibition of p38α within lung myofibroblasts is essential for spontaneous fibrosis resolution. J. Clin. Investig. 134, e172826 (2024).
Chen, J. et al. Surface functionalization-dependent inflammatory potential of polystyrene nanoplastics through the activation of MAPK/ NF-κB signaling pathways in macrophage Raw 264.7. Ecotoxicol. Environ. Saf. 251, 114520 (2023).
Huang, G. et al. Overexpression of STX11 alleviates pulmonary fibrosis by inhibiting fibroblast activation via the PI3K/AKT/mTOR pathway. Signal Transduct. Target. Ther. 9, 306 (2024).
Shen, S. et al. The exoprotein Gbp of Fusobacterium nucleatum promotes THP-1 cell lipid deposition by binding to CypA and activating PI3K-AKT/MAPK/NF-κB pathways. J. Adv. Res. 57, 93–105 (2024).
Kanai, A. J. et al. Relaxin and fibrosis: Emerging targets, challenges, and future directions. Mol. Cell. Endocrinol. 487, 66–74 (2019).
Wu, J. et al. The interaction between STING and NCOA4 exacerbates lethal sepsis by orchestrating ferroptosis and inflammatory responses in macrophages. Cell Death Dis. 13, 653 (2022).
Dmello, C. et al. Checkpoint kinase 1/2 inhibition potentiates anti-tumoral immune response and sensitizes gliomas to immune checkpoint blockade. Nat. Commun. 14, 1566 (2023).
Hegedűs, C. et al. Cyclobutane pyrimidine dimers from UVB exposure induce a hypermetabolic state in keratinocytes via mitochondrial oxidative stress. Redox Biol. 38, 101808 (2021).
Lou, X. et al. An oral bacterial pyroptosis amplifier against malignant colon cancer. Nano Today, 54, 102091 (2024).
Winslow, S. et al. Multi-omics links IL-6 trans-signalling with neutrophil extracellular trap formation and Haemophilus infection in COPD. Eur. Respir. J. 58, 2003312 (2021).
Chen, L. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget 9, 7204–7218 (2018).
Zhang, X. et al. The distribution and structural fingerprints of metals from particulate matters (PM) deposited in human lungs. Ecotoxicol. Environ. Saf. 233, 113324 (2022).
Liu, S. et al. Assessing inhalation exposure to infectious particles in hospitals using a breathing thermal manikin. Chin. Sci. Bull. 69, 1234–1244 (2023).
Wang, Z. et al. SARS-CoV-2-specific CD4+ T cells are associated with long-term persistence of neutralizing antibodies. Signal Transd. Target. Ther. 7, 132 (2022).
Acknowledgements
This work was supported by the Major Science and Technology plan of Hainan Province (ZDYF2022SHFZ321), National Natural Science Foundation of China (42377273), Research start-up Project of Hainan University (XJ2300009832), Hainan Provincial Graduate Innovation Research Project (SA2400003272).Hainan Province Science and Technology Special Fund (ZDYF2022SHFZ321). We would like to express our gratitude to the Biorender website (https://app.biorender.com/) for providing resources for creating illustrations.
Author information
Authors and Affiliations
Contributions
Yu Chen: Conceptualization, Writing-Original Draft, Formal analysis, Data Curation, Methodology, Data Curation, Yingai Zhang: Project administration, Resources. Yuting Zhang: Methodology. Xu Zhang: Data Curation. Shuguo Lv: Revision, Resource. Mohamed Mohsen: Methodology, Writing - Review & Editing. Kai Yin : Supervision, Methodology. Xiaoshan Zhu: Resource. Hailong Zhou: Supervision, Funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Naresh Singh and the other anonymous reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Dario Ummarino.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, Y., Zhang, Y., Zhang, Y. et al. Polystyrene nanoplastics and benzo(a)pyrene synergistically induce lung fibrosis and inflammation via relaxin signalling in mice. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09872-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09872-9