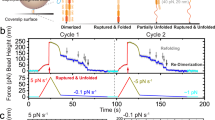
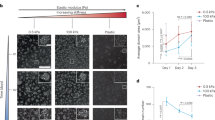
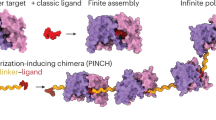
Abstract
Kindlin proteins are central regulators of integrin-mediated cell adhesion, a process essential for various biological and pathological processes. Although structural studies have proposed that kindlins promote integrin activation and clustering via domain-swapped homodimers, this hypothesis has been challenged by the extremely slow in vitro dimerization kinetics, leaving the physiological regulatory mechanism unresolved. Here we show, via multiscale molecular simulations, that mechanical stress acts as a critical trigger for kindlin activation and homodimerization via mechanical allostery. Force accelerates the rate-limiting closed-to-open conformational transition step involved in dimerization, thereby facilitating homodimer assembly. Our simulations pinpoint specific interactions that underlie the high free-energy barrier of conformational transition, which is supported by analytical gel-filtration experiments. We further demonstrate that the relative lengths of linkers between kindlin subdomains strongly influence the propagation of mechanical allostery. Together, these results clarify how mechanical allostery enables kindlin-mediated integrin mechanoactivation and suggest potential therapeutic strategies for adhesion-related disorders.
Similar content being viewed by others
Data availability
The raw data that support this study are available from the corresponding author upon request. The inputfiles for MD simulation and some related files were deposited at https://doi.org/10.6084/m9.figshare.28093673.v2.
Code availability
This study used CafeMol (http://www.cafemol.org/), a non-commercial software for coarse-grained molecular simulations. The source code was minimally modified to apply opposite pair-wise forces in steered simulations. Per the software’s license, neither the original nor modified code may be redistributed. The modified code is available from the corresponding author upon request for verification purposes only.
References
Geiger, B., Bershadsky, A., Pankov, R. & Yamada, K. M. Transmembrane crosstalk between the extracellular matrix and the cytoskeleton. Nat. Rev. Mol. Cell Biol. 2, 793–805 (2001).
Berrier, A. L. & Yamada, K. M. Cell–matrix adhesion. J. Cell Physiol. 213, 565–573 (2007).
Winograd-Katz, S. E., Fässler, R., Geiger, B. & Legate, K. R. The integrin adhesome: from genes and proteins to human disease. Nat. Rev. Mol. Cell Biol. 15, 273–288 (2014).
Hamidi, H. & Ivaska, J. Every step of the way: integrins in cancer progression and metastasis. Nat. Rev. Cancer 18, 533–548 (2018).
Hynes, R. O. Integrins: bidirectional, allosteric signaling machines. Cell 110, 673–687 (2002).
Luo, B.-H., Carman, C. V. & Springer, T. A. Structural basis of integrin regulation and signaling. Annu. Rev. Immunol. 25, 619–647 (2007).
Xiong, J.-P., Stehle, T., Goodman, S. L. & Arnaout, M. A. New insights into the structural basis of integrin activation. Blood 102, 1155–1159 (2003).
Chen, Y. et al. An integrin αiibβ3 intermediate affinity state mediates biomechanical platelet aggregation. Nat. Mater. 18, 760–769 (2019).
Ye, F., Kim, C. & Ginsberg, M. H. Reconstruction of integrin activation. Blood 119, 26–33 (2012).
Su, Y. et al. Relating conformation to function in integrin α5β1. Proc. Natl. Acad. Sci. USA 113, E3872–E3881 (2016).
Humphries, J. D., Chastney, M. R., Askari, J. A. & Humphries, M. J. Signal transduction via integrin adhesion complexes. Curr. Opin. Cell Biol. 56, 14–21 (2019).
Kim, M., Carman, C. V. & Springer, T. A. Bidirectional transmembrane signaling by cytoplasmic domain separation in integrins. Science 301, 1720–1725 (2003).
Kanchanawong, P. et al. Nanoscale architecture of integrin-based cell adhesions. Nature 468, 580–584 (2010).
Elosegui-Artola, A., Trepat, X. & Roca-Cusachs, P. Control of mechanotransduction by molecular clutch dynamics. Trends Cell Biol. 28, 356–367 (2018).
Kechagia, J. Z., Ivaska, J. & Roca-Cusachs, P. Integrins as biomechanical sensors of the microenvironment. Nat. Rev. Mol. Cell Biol. 20, 457–473 (2019).
Tadokoro, S. et al. Talin binding to integrin ß tails: a final common step in integrin activation. Science 302, 103–106 (2003).
Tu, Y., Wu, S., Shi, X., Chen, K. & Wu, C. Migfilin and mig-2 link focal adhesions to filamin and the actin cytoskeleton and function in cell shape modulation. Cell 113, 37–47 (2003).
Calderwood, D. A., Campbell, I. D. & Critchley, D. R. Talins and kindlins: partners in integrin-mediated adhesion. Nat. Rev. Mol. Cell Biol. 14, 503–517 (2013).
Yan, J., Yao, M., Goult, B. T. & Sheetz, M. P. Talin dependent mechanosensitivity of cell focal adhesions. Cell. Mol. Bioeng. 8, 151–159 (2015).
Sun, Z., Costell, M. & Fässler, R. Integrin activation by talin, kindlin and mechanical forces. Nat. Cell Biol. 21, 25–31 (2019).
Zhu, L., Plow, E. F. & Qin, J. Initiation of focal adhesion assembly by talin and kindlin: a dynamic view. Protein Sci. 30, 531–542 (2021).
Mehrbod, M., Trisno, S. & Mofrad, M. R. On the activation of integrin αiibβ3: outside-in and inside-out pathways. Biophys. J. 105, 1304–1315 (2013).
Provasi, D., Negri, A., Coller, B. S. & Filizola, M. Talin-driven inside-out activation mechanism of platelet αiibβ3 integrin probed by multimicrosecond, all-atom molecular dynamics simulations. Proteins 82, 3231–3240 (2014).
Kammerer, P., Aretz, J. & Fässler, R. Lucky kindlin: a cloverleaf at the integrin tail. Proc. Natl. Acad. Sci. USA 114, 9234–9236 (2017).
Bu, W., Levitskaya, Z., Tan, S.-M. & Gao, Y.-G. Emerging evidence for kindlin oligomerization and its role in regulating kindlin function. J. Cell Sci. 134, jcs256115 (2021).
Meves, A., Stremmel, C., Gottschalk, K. & Fässler, R. The kindlin protein family: new members to the club of focal adhesion proteins. Trends Cell Biol. 19, 504–513 (2009).
Ma, Y.-Q., Qin, J., Wu, C. & Plow, E. F. Kindlin-2 (mig-2): a co-activator of β3 integrins. J. Cell Biol. 181, 439–446 (2008).
Rognoni, E., Ruppert, R. & Fässler, R. The kindlin family: functions, signaling properties and implications for human disease. J. Cell Sci. 129, 17–27 (2016).
Montanez, E. et al. Kindlin-2 controls bidirectional signaling of integrins. Genes Dev. 22, 1325–1330 (2008).
Bledzka, K. et al. Spatial coordination of kindlin-2 with talin head domain in interaction with integrin β cytoplasmic tails. J. Biol. Chem. 287, 24585–24594 (2012).
Lu, F. et al. Mechanism of integrin activation by talin and its cooperation with kindlin. Nat. Commun. 13, 2362 (2022).
Haydari, Z., Shams, H., Jahed, Z. & Mofrad, M. R. Kindlin assists talin to promote integrin activation. Biophys. J. 118, 1977–1991 (2020).
Larjava, H., Plow, E. F. & Wu, C. Kindlins: essential regulators of integrin signalling and cell–matrix adhesion. EMBO Rep. 9, 1203–1208 (2008).
Mory, A. et al. Kindlin-3: a new gene involved in the pathogenesis of LAD-III. Blood 112, 2591–2591 (2008).
Ussar, S. et al. Loss of kindlin-1 causes skin atrophy and lethal neonatal intestinal epithelial dysfunction. PLoS Genet. 4, e1000289 (2008).
Malinin, N. L. et al. A point mutation in kindlin3 ablates activation of three integrin subfamilies in humans. Nat. Med. 15, 313–318 (2009).
Lai, Y. et al. Kindlin-2 loss in condylar chondrocytes causes spontaneous osteoarthritic lesions in the temporomandibular joint in mice. Int. J. Oral. Sci. 14, 33 (2022).
Li, H. et al. Structural basis of kindlin-mediated integrin recognition and activation. Proc. Natl. Acad. Sci. USA 114, 9349–9354 (2017).
Bu, W. et al. Structural basis of human full-length kindlin-3 homotrimer in an auto-inhibited state. PLoS Biol. 18, e3000755 (2020).
Sun, J. et al. Structure basis of the ferm domain of kindlin-3 in supporting integrin αiibβ3 activation in platelets. Blood Adv. 4, 3128–3135 (2020).
Qadota, H., Moerman, D. G. & Benian, G. M. A molecular mechanism for the requirement of pat-4 (integrin-linked kinase (ilk)) for the localization of unc-112 (kindlin) to integrin adhesion sites. J. Biol. Chem. 287, 28537–28551 (2012).
Ye, F. et al. The mechanism of kindlin-mediated activation of integrin αiibβ3. Curr. Biol. 23, 2288–2295 (2013).
Yates, L. A., Füzéry, A. K., Bonet, R., Campbell, I. D. & Gilbert, R. J. Biophysical analysis of kindlin-3 reveals an elongated conformation and maps integrin binding to the membrane-distal β-subunit npxy motif. J. Biol. Chem. 287, 37715–37731 (2012).
Kadry, Y. A., Maisuria, E. M., Huet-Calderwood, C. & Calderwood, D. A. Differences in self-association between kindlin-2 and kindlin-3 are associated with differential integrin binding. J. Biol. Chem. 295, 11161–11173 (2020).
Paszek, M. J., Boettiger, D., Weaver, V. M. & Hammer, D. A. Integrin clustering is driven by mechanical resistance from the glycocalyx and the substrate. PLOS Comput. Biol. 5, e1000604 (2009).
Paszek, M. J. et al. The cancer glycocalyx mechanically primes integrin-mediated growth and survival. Nature 511, 319–325 (2014).
Lee, S. E., Kamm, R. D. & Mofrad, M. R. Force-induced activation of talin and its possible role in focal adhesion mechanotransduction. J. Biomech. 40, 2096–2106 (2007).
Haining, A. W. M., Von Essen, M., Attwood, S. J., Hyt'´onen, V. P. & del Río Hernández, A. All subdomains of the talin rod are mechanically vulnerable and may contribute to cellular mechanosensing. ACS Nano. 10, 6648–6658 (2016).
Zhou, J. et al. Mechanism of focal adhesion kinase mechanosensing. PLoS Comput. Biol. 11, e1004593 (2015).
Bauer, M. S. et al. Structural and mechanistic insights into mechanoactivation of focal adhesion kinase. Proc. Natl. Acad. Sci. USA 116, 6766–6774 (2019).
Sawada, Y. et al. Force sensing by mechanical extension of the src family kinase substrate p130cas. Cell 127, 1015–1026 (2006).
Baumann, K. Kindlin’the fate of mesenchymal stem cells. Nat. Rev. Mol. Cell Biol. 19, 279–279 (2018).
Guo, L. et al. Kindlin-2 regulates mesenchymal stem cell differentiation through control of yap1/taz. J. Cell Biol. 217, 1431–1451 (2018).
Jahed, Z., Haydari, Z., Rathish, A. & Mofrad, M. R. Kindlin is mechanosensitive: force-induced conformational switch mediates cross-talk among integrins. Biophys. J. 116, 1011–1024 (2019).
Nordenfelt, P., Elliott, H.-L. & Springer, T. A. Coordinated integrin activation by actin-dependent force during t-cell migration. Nat. Commun. 7, 13119 (2016).
Yao, M. et al. The mechanical response of talin. Nat. Commun. 7, 11966 (2016).
Li, W., Wolynes, P. G. & Takada, S. Frustration, specific sequence dependence, and nonlinearity in large-amplitude fluctuations of allosteric proteins. Proc. Natl. Acad. Sci. USA 108, 3504–3509 (2011).
Li, W., Wang, W. & Takada, S. Energy landscape views for interplays among folding, binding, and allostery of calmodulin domains. Proc. Natl. Acad. Sci. 111, 10550–10555 (2014).
Okazaki, K. -i., Koga, N., Takada, S., Onuchic, J. N. & Wolynes, P. G. Multiple-basin energy landscapes for large-amplitude conformational motions of proteins: Structure-based molecular dynamics simulations. Proc. Natl. Acad. Sci. USA 103, 11844–11849 (2006).
Li, W., Wang, J., Zhang, J., Takada, S. & Wang, W. Overcoming the bottleneck of the enzymatic cycle by steric frustration. Phys. Rev. Lett. 122, 238102 (2019).
Wang, Y., Gan, L., Wang, E. & Wang, J. Exploring the dynamic functional landscape of adenylate kinase modulated by substrates. J. Chem. Theory Comput. 9, 84–95 (2013).
Whitford, P. C., Miyashita, O., Levy, Y. & Onuchic, J. N. Conformational transitions of adenylate kinase: switching by cracking. J. Mol. Biol. 366, 1661–1671 (2007).
Zhang, W., Cao, Y., Li, W. & Wang, W. Allostery and molecular stripping mechanism in profilin regulated actin filament growth. N. J. Phys. 23, 123010 (2021).
Guan, X. et al. Bidirectional allostery mechanism in catch-bond formation of cd44 mediated cell adhesion. J. Phys. Chem. Lett. 15, 10786–10794 (2024).
Yao, X.-Q., Kenzaki, H., Murakami, S. & Takada, S. Drug export and allosteric coupling in a multidrug transporter revealed by molecular simulations. Nat. Commun. 1, 117 (2010).
Sun, Z., Guo, S. S. & Fässler, R. Integrin-mediated mechanotransduction. J. Cell Biol. 215, 445–456 (2016).
Bledzka, K. et al. Kindlin-2 directly binds actin and regulates integrin outside-in signaling. J. Cell Biol. 213, 97–108 (2016).
Böttcher, R. T. et al. Kindlin-2 recruits paxillin and arp2/3 to promote membrane protrusions during initial cell spreading. J. Cell Biol. 216, 3785–3798 (2017).
Liu, J. et al. Structural basis of phosphoinositide binding to kindlin-2 protein pleckstrin homology domain in regulating integrin activation. J. Biol. Chem. 286, 43334–43342 (2011).
Liu, Y., Zhu, Y., Ye, S. & Zhang, R. Crystal structure of kindlin-2 ph domain reveals a conformational transition for its membrane anchoring and regulation of integrin activation. Protein Cell 3, 434–440 (2012).
Nguyen, H. T. et al. Paxillin binding to the ph domain of kindlin-3 in platelets is required to support integrin αiibβ3 outside-in signaling. J. Thromb. Haemost. 19, 3126–3138 (2021).
Ferrara, P. & Caflisch, A. Folding simulations of a three-stranded antiparallel β-sheet peptide. Proc. Natl. Acad. Sci. USA 97, 10780–10785 (2000).
Zhang, Y., Zhang, J. & Wang, W. Atomistic analysis of pseudoknotted RNA unfolding. J. Am. Chem. Soc. 133, 6882–6885 (2011).
Capraro, D. T., Roy, M., Onuchic, J. N. & Jennings, P. A. Backtracking on the folding landscape of the β-trefoil protein interleukin-1β? Proc. Natl. Acad. Sci. USA 105, 14844–14848 (2008).
Tripathi, S., Makhatadze, G. I. & Garcia, A. E. Backtracking due to residual structure in the unfolded state changes the folding of the third fibronectin type iii domain from tenascin-c. J. Phys. Chem. B 117, 800–810 (2013).
Especial, J. N. & Faísca, P. F. A specific set of heterogeneous native interactions yields efficient knotting in protein folding. J. Phys. Chem. B 125, 7359–7367 (2021).
Li, W., Terakawa, T., Wang, W. & Takada, S. Energy landscape and multiroute folding of topologically complex proteins adenylate kinase and 2ouf-knot. Proc. Natl. Acad. Sci. 109, 17789–17794 (2012).
Onuchic, J. et al. Dodging the crisis of folding proteins with knots. Proc. Natl. Acad. Sci. USA 106, 3119–3124 (2009).
McEver, R. P. & Zhu, C. Rolling cell adhesion. Annu. Rev. Cell Dev. Biol. 26, 363–396 (2010).
Sokurenko, E. V., Vogel, V. & Thomas, W. E. Catch-bond mechanism of force-enhanced adhesion: counterintuitive, elusive, but… widespread? Cell Host Microbe 4, 314–323 (2008).
Ziegler, W. H., Liddington, R. C. & Critchley, D. R. The structure and regulation of vinculin. Trends Cell Biol. 16, 453–460 (2006).
Goult, B. T., Yan, J. & Schwartz, M. A. Talin as a mechanosensitive signaling hub. J. Cell Biol. 217, 3776–3784 (2018).
Wang, Y. et al. Force-dependent interactions between talin and full-length vinculin. J. Am. Chem. Soc. 143, 14726–14737 (2021).
Choi, H. K., Kim, H. G., Shon, M. J. & Yoon, T. Y. High-resolution single-molecule magnetic tweezers. Annu. Rev. Biochem. 91, 33–59 (2022).
Bustamante, C. J., Chemla, Y. R., Liu, S. & Wang, M. D. Optical tweezers in single-molecule biophysics. Nat. Rev. Methods Primers 1, 25 (2021).
Pal, K. Unravelling molecular mechanobiology using DNA-based fluorogenic tension sensors. J. Mater. Chem. B 13, 37–53 (2025).
Cost, A. L., Ringer, P., Chrostek-Grashoff, A. & Grashoff, C. How to measure molecular forces in cells: A guide to evaluating genetically-encoded fret-based tension sensors. Cell. Mol. Bioeng. 8, 96–105 (2015).
Tabrizi, M. A., Bhattacharyya, P., Zheng, R. & You, M. Electrochemical DNA-based sensors for measuring cell-generated forces. Biosens. Bioelectron. 253, 116185 (2024).
Chua, G. L., Tan, S. M. & Bhattacharjya, S. NMR characterization and membrane interactions of the loop region of kindlin-3f1 subdomain. PLoS ONE 11, e0153501 (2016).
Huray, K. I. L., Wang, H., Sobott, F. & Kalli, A. C. Systematic simulation of the interactions of pleckstrin homology domains with membranes. Sci. Adv. 8, eabn6992 (2022).
Onuchic, J. N., Luthey-Schulten, Z. & Wolynes, P. G. Theory of protein folding: the energy landscape perspective. Annu. Rev. Phys. Chem. 48, 545–600 (1997).
Clementi, C., Nymeyer, H. & Onuchic, J. N. Topological and energetic factors: what determines the structural details of the transition state ensemble and “en-route” intermediates for protein folding? An investigation for small globular proteins. J. Mol. Biol. 298, 937–953 (2000).
Wang, W., Xu, W.-X., Levy, Y., Trizac, E. & Wolynes, P. Confinement effects on the kinetics and thermodynamics of protein dimerization. Proc. Natl. Acad. Sci. 106, 5517–5522 (2009).
Kenzaki, H. et al. CafeMol: a coarse-grained biomolecular simulator for simulating proteins at work. J. Chem. Theory Comput. 7, 1979–1989 (2011).
Abraham, M. J. et al. GROMACS: high performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX 1, 19–25 (2015).
Bonomi, M. et al. PLUMED: a portable plugin for free-energy calculations with molecular dynamics. Comput. Phys. Commun. 180, 1961–1972 (2009).
DeLano, W. L. et al. Pymol: an open-source molecular graphics tool. CCP4 Newsl. Protein Crystallogr 40, 82–92 (2002).
Durrant, J. D. BlendMol: advanced macromolecular visualization in Blender. Bioinformatics 35, 2323–2325 (2019).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant Nos. 11974173 and 12347102), the Basic Research Program of Jiangsu Province (BK20253050), the grant of Wenzhou Institute, University of Chinese Academy of Sciences (WIUCASQD2021010), and the Shenzhen Science and Technology Program (Grant No. 20231121095506001 to C.Y.). The authors also thank the partial support of HPC Center of Nanjing University and HPC center of Wenzhou Institute, e-Science center of Nanjing University, Nanjing Kunpeng&Ascend Center of Cultivation, and the Nanjing Key Laboratory for Cardiovascular Information and Health Engineering Medicine (funded by the Nanjing Municipal Health Commission) and its Jiangsu counterpart. C.Y. and Z.W. are investigators of SUSTech Institute for Biological Electron Microscopy.
Author information
Authors and Affiliations
Contributions
W.L. conceived the study, W.Z. conducted the MD simulations, H.Y., Z.Y., Z.W., M.Y., and C.Y. conducted the experiments, W.Z., H.Y., Z.Y., Y.B., J.Z., Z.W., M.Y., W.L., C.Y., and W.W. analyzed the results, W.L., C.Y., and W.W. supervised the project. W.Z., W.L. and W.W. wrote the manuscript with input from all authors. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Physics thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, W., Yang, H., Yang, Z. et al. Role of mechanical allostery in kindlin-mediated integrin activation. Commun Phys (2026). https://doi.org/10.1038/s42005-026-02557-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42005-026-02557-z