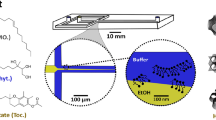
Abstract
Liquid crystal (LC)-aqueous interfaces were shown to respond to the phospholipid interactions via optically observable ordering transitions; however, past attempts lack the quantification of the transport and fusion kinetics of the vesicles at the interfaces. Herein, we investigated the response of flowing LC-aqueous interfaces upon fusion of the vesicles formed by pure 1,2-dilauroyl-sn-glycero-3-phosphocholine (DLPC), 1,2-dioleoyl-sn-glycero-3-phosphocholine (DOPC), 1,2-dipalmitoyl-sn-glycero-3-phosphocholine (DPPC), or egg sphingomyelin or their mixtures with guest molecules. Using stabilized LC-aqueous interfaces in transparent microfluidic chips that allow spatiotemporal quantification using fluorescence, confocal, and polarized light microscopy, we demonstrated that flowing LC interfaces provide a rapid response to lipid adsorption, where their spatiotemporal interfacial distribution differs depending on the mechanical properties of their vesicles. We show that cholesterol-dissolved lipid complexes result in distinct LC-response kinetics, mainly associated to the changes in their rigidities. Considering the critical role of the mechanical properties of cell membranes in proper cellular function, this study is significant as it offers a continuous and rapid early diagnosis platform for detecting minor mechanical alterations in lipid bilayers, which may lead to cell dysfunction that contributes to critical diseases.
Similar content being viewed by others
Data availability
All raw data supporting the findings of this study are openly available at https://doi.org/10.5281/zenodo.18976028.
References
Iwasa, J.; Marshall, W. F.; Karp, G. Karp’s Cell and Molecular Biology: Concepts and Experiments (Wiley, 2020).
Dias, C. & Nylandsted, J. Plasma membrane integrity in health and disease: significance and therapeutic potential. Cell Discov. 7, 1–18 (2021).
Faizi, H. A., Frey, S. L., Steinkühler, J., Dimova, R. & Vlahovska, P. M. Bending rigidity of charged lipid bilayer membranes. Soft Matter 15, 6006–6013 (2019).
Tsuchiya, H. et al. Membrane-rigidifying effects of anti-cancer dietary factors. BioFactors 16, 45–56 (2002).
D’auria, L. et al. Psychosine enhances the shedding of membrane microvesicles: implications in demyelination in Krabbe’s Disease. PLoS ONE 12, 1–19 (2017).
Kashani, A. S. & Packirisamy, M. Cancer cells optimize elasticity for efficient migration: migratory index. R. Soc. 7, 1–16 (2020).
Sarna, M. et al. Cell elasticity is an important indicator of the metastatic phenotype of melanoma cells. Exp. Dermatol. 23, 813–818 (2014).
Kwon, S., Yang, W., Moon, D. & Kim, K. S. Comparison of cancer cell elasticity by cell type. J. Cancer 11, 5403–5412 (2020).
Hayashi, K. & Iwata, M. Stiffness of cancer cells measured with an AFM indentation method. J. Mech. Behav. Biomed. Mater. 49, 105–111 (2015).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Skog, J. et al. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat. Cell Biol. 10, 1470–1476 (2008).
Peinado, H. et al. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat. Med. 18, 883–891 (2012).
Stawarska, A. et al. Extracellular vesicles as next-generation diagnostics and advanced therapy medicinal products. Int. J. Mol. Sci. 25, 1–29 (2024).
Xu, R. et al. Extracellular vesicles in cancer—implications for future improvements in cancer care. Nat. Rev. Clin. Oncol. 15, 617–638 (2018).
Goñi, F. M. & Alonso, A. Effects of ceramide and other simple sphingolipids on membrane lateral structure. Biochim. Biophys. Acta Biomembr. 1788, 169–177 (2009).
Jones, M. N. Surfactant interactions with biomembranes and proteins. Chem. Soc. Rev. 21, 127–136 (1992).
Crane, J. M. & Tamm, L. K. Role of cholesterol in the formation and nature of lipid rafts in planar and spherical model membranes. Biophys. J. 86, 2965–2979 (2004).
Levental, I. & Lyman, E. Regulation of membrane protein structure and function by their lipid nano-environment. Nat. Rev. Mol. Cell Biol. 24, 107–122 (2023).
Song, Y., Liu, J., Zhao, K., Gao, L. & Zhao, J. Cholesterol-induced toxicity: an integrated view of the role of cholesterol in multiple diseases. Cell Metab. 33, 1911–1925 (2021).
Pan, J., Mills, T. T., Tristram-Nagle, S. & Nagle, J. F. Cholesterol perturbs lipid bilayers nonuniversally. Phys. Rev. Lett. 100, 1–4 (2008).
Luo, J., Yang, H. & Song, B. L. Mechanisms and regulation of cholesterol homeostasis. Nat. Rev. Mol. Cell Biol. 21, 225–245 (2020).
Emanuelsson, F., Nordestgaard, B. G. & Benn, M. Familial hypercholesterolemia and risk of peripheral arterial disease and chronic kidney disease. J. Clin. Endocrinol. Metab. 103, 4491–4500 (2018).
Zambón, D. et al. Higher incidence of mild cognitive impairment in familial hypercholesterolemia. Am. J. Med. 123, 267–274 (2010).
Gamerdinger, M., Clement, A. B. & Behl, C. Cholesterol-like effects of selective cyclooxygenase inhibitors and fibrates on cellular membranes and amyloid-β production. Mol. Pharm. 72, 141–151 (2007).
Gan, L. T. et al. Hepatocyte free cholesterol lipotoxicity results from JNK1-mediated mitochondrial injury and is HMGB1 and TLR4-dependent. J. Hepatol. 61, 1376–1384 (2014).
Brake, J. M., Daschner, M. K., Luk, Y. Y. & Abbott, N. L. Biomolecular interactions at phospholipid-decorated surfaces of liquid crystals. Science 302, 2094–2097 (2003).
Bai, Y. & Abbott, N. L. Recent advances in colloidal and interfacial phenomena involving liquid crystals. Langmuir 27, 5719–5738 (2011).
Bukusoglu, E., Pantoja, M. B., Mushenheim, P. C., Wang, X. & Abbott, N. L. Design of responsive and active (soft) materials using liquid crystals. Annu. Rev. Chem. Biomol. Eng. 7, 163–196 (2016).
Gupta, J. K., Meli, M. V., Teren, S. & Abbott, N. L. Elastic energy-driven phase separation of phospholipid monolayers at the nematic liquid-crystal-aqueous interface. Phys. Rev. Lett. 100, 1–4 (2008).
Brake, J. M., Daschner, M. K. & Abbott, N. L. Formation and characterization of phospholipid monolayers spontaneously assembled at interfaces between aqueous phases and thermotropic liquid crystals. Langmuir 21, 2218–2228 (2005).
Lockwood, N. A. & Abbott, N. L. Self-assembly of surfactants and phospholipids at interfaces between aqueous phases and thermotropic liquid crystals. Curr. Opin. Colloid Interface Sci. 10, 111–120 (2005).
Tsuei, M., Shivrayan, M., Kim, Y. K., Thayumanavan, S. & Abbott, N. L. Optical “Blinking” triggered by collisions of single supramolecular assemblies of amphiphilic molecules with interfaces of liquid crystals. J. Am. Chem. Soc. 142, 6139–6148 (2020).
Hartono, D., Qin, W. J., Yang, K. L. & Yung, L. Y. L. Imaging the disruption of phospholipid monolayer by protein-coated nanoparticles using ordering transitions of liquid crystals. Biomaterials 30, 843–849 (2009).
Sadati, M. et al. Liquid crystal enabled early stage detection of beta amyloid formation on lipid monolayers. Adv. Funct. Mater. 25, 6050–6060 (2015).
Devi, M., Verma, I. & Pal, S. K. Distinct interfacial ordering of liquid crystals observed by protein-lipid interactions that enabled the label-free sensing of cytoplasmic protein at the liquid crystal-aqueous interface. Analyst 146, 7152–7159 (2021).
Gupta, T., Lata, K., Chattopadhyay, K. & Pal, S. K. Utilizing an aqueous-liquid crystal interface to investigate membrane protein interactions and mutation effects of a pore-forming toxin. J. Mater. Chem. B 13, 5358–5364 (2025).
Pani, I., Nazreen, F., Sharma, K. M. & Pal, M. S. K. Probing nanoscale lipid-protein interactions at the interface of liquid crystal droplets. Nano Lett. 21, 4546–4553 (2021).
Lin, I. H. et al. Endotoxin-induced structural transformations in liquid crystalline droplets. Science 332, 1297–1300 (2011).
Paterson, D. A. et al. Control of director fields in phospholipid-coated liquid crystal droplets. Langmuir 36, 6436–6446 (2020).
Carter, M. C. D. et al. Synthetic mimics of bacterial lipid a trigger optical transitions in liquid crystal microdroplets at ultralow picogram-per-milliliter concentrations. Langmuir 31, 12850–12855 (2015).
Sharma, A., Gupta, D., Scalia, G. & Lagerwall, J. P. F. Lipid islands on liquid crystal shells. Phys. Rev. Res. 4, 1–24 (2022).
Yang, C. et al. Local high-density distributions of phospholipids induced by the nucleation and growth of smectic liquid crystals at the interface. Chin. Chem. Lett. 33, 3973–3976 (2022).
Lee, H. G., Munir, S. & Park, S. Y. Cholesteric liquid crystal droplets for biosensors. ACS Appl. Mater. Interfaces 8, 26407–26417 (2016).
Concellón, A., Fong, D. & Swager, T. M. Complex liquid crystal emulsions for biosensing. J. Am. Chem. Soc. 143, 9177–9182 (2021).
Sun, H. et al. Development of a liquid crystal-based α-glucosidase assay to detect anti-diabetic drugs. Microchem. J. 167, 1–6 (2021).
Nguyen, D. K. & Jang, C. H. A cationic surfactant-decorated liquid crystal-based aptasensor for label-free detection of malathion pesticides in environmental samples. Biosensors 11, 1–11 (2021).
Song, H. et al. Construction of liquid crystal-based sensors using enzyme-linked dual-functional nucleic acid on magnetic beads. Anal. Chem. 95, 13385–13390 (2023).
Karaman, D., Saylam, A., Akar, E., Özçubukçu, S. & Bukusoglu, E. Liquid crystals that respond to the aqueous phase reactions catalyzed by synthetic enzyme mimics. Adv. Mater. Interfaces 12, 1–13 (2025).
Karaman, D., Akar, E., Saylam, A., Özçubukçu, S. & Bukusoglu, E. Engineered fullerene-based enzyme mimics for enhanced sensing of aqueous-phase reactions using liquid crystals. ACS Appl. Nano Mater. 8, 22798–22808 (2025).
Heerklotz, H. Interactions of surfactants with lipid membranes. Q Rev. Biophys. 41, 205–264 (2008).
Pantaler, E., Kamp, D. & Haest, C. W. M. Acceleration of phospholipid flip-flop in the erythrocyte membrane by detergents differing in polar head group and alkyl chain length. Biochim. Biophys. Acta Biomembr. 1509, 397–408 (2000).
Silva, L., De Almeida, R. F. M., Fedorov, A., Matos, A. P. A. & Prieto, M. Ceramide-platform formation and—induced biophysical changes in a fluid phospholipid membrane. Mol. Membr. Biol. 23, 137–148 (2006).
Gracià, R. S., Bezlyepkina, N., Knorr, R. L., Lipowsky, R. & Dimova, R. Effect of cholesterol on the rigidity of saturated and unsaturated membranes: fluctuation and electrodeformation analysis of giant vesicles. Soft Matter 6, 1472–1482 (2010).
Pan, J., Tristram-Nagle, S. & Nagle, J. F. Effect of cholesterol on structural and mechanical properties of membranes depends on lipid chain saturation. Phys. Rev. E Stat. Nonlin. Soft Matter Phys. 80, 1–2 (2009).
Karal, M. A. S. et al. Effects of cholesterol on the size distribution and bending modulus of lipid vesicles. PLoS ONE 17, 1–24 (2022).
Chakraborty, S. et al. How cholesterol stiffens unsaturated lipid membranes. Proc. Natl. Acad. Sci. USA 117, 21896–21905 (2020).
Kaddah, S., Khreich, N., Kaddah, F., Charcosset, C. & Greige-Gerges, H. Cholesterol modulates the liposome membrane fluidity and permeability for a hydrophilic molecule. Food Chem. Toxicol. 113, 40–48 (2018).
Karal, M. A. S., Billah, M. M., Ahmed, M. & Ahamed, M. K. A review on the measurement of the bending rigidity of lipid membranes. Soft Matter 19, 8285–8304 (2023).
Pöhnl, M., Trollmann, M. F. W. & Böckmann, R. A. Nonuniversal impact of cholesterol on membranes mobility, curvature sensing and elasticity. Nat. Commun. 14, 1–12 (2023).
Espinosa, G., López-Montero, I., Monroy, F. & Langevin, D. Shear rheology of lipid monolayers and insights on membrane fluidity. Proc. Natl. Acad. Sci. USA 108, 6008–6013 (2011).
Vrânceanu, M., Winkler, K., Nirschl, H. & Leneweit, G. Surface rheology and phase transitions of monolayers of phospholipid/cholesterol mixtures. Biophys. J. 94, 3924–3934 (2008).
Beyazkilic, P. et al. Contactless pulsed and continuous microdroplet release using photothermal liquid crystals. Adv. Funct. Mater. 32, 1–29 (2022).
Maiti, S., Roh, S., Cohen, I. & Abbott, N. L. Non-equilibrium ordering of liquid crystalline (LC) films driven by external gradients in surfactant concentration. J. Colloid Interface Sci. 637, 134–146 (2023).
Özşahin, A. N. & Bukusoglu, E. Responsive soft interface liquid crystal microfluidics. Adv. Mater. Interfaces 11, 1–13 (2024).
İlhan, G., Carenza, L. N. & Bukusoglu, E. Shear-induced structural transitions in confined nematic soft interfaces. Commun. Phys. 8, 1–15 (2025).
Gupta, J. K., Zimmerman, J. S., De Pablo, J. J., Caruso, F. & Abbott, N. L. Characterization of adsorbate-induced ordering transitions of liquid crystals within monodisperse droplets. Langmuir 25, 9016–9024 (2009).
Doktorova, M., Harries, D. & Khelashvili, G. Determination of bending rigidity and tilt modulus of lipid membranes from real-space fluctuation analysis of molecular dynamics simulations. Phys. Chem. Chem. Phys. 19, 16806–16818 (2017).
Arunachalam, B., Jaganathan, M., Palanisamy, T. & Dhathathreyan, A. Physico-chemical studies of elastic compliance and adsorption of dopc vesicles and its mixture with charged lipids at fluid/solid interface. Colloids Surf. B Biointerfaces 199, 1–9 (2021).
Pinazo, A., Wen, X., Liao, Y. C., Prosser, A. J. & Franses, E. I. Comparison of DLPC and DPPC in controlling the dynamic adsorption and surface tension of their aqueous dispersions. Langmuir 18, 8888–8896 (2002).
Jiménez-Rojo, N., García-Arribas, A. B., Sot, J., Alonso, A. & Goñi, F. M. Lipid bilayers containing sphingomyelins and ceramides of varying N-acyl lengths: a glimpse into sphingolipid complexity. Biochim. Biophys. Acta Biomembr. 1838, 456–464 (2014).
Choi, S. Q., Steltenkamp, S., Zasadzinski, J. A. & Squires, T. M. Active microrheology and simultaneous visualization of sheared phospholipid monolayers. Nat. Commun. 2, 1–6 (2011).
Et-Thakafy, O., Delorme, N., Guyomarc’h, F. & Lopez, C. Mechanical properties of milk sphingomyelin bilayer membranes in the gel phase: effects of naturally complex heterogeneity, saturation and acyl chain length investigated on liposomes using AFM. Chem. Phys. Lipids 210, 47–59 (2018).
Catapano, E. R., Natale, P., Monroy, F. & López-Montero, I. The enzymatic sphingomyelin to ceramide conversion increases the shear membrane viscosity at the air-water interface. Adv. Colloid Interface Sci. 247, 555–560 (2017).
Alonso, A. & Gõ Ni, F. M. The physical properties of ceramides in membranes. Annu. Rev. Biophys. 176, 633–654 (2024).
Kessel, A., Ben-Tal, N. & May, S. Interactions of cholesterol with lipid bilayers: the preferred configuration and fluctuations. Biophys. J. 81, 643–658 (2001).
Dimova, R. Recent developments in the field of bending rigidity measurements on membranes. Adv. Colloid Interface Sci. 208, 225–234 (2014).
Henriksen, J. et al. Universal behavior of membranes with sterols. Biophys. J. 90, 1639–1649 (2006).
Miyoshi, T. & Kato, S. Detailed analysis of the surface area and elasticity in the saturated 1,2-diacylphosphatidylcholine/cholesterol binary monolayer system. Langmuir 31, 9086–9096 (2015).
Huang, J. & Feigenson, G. W. A microscopic interaction model of maximum solubility of cholesterol in lipid bilayers. Biophys. J. 76, 2142–2157 (1999).
Dai, J., Alwarawrah, M. & Huang, J. instability of cholesterol clusters in lipid bilayers and the cholesterol’s umbrella effect. J. Phys. Chem. B 114, 840–848 (2010).
Sabatini, K., Mattila, J. P. & Kinnunen, P. K. J. Interfacial behavior of cholesterol, ergosterol, and lanosterol in mixtures with DPPC and DMPC. Biophys. J. 95, 2340–2355 (2008).
Bykov, A. G. & Noskov, B. A. Surface dilatational elasticity of pulmonary surfactant solutions in a wide range of surface tensions. Colloid J. 80, 467–473 (2018).
Ortega-Sanchez, F. G. et al. Microfluidic systems in extracellular vesicles single analysis. a systematic review. TrAC Trends Anal. Chem. 159, 116920 (2023).
Lavrentovich, O. D. & Zola, E. Fluorescence confocal polarizing microscopy: three-dimensional imaging of the director. PRAMANA Indian Acad. Sci. 61, 373–384 (2003).
Acknowledgements
Financial support from the European Research Council under Starting Grant, LCFlow (grant agreement no. 101039294; awardee, Emre Bukusoglu) is gratefully acknowledged. This work was partially supported by the Research Fund of the Middle East Technical University. Project Number: 11628. The authors thank Dr. Burak Akdeniz for their help in developing the COMSOL simulations.
Author information
Authors and Affiliations
Contributions
C.D. conducted experiments and characterizations. E.B. supervised the research. All authors contributed to data interpretation, discussions, and manuscript preparation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Materials thanks Carmelo La Rosa and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dedeoglu, C., Bukusoglu, E. Soft-interfaced liquid crystal microfluidics can probe the rigidity of lipid vesicles. Commun Mater (2026). https://doi.org/10.1038/s43246-026-01128-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43246-026-01128-7