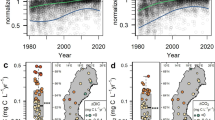
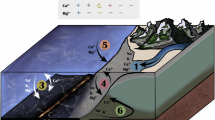
Abstract
The coastal ocean links land and sea through rivers and submarine groundwater discharge, which contribute to the coastal carbon budget. Groundwater discharge, including fresh groundwater and recirculated seawater, remains poorly constrained globally. Here, we compile a global dataset of coastal groundwater chemistry and estimate fluxes of dissolved inorganic carbon and total alkalinity. Using conceptual reaction models, we analyze the alkalinity-carbon relationship to identify dominant processes. These patterns reflect carbonate dissolution and precipitation, and organic matter remineralization under oxic and anoxic conditions, indicating that coastal aquifers function as geochemical reactors. Recirculated seawater sampled inland is more enriched than nearshore groundwater, consistent with longer residence time and enhanced water-rock interaction. Groundwater contributes 3–7 percent of riverine dissolved inorganic carbon flux, equivalent to 2.7–2.9 and 2.2-2.4 trillion moles per year of dissolved inorganic carbon and alkalinity, depending on lithology and redox conditions. This emphasizes the importance of incorporating groundwater fluxes into Earth system models.
Similar content being viewed by others
Data availability
The study uses compiled groundwater geochemical data (dissolved inorganic carbon (DIC), total alkalinity (TA), pH, major ions concentration, salinity classification, lithology, and derived mixing) from 58 previously published studies. The processed and harmonized dataset used for the global submarine groundwater discharge (SGD) flux calculations has been deposited in the Zenodo repository and is publicly available at https://doi.org/10.5281/zenodo.18833297.
References
Friedlingstein, P. Carbon cycle feedbacks and future climate change. Philos. Trans. R. Soc. A: Math. Phys. Eng. Sci. 373, 20140421 (2015).
Berner, R. A., Lasaga, A. C. & Garrels, R. M. Carbonate-silicate geochemical cycle and its effect on atmospheric carbon dioxide over the past 100 million years. Am. J. Sci. 283, 641–683 (1983).
Isson, T. T. et al. Evolution of the global carbon cycle and climate regulation on Earth. Glob. Biogeochem. Cycles 34, e2018GB006061 (2020).
Devries, T. The ocean carbon cycle. Annu. Rev. Environ. Resour. 47, 317–341 (2022).
Bauer, J. E. et al. The changing carbon cycle of the coastal ocean. Nature 504, 61–70 (2013).
Burnett, W. C., Bokuniewicz, H., Moore, W. S. & Taniguchi, M. Groundwater and pore water inputs to the coastal zone. Biogeochemistry 66, 3–33 (2003).
Moore, W. S. The effect of submarine groundwater discharge on the ocean. Ann. Rev. Mar. Sci. 2, 59–88 (2010).
Kwon, E. Y. et al. Global estimate of submarine groundwater discharge based on an observationally constrained radium isotope model. Geophys. Res. Lett. 41, 8438–8444 (2014).
Wilson, S. J. et al. Global subterranean estuaries modify groundwater nutrient loading to the ocean. Limnol. Oceanogr. Lett. 9, 411–422 (2024).
Kiro, Y. Coastal aquifers key contributors to ocean chemistry through solute fluxes. Nat. Commun. 16, 7082 (2025).
Post, V. & Abarca, E. Preface: saltwater and freshwater interactions in coastal aquifers. Hydrogeol. J. 18, 1–4 (2010).
Santos, I. R., Eyre, B. D. & Huettel, M. The driving forces of porewater and groundwater flow in permeable coastal sediments: a review. Estuar. Coast. Shelf Sci. 98, 1–15 (2012).
Wood, W. W. & Hyndman, D. W. Groundwater depletion: a significant unreported source of atmospheric carbon dioxide. Earth’s. Future 5, 1133–1135 (2017).
Robinson, C. E. et al. Groundwater dynamics in subterranean estuaries of coastal unconfined aquifers: controls on submarine groundwater discharge and chemical inputs to the ocean. Adv. Water Resour. 115, 315–331 (2018).
Zhang, S. & Planavsky, N. J. Revisiting groundwater carbon fluxes to the ocean with implications for the carbon cycle. Geology 48, 67–71 (2019).
Middelburg, J. J., Soetaert, K. & Hagens, M. Ocean alkalinity, buffering and biogeochemical processes. Rev. Geophys. 58, e2019RG000681 (2020).
Liu, M. et al. Global riverine land-to-ocean carbon export constrained by observations and multi-model assessment. Nat. Geosci. 17, 896–904 (2024).
Cole, J. J. et al. Plumbing the global carbon cycle: integrating inland waters into the terrestrial carbon budget. Ecosystems 10, 171–184 (2007).
Luijendijk, E., Gleeson, T. & Moosdorf, N. Fresh groundwater discharge insignificant for the world’s oceans but important for coastal ecosystems. Nat. Commun. 11, 1260 (2020).
Liu, Q. et al. Effect of submarine groundwater discharge on the coastal ocean inorganic carbon cycle. Limnol. Oceanogr. 59, 1529–1554 (2014).
Stewart, B. T., Santos, I. R., Tait, D. R., Macklin, P. A. & Maher, D. T. Submarine groundwater discharge and associated fluxes of alkalinity and dissolved carbon into Moreton Bay (Australia) estimated via radium isotopes. Mar. Chem. 174, 1–12 (2015).
Cai, W. J., Wang, Y., Krest, J. & Moore, W. S. The geochemistry of dissolved inorganic carbon in a surficial groundwater aquifer in North Inlet, South Carolina, and the Carbon fluxes to the coastal ocean. Geochim. Cosmochim. Acta 67, 631–639 (2003).
Szymczycha, B., Maciejewska, A., Winogradow, A. & Pempkowiak, J. Could submarine groundwater discharge be a significant carbon source to the southern Baltic Sea? Oceanologia 56, 327–347 (2014).
Chen, X. et al. Submarine Groundwater discharge-derived carbon fluxes in mangroves: an important component of blue carbon budgets? J. Geophys. Res. Oceans 123, 6962–6979 (2018).
Chaillou, G., Couturier, M., Tommi-Morin, G. & Rao, A. M. F. Total alkalinity and dissolved inorganic carbon production in groundwaters discharging through a sandy beach. Procedia Earth Planet. Sci. 10, 88–99 (2014).
Regnier, P., Resplandy, L., Najjar, R. G. & Ciais, P. The land-to-ocean loops of the global carbon cycle. Nature 603, 401–410 (2022).
Robinson, C., Gibbes, B. & Li, L. Driving mechanisms for groundwater flow and salt transport in a subterranean estuary. Geophys. Res. Lett. 33, L03402 (2006).
King, J. N., Mehta, A. J. & Dean, R. G. Analytical models for the groundwater tidal prism and associated benthic water flux. Hydrogeol. J. 18, 203–215 (2010).
Russoniello, C. J., Heiss, J. W. & Michael, H. A. Variability in benthic exchange rate, depth, and residence time beneath a shallow coastal estuary. J. Geophys. Res. Oceans 123, 1860–1876 (2018).
Sivan, O., Yechieli, Y., Herut, B. & Lazar, B. Geochemical evolution and timescale of seawater intrusion into the coastal aquifer of Israel. Geochim. Cosmochim. Acta 69, 579–592 (2005).
Caschetto, M., Colombani, N., Mastrocicco, M., Petitta, M. & Aravena, R. Nitrogen and sulphur cycling in the saline coastal aquifer of Ferrara, Italy. A multi-isotope approach. Appl. Geochem. 76, 88–98 (2017).
Liu, Q. et al. Carbonate system biogeochemistry in a subterranean estuary – Waquoit Bay, USA. Geochim. Cosmochim. Acta 203, 422–439 (2017).
Sola, F. & Vallejos, A. Long and short-term cation exchange linked to a negative hydraulic barrier in a coastal aquifer. Sci. Total Environ. 819, 152013 (2022).
Stein, S., Sivan, O., Yechieli, Y. & Kasher, R. Redox condition of saline groundwater from coastal aquifers influences reverse osmosis desalination process. Water Res. 188, 116508 (2021).
Anschutz, P. et al. Terrestrial groundwater and nutrient discharge along the 240-km-long Aquitanian coast. Mar. Chem. 185, 38–47 (2016).
Meredith, K. T. et al. Isotopic and chromatographic fingerprinting of the sources of dissolved organic carbon in a shallow coastal aquifer. Hydrol. Earth Syst. Sci. 24, 2167–2178 (2020).
Smith, R. L., Kent, D. B., Repert, D. A. & Böhlke, J. K. Anoxic nitrate reduction coupled with iron oxidation and attenuation of dissolved arsenic and phosphate in a sand and gravel aquifer. Geochim. Cosmochim. Acta 196, 102–120 (2017).
Roy, M., Martin, J. B., Smith, C. G. & Cable, J. E. Reactive-transport modeling of iron diagenesis and associated organic carbon remineralization in a Florida (USA) subterranean estuary. Earth Planet. Sci. Lett. 304, 191–201 (2011).
Roy, M., Martin, J. B., Cherrier, J., Cable, J. E. & Smith, C. G. Influence of sea level rise on iron diagenesis in an east Florida subterranean estuary. Geochim. Cosmochim. Acta 74, 5560–5573 (2010).
Charette, M. A. & Sholkovitz, E. R. Trace element cycling in a subterranean estuary: Part 2. Geochemistry of the pore water. Geochim. Cosmochim. Acta 70, 811–826 (2006).
Andersen, M. S., Nyvang, V., Jakobsen, R. & Postma, D. Geochemical processes and solute transport at the seawater/freshwater interface of a sandy aquifer. Geochim. Cosmochim. Acta 69, 3979–3994 (2005).
Hartmann, J. & Moosdorf, N. The new global lithological map database GLiM: a representation of rock properties at the Earth surface. Geochem. Geophys. Geosyst. 13, Q12004 (2012).
Zhou, Y. Q., Sawyer, A. H., David, C. H. & Famiglietti, J. S. Fresh submarine groundwater discharge to the near-global coast. Geophys. Res. Lett. 46, 5855–5863 (2019).
Reznik, I. J. et al. Fresh and saline groundwater ages and flow dynamics in a perturbed coastal aquifer. J. Hydrol. 597, 125721 (2021).
Riedl, R. J., Huang, N. & Machan, R. The subtidal pump: a mechanism of interstitial water exchange by wave action. Mar. Biol. 13, 210–221 (1972).
Wang, G. et al. Net subterranean estuarine export fluxes of dissolved inorganic C, N, P, Si, and total alkalinity into the Jiulong River estuary, China. Geochim. Cosmochim. Acta 149, 103–114 (2015).
Pain, A. J., Martin, J. B., Young, C. R., Valle-Levinson, A. & Mariño-Tapia, I. Carbon and phosphorus processing in a carbonate karst aquifer and delivery to the coastal ocean. Geochim. Cosmochim. Acta 269, 484–495 (2020).
Russoniello, C. J. Exploring submarine groundwater discharge into the Delaware Inland Bays over diverse scales with direct measurements and modeling. PhD thesis, University of Delaware, Newark, USA, (2012).
Friedlingstein, P. et al. Global Carbon Budget 2020. Earth Syst. Sci. Data 12, 3269–3340 (2020).
Canadell, J. G. et al. Global carbon and other biogeochemical cycles and feedbacks. in IPCC AR6 WGI, final government distribution 673–816. https://doi.org/10.1017/9781009157896.007. (Cambridge University Press, 2021).
Sawyer, A. H. et al. Stratigraphic controls on fluid and solute fluxes across the sediment-water interface of an estuary. Limnol. Oceanogr. 59, 997–1010 (2014).
Kiro, Y., Weinstein, Y., Starinsky, A. & Yechieli, Y. Application of radon and radium isotopes to groundwater flow dynamics: an example from the Dead Sea. Chem. Geol. 411, 155–171 (2015).
Parkhurst, D. L. & Appelo, C. A. J. User’s guide to PHREEQC (version 2): A computer program for speciation,batch-reaction, one-dimensional transport, and inverse geochemical calculations. U.S. Geological Survey Water-Resources Investigations Report 99–4259 (1999).
Acknowledgements
We want to thank the Sustainability and Energy Research Initiative (SAERI) grant, the Helen Kimmel Center for Planetary Science grant, the Minerva Foundation, the Angel Faivovich Foundation for ecological research, and the De Botton Center for Marine Sciences grant for their funding support for this study. Additionally, we acknowledge the support from the Zuckerman Faculty Scholars program, the Center for Scientific Excellence research grant, the Raymond Lapon Fund research grant, the Estate of David Levinson, the Paul and Tina Gardner research grant, and the Center for New Scientists at the Weizmann Institute of Science research grant.
Author information
Authors and Affiliations
Contributions
Nurit Weber designed the research, compiled and analysed the global groundwater dataset, performed the geochemical and flux calculations, and wrote the manuscript. Yael Kiro contributed to the conceptual framework and research design, interpreted the results, contributed to the development of the reaction-based modeling approach, and revised and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Earth and Environment thanks Gwénaëlle Chaillou and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Nicola Colombo. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
43247_2026_3430_MOESM2_ESM.pdf (download PDF )
Supplementary Information for „Alkalinity and carbon fluxes from coastal aquifers to the ocean via submarine groundwater discharge“
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Weber, N., Kiro, Y. Alkalinity and carbon fluxes from coastal aquifers to the ocean via submarine groundwater discharge. Commun Earth Environ (2026). https://doi.org/10.1038/s43247-026-03430-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43247-026-03430-1