Abstract
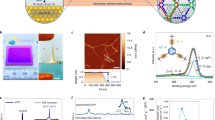
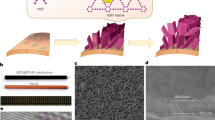
Developing artificial membranes with stable and uniform angstrom-scale channels that can effectively reject hydrated ions is a substantial challenge but important in water desalination and energy conversion/storage applications. Achieving precise water/ions separation while maintaining high water flux requires a membrane microstructure engineered with molecular precision. This study reports the successful synthesis of ultra-thin, centimetre-scale graphdiyne (GDY) films with ordered one-dimensional (1D) channels using single-crystalline Cu (111) as the growth substrate and demonstrates their exceptional performance as molecular sieves for highly efficient water/ion separation. The optimized membrane exhibits an ultra-high water/NaCl selectivity of 5.96 × 104, outperforming state-of-the-art membranes, at a water permeance of ∼32.9 mol m−2 h−1 bar−1 and a salt rejection exceeding 99.7% for small ions in seawater. Mechanism studies reveal that the hydrophobic angstrom-scale channels in GDY crystals force water molecules into a single-file configuration with 1D hydrogen bond during water permeation. The 1D water chain enables the GDY membrane to facilitate rapid (diffusion constant as high as 1.3 × 10−4 cm2 s−1) and selective proton transport via the Grotthuss mechanism. This work contributes to the development of carbon nanomaterial membranes for precise molecular sieving and biomimetic protonophores.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the findings of this study are available in the paper and its Supplementary Information. Source data are provided with this paper.
Change history
26 February 2025
A Correction to this paper has been published: https://doi.org/10.1038/s44221-025-00415-w
References
Elimelech, M. & Phillip, W. A. The future of seawater desalination: energy, technology, and the environment. Science 333, 712–717 (2011).
Koros, W. J. & Zhang, C. Materials for next-generation molecularly selective synthetic membranes. Nat. Mater. 16, 289–297 (2017).
Lively, R. P. & Sholl, D. S. From water to organics in membrane separations. Nat. Mater. 16, 276–279 (2017).
Murata, K. et al. Structural determinants of water permeation through aquaporin-1. Nature 407, 599–605 (2000).
Tunuguntla, R. H. et al. Enhanced water permeability and tunable ion selectivity in subnanometer carbon nanotube porins. Science 357, 792–796 (2017).
Gopinadhan, K. et al. Complete steric exclusion of ions and proton transport through confined monolayer water. Science 363, 145–148 (2019).
Abraham, J. et al. Tunable sieving of ions using graphene oxide membranes. Nat. Nanotechnol. 12, 546–550 (2017).
Ries, L. et al. Enhanced sieving from exfoliated MoS2 membranes via covalent functionalization. Nat. Mater. 18, 1112–1117 (2019).
Ding, L. et al. Effective ion sieving with Ti3C2Tx MXene membranes for production of drinking water from seawater. Nat. Sustain. 3, 296–302 (2020).
Joshi, R. K. et al. Precise and ultrafast molecular sieving through graphene oxide membranes. Science 343, 752–754 (2014).
Lu, Z. et al. Self-crosslinked MXene (Ti3C2Tx) membranes with good antiswelling property for monovalent metal ion exclusion. ACS Nano 13, 10535–10544 (2019).
Deng, M., Kwac, K., Li, M., Jung, Y. & Park, H. G. Stability, molecular sieving, and ion diffusion selectivity of a lamellar membrane from two-dimensional molybdenum disulfide. Nano Lett. 17, 2342–2348 (2017).
Chen, L. et al. Ion sieving in graphene oxide membranes via cationic control of interlayer spacing. Nature 550, 380–383 (2017).
Zhang, H. et al. Electrostatic-induced ion-confined partitioning in graphene nanolaminate membrane for breaking anion-cation co-transport to enhance desalination. Nat. Commun. 15, 4324 (2024).
Morelos-Gomez, A. et al. Effective NaCl and dye rejection of hybrid graphene oxide/graphene layered membranes. Nat. Nanotechnol. 12, 1083–1088 (2017).
Sapkota, B. et al. High permeability sub-nanometre sieve composite MoS2 membranes. Nat. Commun. 11, 2747 (2020).
Qian, Y. et al. Enhanced ion sieving of graphene oxide membranes via surface amine functionalization. J. Am. Chem. Soc. 143, 5080–5090 (2021).
Yuan, S., Li, Y., Xia, Y., Selomulya, C. & Zhang, X. Stable cation-controlled reduced graphene oxide membranes for improved NaCl rejection. J. Membr. Sci. 621, 118995 (2021).
Liu, H., Wang, H. & Zhang, X. Facile fabrication of freestanding ultrathin reduced graphene oxide membranes for water purification. Adv. Mater. 27, 249–254 (2015).
O’Hern, S. C. et al. Nanofiltration across defect-sealed nanoporous monolayer graphene. Nano Lett. 15, 3254–3260 (2015).
Yang, Y. et al. Large-area graphene-nanomesh/carbon-nanotube hybrid membranes for ionic and molecular nanofiltration. Science 364, 1057–1062 (2019).
Zhao, J. et al. Etching gas-sieving nanopores in single-layer graphene with an angstrom precision for high-performance gas mixture separation. Sci. Adv. 5, eaav1851 (2019).
Celebi, K. et al. Ultimate permeation across atomically thin porous graphene. Science 344, 289–292 (2014).
Zhou, Z. et al. Gas permeation through graphdiyne-based nanoporous membranes. Nat. Commun. 13, 4031 (2022).
Shen, J. et al. Fast water transport and molecular sieving through ultrathin ordered conjugated-polymer-framework membranes. Nat. Mater. 21, 1183–1190 (2022).
Liu, G. et al. Eliminating lattice defects in metal-organic framework molecular-sieving membranes. Nat. Mater. 22, 769–776 (2023).
Xu, J. Y. et al. Transparent proton transport through a two-dimensional nanomesh material. Nat. Commun. 10, 3971 (2019).
Li, G. et al. Architecture of graphdiyne nanoscale films. Chem. Commun. 46, 3256–3258, (2010).
Li, J. et al. Space-confined synthesis of monolayer graphdiyne in MXene interlayer. Adv. Mater. 36, 2308429 (2023).
Bartolomei, M. et al. Penetration barrier of water through graphynes’ pores: first-principles predictions and force field optimization. J. Phys. Chem. Lett. 5, 751–755 (2014).
Xu, J. Y. et al. Water transport through subnanopores in the ultimate size limit: mechanism from molecular dynamics. Nano Res. 12, 587–592 (2019).
Xu, J. Y. & Meng, S. Molecular transport across a two-dimensional nanomesh membrane–graphdiyne. J. Phys. D 53, 493003 (2020).
Chen, H. et al. Ultrahigh-water-flux desalination on graphdiyne membranes. Nat. Water 1, 800–807 (2023).
Zhou, J. et al. Synthesis of graphdiyne nanowalls using acetylenic coupling reaction. J. Am. Chem. Soc. 137, 7596–7599 (2015).
Li, J. et al. Synthesis of wafer-scale ultrathin graphdiyne for flexible optoelectronic memory with over 256 storage levels. Chem 7, 1284–1296 (2021).
Li, J. et al. Wafer-scale single-crystal monolayer graphene grown on sapphire substrate. Nat. Mater. 21, 740–747 (2022).
Chen, T. A. et al. Wafer-scale single-crystal hexagonal boron nitride monolayers on Cu (111). Nature 579, 219–223 (2020).
Tang, Y. Q., Yang, H. Y. & Yang, P. Investigation on the contact between graphdiyne and Cu (111) surface. Carbon 117, 246–251 (2017).
Gao, X. et al. Ultrathin graphdiyne film on graphene through solution phase Van der Waals epitaxy. Sci. Adv. 4, eaat6378 (2018).
Zhang, S. et al. Raman spectra and corresponding strain effects in graphyne and graphdiyne. J. Phys. Chem. C 120, 10605–10613 (2016).
Li, J. et al. Architecture of beta-graphdiyne-containing thin film using modified Glaser–Hay coupling reaction for enhanced photocatalytic property of TiO2. Adv. Mater. 29, 1700421 (2017).
Matsuoka, R. et al. Crystalline graphdiyne nanosheets produced at a gas/liquid or liquid/liquid interface. J. Am. Chem. Soc. 139, 3145–3152 (2017).
Zhong, J. et al. Electronic structure of graphdiyne probed by X-ray absorption spectroscopy and scanning transmission X-ray microscopy. J. Phys. Chem. C 117, 5931–5936 (2013).
Luo, G., Zheng, Q., Mei, W.-N., Lu, J. & Nagase, S. Structural, electronic, and optical properties of bulk graphdiyne. J. Phys. Chem. C 117, 13072–13079 (2013).
Zhou, J., Li, J., Liu, Z. & Zhang, J. Exploring approaches for the synthesis of few-layered graphdiyne. Adv. Mater. 31, 1803758 (2019).
Holt, J. K. et al. Fast mass transport through sub-2-nanometer carbon nanotubes. Science 312, 1034–1037 (2006).
Pang, J. et al. Exploring the sandwich antibacterial membranes based on UiO-66/graphene oxide for forward osmosis performance. Carbon 144, 321–332 (2019).
Baghbani, N. B., Azamat, J., Erfan-Niya, H., Majidi, S. & Khazini, L. Molecular insights into water desalination performance of pristine graphdiyne nanosheet membrane. J. Mol. Graph. Model. 101, 107729 (2020).
Ren, J. & McCutcheon, J. R. A new commercial thin film composite membrane for forward osmosis. Desalination 343, 187–193 (2014).
Chen, C. et al. Self‐assembled free‐standing graphite oxide membrane. Adv. Mater. 21, 3007–3011 (2009).
Li, W. et al. Ultrastrong MXene film induced by sequential bridging with liquid metal. Science 385, 62–68 (2024).
Hu, M. & Mi, B. Enabling graphene oxide nanosheets as water separation membranes. Environ. Sci. Technol. 47, 3715–3723 (2013).
Han, Y., Xu, Z. & Gao, C. Ultrathin graphene nanofiltration membrane for water purification. Adv. Funct. Mater. 23, 3693–3700 (2013).
Liu, X., Demir, N. K., Wu, Z. & Li, K. Highly water-stable zirconium metal–organic framework UiO-66 membranes supported on alumina hollow fibers for desalination. J. Am. Chem. Soc. 137, 6999–7002 (2015).
Zhang, Y. et al. Molecularly soldered covalent organic frameworks for ultrafast precision sieving. Sci. Adv. 7, eabe8706 (2021).
Schwegler, E., Grossman, J. C., Gygi, F. & Galli, G. Towards an assessment of the accuracy of density functional theory for first-principles simulations of water. J. Chem. Phys. 121, 5400–5409 (2004).
Bregante, D. T. et al. The shape of water in zeolites and its impact on epoxidation catalysis. Nat. Catal. 4, 797–808 (2021).
Uedaira, H. & Uedaira, H. Role of hydration of polyhydroxy compounds in biological systems. Cell. Mol. Biol. 47, 823–829 (2001).
Marx, D., Tuckerman, M. E., Hutter, J. & Parrinello, M. The nature of the hydrated excess proton in water. Nature 397, 601–604 (1999).
Tunuguntla, R. H., Allen, F. I., Kim, K., Belliveau, A. & Noy, A. Ultrafast proton transport in sub-1-nm diameter carbon nanotube porins. Nat. Nanotechnol. 11, 639–644 (2016).
Chen, M. et al. Hydroxide diffuses slower than hydronium in water because its solvated structure inhibits correlated proton transfer. Nat. Chem. 10, 413–419 (2018).
Acknowledgements
Financial support for this work was provided by Baseline Funds (BAS/1/1375-01-01 and FCC/1/5937-05-01 to Z.L.) from King Abdullah University of Science and Technology (KAUST). Y.H. acknowledges the support from the GJYC programme of Guangzhou City (2024D03J0001).
Author information
Authors and Affiliations
Contributions
Z.L., Y.H. and J.L. conceived and designed the experiments. J.L. and B.T. prepared the Cu (111)/Al2O3(0001) substrate. X.Z. provided important guidelines for Cu (111) substrate preparation. J.L., Q.L., X.L. and L.C. performed the synthesis of GDY materials, characterizations and transport measurements. K.Z. performed the ab initio molecular dynamics simulations and calculation of the potential of means force. H.C. performed the electronic measurements. G.L. performed the TEM measurement. All authors were involved in the analysis and discussion of the results. Z.L., Y.H., J.L., L.C. and Q.L. wrote the paper, and all authors commented on the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Water thanks Jin Zhang, Junyong Zhu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Methods, Figs. 1–53, Tables 1–6 and description of the videos.
Supplementary Video 1 (download MP4 )
A free-standing c-GDY film floating on the water indicates good mechanical strength as its macrostructure remains integrated despite external disturbances.
Supplementary Video 2 (download MP4 )
Water molecules moving in the channels to form regular 1D chains inside the membrane.
Supplementary Video 3 (download MP4 )
Density-functional-theory-calculated diffusion of proton in GDY membrane.
Supplementary Data 1
Chembiodraw structure of Fig. 1.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, J., Zhou, K., Liu, Q. et al. Synthesis of two-dimensional ordered graphdiyne membranes for highly efficient and selective water transport. Nat Water 3, 307–318 (2025). https://doi.org/10.1038/s44221-025-00397-9
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44221-025-00397-9
This article is cited by
-
Molecular dynamics insights into interfacial water accumulation for optimizing ceramic membrane-based air dehumidification
Building Simulation (2026)
-
High-strength conjugated polymer framework membranes for ultrafast and precise separations
Nature Sustainability (2025)
-
Energy-autonomous KCl extraction from brine via biomimetic covalent-organic-framework membranes and redox-driven ion transport
Nature Communications (2025)
-
Pre-pilot-scale porous graphene membrane for CO2 separation
Nature Chemical Engineering (2025)