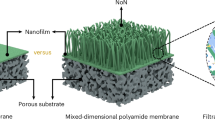
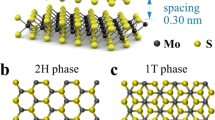
Abstract
Membranes with nanometre- and subnanometre-scale pores play a vital role in aqueous separations across applications ranging from desalination and wastewater reuse to resource recovery and green hydrogen production. Despite their widespread use, the molecular-level mechanisms that govern water and solute transport in these membranes remain inadequately understood. In this Perspective, we examine advances in membrane and nanochannel transport across macroscopic, microscopic and molecular scales to establish a unified mechanistic framework. We begin by analysing current macroscopic models, highlighting their simplifying assumptions and inherent limitations. We then explore insights from nano- and ångström-scale fluidic studies, revealing unconventional transport phenomena that are not captured by classical continuum theories. Next, we describe how molecular simulations offer atomistic resolution of transport processes, providing mechanistic insight into how water and ions traverse the dynamic, heterogeneous porous networks of real-world, state-of-the-art polymer membranes. Finally, we discuss how to integrate these molecular, microscopic and macroscopic scales to advance theoretical understanding and inform the rational design of next-generation membranes. We conclude by identifying key knowledge gaps and outlining emerging opportunities to bridge scales through advanced characterization techniques and multiscale modelling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Elimelech, M. & Phillip, W. A. The future of seawater desalination: energy, technology, and the environment. Science 333, 712–717 (2011).
Liu, W. et al. Pressure-driven membrane desalination. Nat. Rev. Methods Primers 4, 10 (2024).
Mohammad, A. W. et al. Nanofiltration membranes review: recent advances and future prospects. Desalination 356, 226–254 (2015).
Zhao, Y. et al. Differentiating solutes with precise nanofiltration for next generation environmental separations: a review. Environ. Sci. Technol. 55, 1359–1376 (2021).
Lu, X. & Elimelech, M. Fabrication of desalination membranes by interfacial polymerization: history, current efforts, and future directions. Chem. Soc. Rev. 50, 6290–6307 (2021).
Heiranian, M., Fan, H., Wang, L., Lu, X. & Elimelech, M. Mechanisms and models for water transport in reverse osmosis membranes: history, critical assessment, and recent developments. Chem. Soc. Rev. 52, 8455–8480 (2023).
Wang, R., He, R., He, T., Elimelech, M. & Lin, S. Performance metrics for nanofiltration-based selective separation for resource extraction and recovery. Nat. Water 1, 291–300 (2023).
Kononenko, N. et al. Porous structure of ion exchange membranes investigated by various techniques. Adv. Colloid Interface Sci. 246, 196–216 (2017).
Li, X., Zuo, P., Ge, X., Yang, Z. & Xu, T. Constructing new-generation ion exchange membranes under confinement regime. Natl Sci. Rev. https://doi.org/10.1093/nsr/nwae439 (2024).
Du, N. et al. Anion-exchange membrane water electrolyzers. Chem. Rev. 122, 11830–11895 (2022).
Park, E. J., Arges, C. G., Xu, H. & Kim, Y. S. Membrane strategies for water electrolysis. ACS Energy Lett. 7, 3447–3457 (2022).
Paidar, M., Fateev, V. & Bouzek, K. Membrane electrolysis—history, current status and perspective. Electrochim. Acta 209, 737–756 (2016).
Wang, L. et al. Water transport in reverse osmosis membranes is governed by pore flow, not a solution-diffusion mechanism. Sci. Adv. 9, eadf8488 (2023).
Fan, H., Heiranian, M. & Elimelech, M. The solution-diffusion model for water transport in reverse osmosis: what went wrong? Desalination 580, 117575 (2024).
Wang, L. et al. Salt and water transport in reverse osmosis membranes: beyond the solution-diffusion model. Environ. Sci. Technol. 55, 16665–16675 (2021).
Biesheuvel, P. M., Porada, S., Elimelech, M. & Dykstra, J. E. Tutorial review of reverse osmosis and electrodialysis. J. Membr. Sci. 647, 120221 (2022).
Kavokine, N., Netz, R. R. & Bocquet, L. Fluids at the nanoscale: from continuum to subcontinuum transport. Annu. Rev. Fluid Mech. 53, 377–410 (2021).
Aluru, N. R. et al. Fluids and electrolytes under confinement in single-digit nanopores. Chem. Rev. 123, 2737–2831 (2023).
Emmerich, T. et al. Nanofluidics. Nat. Rev. Methods Primers 4, 69 (2024).
Fujioka, T., Oshima, N., Suzuki, R., Price, W. E. & Nghiem, L. D. Probing the internal structure of reverse osmosis membranes by positron annihilation spectroscopy: gaining more insight into the transport of water and small solutes. J. Membr. Sci. 486, 106–118 (2015).
Pipich, V. et al. Morphology and porous structure of standalone aromatic polyamide films as used in RO membranes – an exploration with SANS, PALS, and SEM. J. Membr. Sci. 573, 167–176 (2019).
Heiranian, M., DuChanois, R. M., Ritt, C. L., Violet, C. & Elimelech, M. Molecular simulations to elucidate transport phenomena in polymeric membranes. Environ. Sci. Technol. 56, 3313–3323 (2022).
Mahajan, S., Hoek, E. M. V., McCutcheon, J. R. & Li, Y. Benchmarking forcefields for molecular dynamics simulations of polyamide-based reverse-osmosis membranes. J. Membr. Sci. 716, 123493 (2025).
Shen, M., Keten, S. & Lueptow, R. M. Dynamics of water and solute transport in polymeric reverse osmosis membranes via molecular dynamics simulations. J. Membr. Sci. 506, 95–108 (2016).
Ding, M., Ghoufi, A. & Szymczyk, A. Molecular simulations of polyamide reverse osmosis membranes. Desalination 343, 48–53 (2014).
Biesheuvel, P. M. et al. Ion selectivity in brackish water desalination by reverse osmosis: theory, measurements, and implications. Environ. Sci. Technol. Lett. 7, 42–47 (2020).
Wang, R. & Lin, S. Membrane design principles for ion-selective electrodialysis: an analysis for Li/Mg separation. Environ. Sci. Technol. 58, 3552–3563 (2024).
Wang, Z., Deshmukh, A., Du, Y. & Elimelech, M. Minimal and zero liquid discharge with reverse osmosis using low-salt-rejection membranes. Water Res. 170, 115317 (2020).
Du, Y., Wang, Z., Cooper, N. J., Gilron, J. & Elimelech, M. Module-scale analysis of low-salt-rejection reverse osmosis: design guidelines and system performance. Water Res. 209, 117936 (2022).
Atia, A. A., Allen, J., Young, E., Knueven, B. & Bartholomew, T. V. Cost optimization of low-salt-rejection reverse osmosis. Desalination 551, 116407 (2023).
Ritt, C. L. et al. The open membrane database: synthesis–structure–performance relationships of reverse osmosis membranes. J. Membr. Sci. 641, 119927 (2022).
Biesheuvel, P. M., Rutten, S. B., Ryzhkov, I. I., Porada, S. & Elimelech, M. Theory for salt transport in charged reverse osmosis membranes: novel analytical equations for desalination performance and experimental validation. Desalination 557, 116580 (2023).
Long, L. et al. Assessment of permeance and selectivity of thin-film composite polyamide membranes for diverse applications. Nat. Water 3, 668–682 (2025).
Wijmans, J. G. & Baker, R. W. The solution-diffusion model: a review. J. Membr. Sci. 107, 1–21 (1995).
Fan, H. et al. The physical basis for solvent flow in organic solvent nanofiltration. Sci. Adv. 10, eado4332 (2024).
He, J., Fan, H., Elimelech, M. & Li, Y. Molecular simulations of organic solvent transport in dense polymer membranes: solution-diffusion or pore-flow mechanism? J. Membr. Sci. 708, 123055 (2024).
Bowen, W. R. & Welfoot, J. S. Modelling the performance of membrane nanofiltration—critical assessment and model development. Chem. Eng. Sci. 57, 1121–1137 (2002).
Wang, R. & Lin, S. Pore model for nanofiltration: history, theoretical framework, key predictions, limitations, and prospects. J. Membr. Sci. 620, 118809 (2021).
Oren, Y. S. & Biesheuvel, P. M. Theory of ion and water transport in reverse-osmosis membranes. Phys. Rev. Appl. 9, 024034 (2018).
Park, H. B., Kamcev, J., Robeson, L. M., Elimelech, M. & Freeman, B. D. Maximizing the right stuff: the trade-off between membrane permeability and selectivity. Science 356, eaab0530 (2017).
Tsuru, T., Urairi, M., Nakao, S.-I. & Kimura, S. Negative rejection of anions in the loose reverse osmosis separation of mono- and divalent ion mixtures. Desalination 81, 219–227 (1991).
Gilron, J., Gara, N. & Kedem, O. Experimental analysis of negative salt rejection in nanofiltration membranes. J. Membr. Sci. 185, 223–236 (2001).
Yaroshchuk, A. E. Negative rejection of ions in pressure-driven membrane processes. Adv. Colloid Interface Sci. 139, 150–173 (2008).
Yaroshchuk, A., Bruening, M. L. & Licón Bernal, E. E. Solution-diffusion–electro-migration model and its uses for analysis of nanofiltration, pressure-retarded osmosis and forward osmosis in multi-ionic solutions. J. Membr. Sci. 447, 463–476 (2013).
Bandini, S. & Vezzani, D. Nanofiltration modeling: the role of dielectric exclusion in membrane characterization. Chem. Eng. Sci. 58, 3303–3326 (2003).
Szymczyk, A. & Fievet, P. Investigating transport properties of nanofiltration membranes by means of a steric, electric and dielectric exclusion model. J. Membr. Sci. 252, 77–88 (2005).
Wang, L. et al. Significance of co-ion partitioning in salt transport through polyamide reverse osmosis membranes. Environ. Sci. Technol. 57, 3930–3939 (2023).
Cao, T., Wang, L., Pataroque, K. E., Wang, R. & Elimelech, M. Relating solute–membrane electrostatic interactions to solute permeability in reverse osmosis membranes. Environ. Sci. Technol. 59, 5819–5828 (2025).
Wang, R. & Elimelech, M. Revisiting solute transport in polyamide membranes: insights from neutral solute partitioning. J. Membr. Sci. 728, 124117 (2025).
Majumder, M., Chopra, N., Andrews, R. & Hinds, B. J. Nanoscale hydrodynamics: enhanced flow in carbon nanotubes. Nature 438, 44–44 (2005).
Secchi, E. et al. Massive radius-dependent flow slippage in carbon nanotubes. Nature 537, 210–213 (2016).
Tunuguntla, R. et al. Enhanced water permeability and tunable ion selectivity in sub-nanometer carbon nanotube porins. Science 357, 792–796 (2017).
Radha, B. et al. Molecular transport through capillaries made with atomic-scale precision. Nature 538, 222–225 (2016).
Licsandru, E. et al. Salt-excluding artificial water channels exhibiting enhanced dipolar water and proton translocation. J. Am. Chem. Soc. 138, 5403–5409 (2016).
Murail, S. et al. Water permeation across artificial I-quartet membrane channels: from structure to disorder. Faraday Discuss. 209, 125–148 (2018).
Huang, L.-B. et al. Hydroxy channels–adaptive pathways for selective water cluster permeation. J. Am. Chem. Soc. 143, 4224–4233 (2021).
Fumagalli, L. et al. Anomalously low dielectric constant of confined water. Science 360, 1339–1342 (2018).
Kavokine, N., Bocquet, M.-L. & Bocquet, L. Fluctuation-induced quantum friction in nanoscale water flows. Nature 602, 84–90 (2022).
Di Vincenzo, M. et al. Biomimetic artificial water channel membranes for enhanced desalination. Nat. Nanotechnol. 16, 190–196 (2021).
Di Vincenzo, M. et al. Tunable membranes incorporating artificial water channels for high-performance brackish/low-salinity water reverse osmosis desalination. Proc. Natl Acad. Sci. USA 118, e2022200118 (2021).
Kocsis, I. et al. Oriented chiral water wires in artificial transmembrane channels. Sci. Adv. 4, eaao5603 (2018).
Huang, L.-B. et al. Water-pore flow permeation through multivalent H-bonding pyridine-2, 6-dicarboxamide-histamine/histidine water channels. J. Am. Chem. Soc. 147, 678–686 (2024).
Mouterde, T. et al. Molecular streaming and its voltage control in ångström-scale channels. Nature 567, 87–90 (2019).
Li, Z. et al. Breakdown of Nernst–Einstein relation in carbon nanotube porins. Nat. Nanotechnol. 18, 177–183 (2023).
Li, Y. et al. Water-ion permselectivity of narrow-diameter carbon nanotubes. Sci. Adv. 6, eaba9966 (2020).
Murata, K. et al. Structural determinants of water permeation through aquaporin-1. Nature 407, 599–605 (2000).
Tunuguntla, R., Allen, F., Kim, K., Bellivieau, A. & Noy, A. Ultrafast proton transport in sub-1-nm diameter carbon nanotube porins. Nat. Nanotechnol. 11, 639–644 (2016).
Andrei, I. M., Chen, W., Baaden, M., Vincent, S. P. & Barboiu, M. Proton-versus cation-selective transport of saccharide rim-appended pillar [5] arene artificial water channels. J. Am. Chem. Soc. 145, 21904–21914 (2023).
Gopinadhan, K. et al. Complete steric exclusion of ions and proton transport through confined monolayer water. Science 363, 145–148 (2019).
Esfandiar, A. et al. Size effect in ion transport through angstrom-scale slits. Science 358, 511–513 (2017).
Abdelghani-Idrissi, S. et al. Resonant osmotic diodes for voltage-induced water filtration across composite membranes. Nat. Mater. 24, 1109–1115 (2025).
Li, Y. et al. Molecular transport enhancement in pure metallic carbon nanotube porins. Nat. Mater. 23, 1123–1130 (2024).
Dischinger, S. M., Miller, D. J., Vermaas, D. A. & Kingsbury, R. S. Unifying the conversation: membrane separation performance in energy, water, and industrial applications. ACS ES&T Eng. 4, 277–289 (2024).
Coquinot, B. et al. Momentum tunnelling between nanoscale liquid flows. Nat. Nanotechnol. 20, 397–403 (2025).
Alkhadra, M. A., Gao, T., Conforti, K. M., Tian, H. & Bazant, M. Z. Small-scale desalination of seawater by shock electrodialysis. Desalination 476, 114219 (2020).
Kim, S. J., Ko, S. H., Kang, K. H. & Han, J. Direct seawater desalination by ion concentration polarization. Nat. Nanotechnol. 5, 297–301 (2010).
Liu, G., Jin, W. & Xu, N. Two-dimensional-material membranes: a new family of high-performance separation membranes. Angew. Chem. Int. Ed. 55, 13384–13397 (2016).
Pagliero, D. et al. Slow water in engineered nanochannels revealed by color-center-enabled sensing. Nano Lett. 25, 9960–9966 (2025).
Ridgway, H. F., Orbell, J. & Gray, S. Molecular simulations of polyamide membrane materials used in desalination and water reuse applications: recent developments and future prospects. J. Membr. Sci. 524, 436–448 (2017).
Ghoufi, A., Dražević, E. & Szymczyk, A. Interactions of organics within hydrated selective layer of reverse osmosis desalination membrane: a combined experimental and computational study. Environ. Sci. Technol. 51, 2714–2719 (2017).
He, J., Yang, J., McCutcheon, J. R. & Li, Y. Molecular insights into the structure-property relationships of 3D printed polyamide reverse-osmosis membrane for desalination. J. Membr. Sci. 658, 120731 (2022).
Wu, J. et al. Polyamide reverse osmosis membrane compaction and relaxation: mechanisms and implications for desalination performance. J. Membr. Sci. 706, 122893 (2024).
Shefer, I., Lopez, K., Straub, A. P. & Epsztein, R. Applying transition-state theory to explore transport and selectivity in salt-rejecting membranes: a critical review. Environ. Sci. Technol. 56, 7467–7483 (2022).
Richards, L. A., Schäfer, A. I., Richards, B. S. & Corry, B. The importance of dehydration in determining ion transport in narrow pores. Small 8, 1701–1709 (2012).
Kingsbury, R. S. et al. Kinetic barrier networks reveal rate limitations in ion-selective membranes. Matter 7, 2161–2183 (2024).
Lu, C. et al. Dehydration-enhanced ion-pore interactions dominate anion transport and selectivity in nanochannels. Sci. Adv. 9, eadf8412 (2023).
Wang, A. et al. Selective ion transport through hydrated micropores in polymer membranes. Nature 635, 353–358 (2024).
Yang, D. et al. Solution-processable polymer membranes with hydrophilic subnanometre pores for sustainable lithium extraction. Nat. Water, 3, 319–333 (2025).
Liu, S. et al. Lithium-ion extraction using electro-driven freestanding graphene oxide composite membranes. J. Membr. Sci. 672, 121448 (2023).
Warnock, S. J. et al. Engineering Li/Na selectivity in 12-Crown-4–functionalized polymer membranes. Proc. Natl Acad. Sci. USA 118, e2022197118 (2021).
Guo, Y. et al. Deciphering co-ion and counterion transport in polyamide desalination membranes reveals ion selectivity mechanisms. Sci. Adv. 11, eadu8302 (2025).
Scheiderer, L., Marin, Z. & Ries, J. MINFLUX achieves molecular resolution with minimal photons. Nat. Photon. 19, 238–247 (2025).
Zewail, A. H. 4D ultrafast electron diffraction, crystallography, and microscopy. Annu. Rev. Phys. Chem. 57, 65–103 (2006).
King, W. E. et al. Ultrafast electron microscopy in materials science, biology, and chemistry. J. Appl. Phy. 97, 111101 (2005).
Freed, J. H. New technologies in electron spin resonance. Annu. Rev. Phys. Chem. 51, 655–689 (2000).
Borbat, P., Costa-Filho, A., Earle, K., Moscicki, J. & Freed, J. Electron spin resonance in studies of membranes and proteins. Science 291, 266–269 (2001).
Moerner, W., Shechtman, Y. & Wang, Q. Single-molecule spectroscopy and imaging over the decades. Faraday Discuss. 184, 9–36 (2015).
Agrawal, K. V., Shimizu, S., Drahushuk, L. W., Kilcoyne, D. & Strano, M. S. Observation of extreme phase transition temperatures of water confined inside isolated carbon nanotubes. Nat. Nanotechnol. 12, 267–273 (2017).
Ishiyama, T., Imamura, T. & Morita, A. Theoretical studies of structures and vibrational sum frequency generation spectra at aqueous interfaces. Chem. Rev. 114, 8447–8470 (2014).
Chen, Z., Shen, Y. & Somorjai, G. A. Studies of polymer surfaces by sum frequency generation vibrational spectroscopy. Annu. Rev. Phys. Chem. 53, 437–465 (2002).
Kim, S. H., Kwak, S.-Y. & Suzuki, T. Positron annihilation spectroscopic evidence to demonstrate the flux-enhancement mechanism in morphology-controlled thin-film-composite (TFC) membrane. Environ. Sci. Technol. 39, 1764–1770 (2005).
Pipich, V. et al. The porous system of a reverse osmosis membrane − operando studies with small-angle neutron scattering. Chem. Eng. J. 495, 153304 (2024).
Fu, Q. et al. Molecular structure of aromatic reverse osmosis polyamide barrier layers. ACS Macro Lett. 8, 352–356 (2019).
Zhang, W., Xu, S., Han, X. & Bao, X. In situ solid-state NMR for heterogeneous catalysis: a joint experimental and theoretical approach. Chem. Soc. Rev. 41, 192–210 (2012).
Yao, X. et al. Nondissociative activated dihydrogen binding on CeO2 revealed by high-pressure operando solid-state NMR spectroscopy. J. Am. Chem. Soc. 146, 24609–24618 (2024).
Rehman, D. & Lienhard, J. H. Physics-informed deep learning for multi-species membrane separations. Chem. Eng. J. 485, 149806 (2024).
Unke, O. T. et al. Machine learning force fields. Chem. Rev. 121, 10142–10186 (2021).
Ingólfsson, H. I. et al. The power of coarse graining in biomolecular simulations. WIREs Comput. Mol. Sci. 4, 225–248 (2014).
Wang, J. et al. Machine learning of coarse-grained molecular dynamics force fields. ACS Central Sci. 5, 755–767 (2019).
Ye, H., Shen, Z. & Li, Y. Computational modeling of magnetic particle margination within blood flow through LAMMPS. Comput. Mech. 62, 457–476 (2018).
Islam, K., Razizadeh, M. & Liu, Y. Coarse-grained molecular simulation of extracellular vesicle squeezing for drug loading. Phys. Chem. Chem. Phys. 25, 12308–12321 (2023).
Fan, H. & Elimelech, M. Solvent transport in disordered and dynamic membrane pores: implications for reverse osmosis and nanofiltration membranes. Environ. Sci. Technol. 59, 17922–17931 (2025).
Hughes, Z. E. & Gale, J. D. A computational investigation of the properties of a reverse osmosis membrane. J. Mater. Chem. 20, 7788–7799 (2010).
Boya, R., Keerthi, A. & Parambath, M. S. The wonderland of angstrofluidics. Phys. Today 77, 26–33 (2024).
You, Y. et al. Angstrofluidics: walking to the limit. Annu. Rev. Mater. Res. 52, 189–218 (2022).
Zhou, X. et al. Intrapore energy barriers govern ion transport and selectivity of desalination membranes. Sci. Adv. 6, eabd9045 (2020).
Goutham, S. et al. Beyond steric selectivity of ions using ångström-scale capillaries. Nat. Nanotechnol. 18, 596–601 (2023).
Li, Z. et al. Strong differential monovalent anion selectivity in narrow diameter carbon nanotube porins. ACS Nano 14, 6269–6275 (2020).
Keerthi, A. et al. Water friction in nanofluidic channels made from two-dimensional crystals. Nat. Commun. 12, 3092 (2021).
Dror, R. O., Dirks, R. M., Grossman, J., Xu, H. & Shaw, D. E. Biomolecular simulation: a computational microscope for molecular biology. Annu. Rev. Biophys. 41, 429–452 (2012).
Liu, S., Foo, Z., Lienhard, J. H., Keten, S. & Lueptow, R. M. Membrane charge effects on solute transport in nanofiltration: experiments and molecular dynamics simulations. Membranes 15, 184 (2025).
Lu, D. et al. Impact of charge homogeneity on ion selectivity in polyamide membranes. Nat. Water 3, 978–991 (2025).
Kawada, R., Endo, K., Yasuoka, K., Kojima, H. & Matubayasi, N. Prediction of water diffusion in wide varieties of polymers with all-atom molecular dynamics simulations and deep generative models. J. Chem. Inf. Model. 63, 76–86 (2022).
Gao, H. et al. Revolutionizing membrane design using machine learning-bayesian optimization. Environ. Sci. Technol. 56, 2572–2581 (2021).
Giro, R. et al. AI powered, automated discovery of polymer membranes for carbon capture. npj Comput. Mater. 9, 133 (2023).
Yang, J., Tao, L., He, J., McCutcheon, J. R. & Li, Y. Machine learning enables interpretable discovery of innovative polymers for gas separation membranes. Sci. Adv. 8, eabn9545 (2022).
Mahajan, S. & Li, Y. Toward molecular simulation guided design of next-generation membranes: challenges and opportunities. Langmuir 41, 12388–12402 (2025).
Cao, Z., Barati Farimani, O., Ock, J. & Barati Farimani, A. Machine learning in membrane design: from property prediction to AI-guided optimization. Nano Lett. 24, 2953–2960 (2024).
Dangayach, R. et al. Machine learning-aided inverse design and discovery of novel polymeric materials for membrane separation. Environ. Sci. Technol. 59, 993–1012 (2024).
Acknowledgements
This Perspective is based on discussions from the ‘Interdisciplinary Perspectives on Transport Mechanisms in Membranes and Nanopores’ workshop held at the Rice Global Paris Center on 17–18 March 2025. We gratefully acknowledge support from the Rice WaTER Institute and the Rice Center for Membrane Excellence (RiCeME) at Rice University, which provided funding to attend the symposium.
Author information
Authors and Affiliations
Contributions
All authors contributed to the discussions, drafting and analysis of the Perspective. M.E. and H.F. edited the final version of the manuscript. M.P. and H.F. prepared the illustrations.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Water thanks Kecheng Guan, Wooyoung Shim and Liping Wen for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fan, H., Parkinson, M., Agrawal, K.V. et al. A multiscale perspective for understanding transport mechanisms in desalination and ion-selective membranes. Nat Water 4, 120–137 (2026). https://doi.org/10.1038/s44221-026-00585-1
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44221-026-00585-1