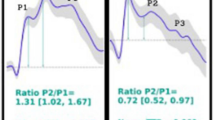
Abstract
Continuous neuromonitoring is essential in neurocritical care units, providing real-time insights into dynamic cerebral physiology for patients with neurological conditions, such as stroke and neurotrauma. Multiple modalities such as intracranial pressure, arterial blood pressure and near-infrared spectroscopy enable high-resolution dynamic tracking of cerebral perfusion pressure, and related variables critical for life-saving decisions. However, inherent, commonly unavoidable artefacts obscure insights into patient states and complicate treatment decisions. In this Review, we explore primary sources of artefacts in continuous neuromonitoring modalities, including clinical procedure activities, patient-related physiology, technical equipment properties and environmental factors, and their impacts on data integrity and clinical implications. We discuss emerging artefact management strategies, including domain knowledge and data-driven methods to mitigate impact and enhance data reliability. Additionally, we identify key translational challenges, indications for neurosensor design, harmonization and future artificial intelligence pathways, highlighting the need for robust, automated, real-time artefact management to enable precise, individualized patient care.
Key points
-
Continuous neuromonitoring integrates invasive and non-invasive high-resolution techniques for real-time multimodal assessment of neural function.
-
Continuous neuromonitoring data inherently contain artefacts arising from clinical procedures, patient physiology, technical limitations and environmental factors.
-
Artefacts can cumulatively compound and interactively interfere with clinical interpretation and, potentially, diagnosis and treatment decisions, and prognosis.
-
Effective artefact management requires close clinical collaboration, robust domain knowledge and data-driven methods.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Zoerle, T. et al. Intracranial pressure monitoring in adult patients with traumatic brain injury: challenges and innovations. Lancet Neurol. 23, 938–950 (2024).
Claassen, J. & Park, S. Spontaneous subarachnoid haemorrhage. Lancet 400, 846–862 (2022).
van den Bent, M. J. et al. Primary brain tumours in adults. Lancet 402, 1564–1579 (2023).
Maas, A. I. et al. Traumatic brain injury: progress and challenges in prevention, clinical care, and research. Lancet Neurol. 21, 1004–1060 (2022).
Mirza, A. B. et al. Preoperative and intraoperative neuromonitoring and mapping techniques impact oncological and functional outcomes in supratentorial function-eloquent brain tumours: a systematic review and meta-analysis. eClinicalMedicine 80, 103055 (2025).
Hemphill, J. C., Andrews, P. & De Georgia, M. Multimodal monitoring and neurocritical care bioinformatics. Nat. Rev. Neurol. 7, 451–460 (2011).
Foreman, B. et al. Practice standards for the use of multimodality neuromonitoring: a Delphi consensus process. Crit. Care Med. 51, 1740 (2023).
Robba, C. et al. Intracranial pressure monitoring in patients with acute brain injury in the intensive care unit (SYNAPSE-ICU): an international, prospective observational cohort study. Lancet Neurol. 20, 548–558 (2021).
Stocchetti, N. et al. Clinical review: neuromonitoring—an update. Crit. Care 17, 201 (2013).
Liang, W. et al. Advances, challenges and opportunities in creating data for trustworthy AI. Nat. Mach. Intell. 4, 669–677 (2022).
Foreman, B., Lissak, I. A., Kamireddi, N., Moberg, D. & Rosenthal, E. S. Challenges and opportunities in multimodal monitoring and data analytics in traumatic brain injury. Curr. Neurol. Neurosci. Rep. 21, 6 (2021).
Marina, N. et al. Astrocytes monitor cerebral perfusion and control systemic circulation to maintain brain blood flow. Nat. Commun. 11, 131 (2020).
Grubb, S. et al. Precapillary sphincters maintain perfusion in the cerebral cortex. Nat. Commun. 11, 395 (2020).
Zhang, Z., Hwang, M., Kilbaugh, T. J., Sridharan, A. & Katz, J. Cerebral microcirculation mapped by echo particle tracking velocimetry quantifies the intracranial pressure and detects ischemia. Nat. Commun. 13, 666 (2022).
Menon, D. K. & Ercole, A. in Handbook of Clinical Neurology Vol. 140 (eds Wijdicks, E. F. M. & Kramer, A. H.) Ch. 14 (Elsevier, 2017).
McGhee, B. & Bridges, E. Monitoring arterial blood pressure: what you may not know. Crit. Care Nurse 22, 60–64, 66–70, 73 passim (2002).
Evensen, K. B. & Eide, P. K. Measuring intracranial pressure by invasive, less invasive or non-invasive means: limitations and avenues for improvement. Fluids Barriers CNS 17, 34 (2020).
Hawryluk, G. W. J. et al. Intracranial pressure: current perspectives on physiology and monitoring. Intensive Care Med. 48, 1471–1481 (2022).
Liang, X.-Z., Zhang, Y. & Lunte, C. E. Monitoring oxygen partial pressure in tissue using microdialysis sampling and membrane coated oxygen sensors. J. Pharm. Biomed. Anal. 16, 1143–1152 (1998).
Cifrek, M. & Jelić, L. in Inspection of Medical Devices: For Regulatory Purposes (eds Badnjević, A. et al.) 241–269 (Springer Nature, 2024).
Brash, J. L., Horbett, T. A., Latour, R. A. & Tengvall, P. The blood compatibility challenge. Part 2: protein adsorption phenomena governing blood reactivity. Acta Biomater. 94, 11–24 (2019).
Gurbel, P. A. et al. Platelet reactivity in patients and recurrent events post-stenting. JACC 46, 1820–1826 (2005).
Papadopoulou, V., Tang, M.-X., Balestra, C., Eckersley, R. J. & Karapantsios, T. D. Circulatory bubble dynamics: from physical to biological aspects. Adv. Colloid Interface Sci. 206, 239–249 (2014).
Yin, J., Wang, S., Tat, T. & Chen, J. Motion artefact management for soft bioelectronics. Nat. Rev. Bioeng. 2, 541–558 (2024).
Wang, Y. et al. Skin bioelectronics towards long-term, continuous health monitoring. Chem. Soc. Rev. 51, 3759–3793 (2022).
Fantini, S. et al. Non-invasive optical monitoring of the newborn piglet brain using continuous-wave and frequency-domain spectroscopy. Phys. Med. Biol. 44, 1543–1563 (1999).
Hepburn-Smith, M. et al. Establishment of an external ventricular drain best practice guideline: the quest for a comprehensive, universal standard for external ventricular drain care. J. Neurosci. Nurs. 48, 54 (2016).
Li, J., Li, Z. & Wu, X. The practice and facilitators of, and barriers to, implementing individualized repositioning frequency: a national cross-sectional survey of critical care nurses. J. Tissue Viability 33, 89–95 (2024).
Blakeman, T. C., Scott, J. B., Yoder, M. A., Capellari, E. & Strickland, S. L. AARC clinical practice guidelines: artificial airway suctioning. Respir. Care 67, 258–271 (2022).
Hickey, J. V., Olson, D. M. & Turner, D. A. Intracranial pressure waveform analysis during rest and suctioning. Biol. Res. Nurs. 11, 174–186 (2009).
Kirkness, C. J., Mitchell, P. H., Burr, R. L., March, K. S. & Newell, D. W. Intracranial pressure waveform analysis: clinical and research implications. J. Neurosci. Nurs. 32, 271 (2000).
Khan, J. M., Maslove, D. M. & Boyd, J. G. Optimized arterial line artifact identification algorithm cleans high-frequency arterial line data with high accuracy in critically Ill patients. Crit. Care Explor. 4, e0814 (2022).
Romagnoli, S. et al. Accuracy of invasive arterial pressure monitoring in cardiovascular patients: an observational study. Crit. Care Lond. Engl. 18, 644 (2014).
Li, Q., Mark, R. G. & Clifford, G. D. Artificial arterial blood pressure artifact models and an evaluation of a robust blood pressure and heart rate estimator. Biomed. Eng. Online 8, 13 (2009).
Howells, T., Johnson, U., McKelvey, T., Ronne-Engström, E. & Enblad, P. The effects of ventricular drainage on the intracranial pressure signal and the pressure reactivity index. J. Clin. Monit. Comput. 31, 469–478 (2017).
Nag, D. S., Sahu, S., Swain, A. & Kant, S. Intracranial pressure monitoring: gold standard and recent innovations. World J. Clin. Cases 7, 1535–1553 (2019).
Liu, X. et al. Intracranial pressure monitoring via external ventricular drain: are we waiting long enough before recording the real value? J. Neurosci. Nurs. 52, 37 (2020).
Teichmann, D., Lynch, J. C. & Heldt, T. Distortion of the intracranial pressure waveform by extraventricular drainage system. IEEE Trans. Biomed. Eng. 68, 1646–1657 (2021).
Hornberger, C. & Wabnitz, H. Approaches for calibration and validation of near-infrared optical methods for oxygenation monitoring. Biomed. Eng. Biomed. Tech. 63, 537–546 (2018).
Agelet, L. E. & Hurburgh, C. R. A tutorial on near infrared spectroscopy and its calibration. Crit. Rev. Anal. Chem. 40, 246–260 (2010).
Hemker, H. C. & Kahn, M. J. P. Reaction sequence of blood coagulation. Nature 215, 1201–1202 (1967).
Saugel, B., Kouz, K., Meidert, A. S., Schulte-Uentrop, L. & Romagnoli, S. How to measure blood pressure using an arterial catheter: a systematic 5-step approach. Crit. Care Lond. Engl. 24, 172 (2020).
Conen, D. et al. Age-specific differences between conventional and ambulatory daytime blood pressure values. Hypertension 64, 1073–1079 (2014).
Zhang, F. et al. Investigation of artifacts and optimization in proton resonance frequency thermometry towards heating risk monitoring of implantable medical devices in magnetic resonance imaging. IEEE Trans. Biomed. Eng. 68, 3638–3646 (2021).
Stewart, C. et al. The new Licox combined brain tissue oxygen and brain temperature monitor: assessment of in vitro accuracy and clinical experience in severe traumatic brain injury. Neurosurgery 63, 1159 (2008).
Li, Z., Li, X. & Li, T. Noninvasive monitoring local variations of fever and edema on human: potential for point-of-care inflammation assessment. In Proc. Light-Based Diagnosis and Treatment of Infectious Diseases Vol. 10479, 21–27 (Curran, 2018).
Mader, M. M. et al. Evaluation of a new multiparameter brain probe for simultaneous measurement of brain tissue oxygenation, cerebral blood flow, intracranial pressure, and brain temperature in a porcine model. Neurocrit. Care 29, 291–301 (2018).
Spencer, G. S., Smith, J. A., Chowdhury, M. E. H., Bowtell, R. & Mullinger, K. J. Exploring the origins of EEG motion artefacts during simultaneous fMRI acquisition: implications for motion artefact correction. NeuroImage 173, 188–198 (2018).
Mauritzon, S., Ginstman, F., Hillman, J. & Wårdell, K. Analysis of laser Doppler flowmetry long-term recordings for investigation of cerebral microcirculation during neurointensive care. Front. Neurosci. 16, 1030805 (2022).
Aquilina, K. et al. Preliminary evaluation of a novel intraparenchymal capacitive intracranial pressure monitor: laboratory investigation. J. Neurosurg. 115, 561–569 (2011).
Kirkman, M. A. & Smith, M. Intracranial pressure monitoring, cerebral perfusion pressure estimation, and ICP/CPP-guided therapy: a standard of care or optional extra after brain injury? BJA Br. J. Anaesth. 112, 35–46 (2014).
Hoelper, B. M., Alessandri, B., Heimann, A., Behr, R. & Kempski, O. Brain oxygen monitoring: in-vitro accuracy, long-term drift and response-time of Licox- and Neurotrend sensors. Acta Neurochir. 147, 767–774 (2005).
Strangman, G. E., Ivkovic, V. & Zhang, Q. Wearable brain imaging with multimodal physiological monitoring. J. Appl. Physiol. 124, 564–572 (2018).
Ferree, T. C., Luu, P., Russell, G. S. & Tucker, D. M. Scalp electrode impedance, infection risk, and EEG data quality. Clin. Neurophysiol. 112, 536–544 (2001).
Thomas, R., Shin, S. S. & Balu, R. Applications of near-infrared spectroscopy in neurocritical care. Neurophotonics 10, 023522 (2023).
Czosnyka, M. Monitoring and interpretation of intracranial pressure. J. Neurol. Neurosurg. Psychiatry 75, 813–821 (2004).
Brasil, S. et al. Intracranial compliance assessed by intracranial pressure pulse waveform. Brain Sci. 11, 971 (2021).
Sykora, M. et al. Heart rate entropy is associated with mortality after intracereberal hemorrhage. J. Neurol. Sci. 418, 117033 (2020).
Mahdi, A., Rutter, E. M. & Payne, S. J. Effects of non-physiological blood pressure artefacts on cerebral autoregulation. Med. Eng. Phys. 47, 218–221 (2017).
Dai, H., Jia, X., Pahren, L., Lee, J. & Foreman, B. Intracranial pressure monitoring signals after traumatic brain injury: a narrative overview and conceptual data science framework. Front. Neurol. 11, 959 (2020).
Feng, M., Loy, L. Y., Zhang, F. & Guan, C. Artifact removal for intracranial pressure monitoring signals: a robust solution with signal decomposition. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2011, 797–801 (2011).
Czosnyka, M. & Czosnyka, Z. Origin of intracranial pressure pulse waveform. Acta Neurochir. 162, 1815–1817 (2020).
Wang, S.-C. et al. Arterial blood pressure waveform in liver transplant surgery possesses variability of morphology reflecting recipients’ acuity and predicting short term outcomes. J. Clin. Monit. Comput. 37, 1521–1531 (2023).
Byrd-Raynor, S. & Parker, K. New transcranial Doppler (TCD) waveform shape parameters: a repeatability/reproducibility study. J. Vasc. Ultrasound 31, 193–205 (2007).
Gersten, A., Perle, J., Heimer, D., Raz, A. & Fried, R. in Infrared Spectroscopy: Life and Biomedical Sciences (ed. Theophile, T.) Ch. 8 (2012).
Bergmann, T. et al. Artifact management for cerebral near-infrared spectroscopy signals: a systematic scoping review. Bioengineering 11, 933 (2024).
Zacchetti, L., Magnoni, S., Di Corte, F., Zanier, E. R. & Stocchetti, N. Accuracy of intracranial pressure monitoring: systematic review and meta-analysis. Crit. Care 19, 420 (2015).
Rozanek, M., Skola, J., Horakova, L. & Trukhan, V. Effect of artifacts upon the pressure reactivity index. Sci. Rep. 12, 15131 (2022).
Deburchgraeve, W. et al. Automated neonatal seizure detection mimicking a human observer reading EEG. Clin. Neurophysiol. 119, 2447–2454 (2008).
Gasser, T., Ziegler, P. & Gattaz, W. F. The deleterious effect of ocular artefacts on the quantitative EEG, and a remedy. Eur. Arch. Psychiatry Clin. Neurosci. 241, 352–356 (1992).
Zanon, C., Bini, C., Toniolo, A., Benetti, T. & Quaia, E. Radiation overuse in intensive care units. Tomography 10, 193–202 (2024).
Su, Y.-T. et al. Unnecessary radiation exposure during diagnostic radiography in infants in a neonatal intensive care unit: a retrospective cohort study. Eur. J. Pediatr. 182, 343–352 (2023).
Moxham, I. Understanding arterial pressure waveforms. South. Afr. J. Anaesth. Analg. 9, 40–42 (2003).
Crunkhorn, S. Developing closed-loop neuromodulatory devices. Nat. Rev. Drug Discov. 18, 98–98 (2019).
Bögli, S. Y. et al. Dynamic assessment of signal entropy for prognostication and secondary brain insult detection after traumatic brain injury. Crit. Care 28, 436 (2024).
Murray, M. L. et al. Data provenance and integrity of health-care systems data for clinical trials. Lancet Digit. Health 4, e567–e568 (2022).
Harrois, A. et al. Effects of routine position changes and tracheal suctioning on intracranial pressure in traumatic brain injury patients. J. Neurotrauma 37, 2227–2233 (2020).
Lee, S.-B. et al. Artifact removal from neurophysiological signals: impact on intracranial and arterial pressure monitoring in traumatic brain injury. J. Neurosurg. 132, 1952–1960 (2020).
Czosnyka, M. et al. Continuous assessment of the cerebral vasomotor reactivity in head injury. Neurosurgery 41, 11 (1997).
Park, S. et al. Inaugural state of the union: continuous cerebral autoregulation monitoring in the clinical practice of neurocritical care and anesthesia. Neurocrit. Care 40, 855–864 (2024).
Bögli, S. Y. et al. Unlocking the potential of high-resolution multimodality neuromonitoring for traumatic brain injury management: lessons and insights from cases, events, and patterns. Crit. Care 29, 139 (2025).
Ben-Hamouda, N., Ltaief, Z., Kirsch, M., Chiche, J.-D. & Rossetti, A. O. NIRS monitoring missing brain death in an ECMO patient. J. Clin. Monit. Comput. 39, 641–643 (2025).
Pelah, A. I. et al. Accuracy of intracranial pressure monitoring-single centre observational study and literature review. Sensors 23, 3397 (2023).
Nizami, S., Green, J. R. & McGregor, C. Implementation of artifact detection in critical care: a methodological review. IEEE Rev. Biomed. Eng. 6, 127–142 (2013).
Wang, X., Gao, Y., Lin, J., Rangwala, H. & Mittu, R. A machine learning approach to false alarm detection for critical arrhythmia alarms. In Proc. 2015 IEEE 14th International Conference on Machine Learning and Applications (ICMLA) 202–207 (IEEE, 2015).
Svedung Wettervik, T. et al. Brain tissue oxygen monitoring in traumatic brain injury: part I—to what extent does PbtO2 reflect global cerebral physiology? Crit. Care 27, 339 (2023).
J, Z. et al. Application of translation wavelet transform with new threshold function in pulse wave signal denoising. Technol. Health Care 31, 551–563 (2023).
Megjhani, M. et al. Automatic identification of intracranial pressure waveform during external ventricular drainage clamping: segmentation via wavelet analysis. Physiol. Meas. https://doi.org/10.1088/1361-6579/acdf3b (2023).
Wang, X. et al. Early qualitative and quantitative amplitude-integrated electroencephalogram and raw electroencephalogram for predicting long-term neurodevelopmental outcomes in extremely preterm infants in the Netherlands: a 10-year cohort study. Lancet Digit. Health 5, e895–e904 (2023).
Fló, A., Gennari, G., Benjamin, L. & Dehaene-Lambertz, G. Automated Pipeline for Infants Continuous EEG (APICE): a flexible pipeline for developmental cognitive studies. Dev. Cogn. Neurosci. 54, 101077 (2022).
Mataczynski, C. et al. End-to-end automatic morphological classification of intracranial pressure pulse waveforms using deep learning. IEEE J. Biomed. Health Inform. 26, 494–504 (2022).
Keenan, D. & Wilhelm, F. Adaptive and wavelet filtering methods for improving accuracy of respiratory measurement. Biomed. Sci. Instrum. 41, 37–42 (2005).
Puspasari, I., Mengko, T. L. R., Setiawan, A. W., Adiono, T. & Pramudyo, M. Denoising of heart sound signal for myocardial infarction detection based on adaptive filtering. In Proc. 2023 IEEE 5th Eurasia Conference on Biomedical Engineering, Healthcare and Sustainability (ECBIOS) 213–216 (IEEE, 2023).
Hu, X. et al. Intracranial pressure pulse morphological features improved detection of decreased cerebral blood flow. Physiol. Meas. 31, 679 (2010).
Xin, Y. et al. A vital sign signal noise suppression method for wearable piezoelectric devices. Rev. Sci. Instrum. 94, 095104 (2023).
Klug, M., Berg, T. & Gramann, K. Optimizing EEG ICA decomposition with data cleaning in stationary and mobile experiments. Sci. Rep. 14, 14119 (2024).
Occhipinti, E., Davies, H. J., Hammour, G. & Mandic, D. P. Hearables: artefact removal in Ear-EEG for continuous 24/7 monitoring. In Proc. International Joint Conference on Neural Networks (IJCNN) 1–6 (IEEE, 2022).
Smielewski, P. et al. Advanced neuromonitoring powered by ICM+ and its place in the Brand New AI World, reflections at the 20th anniversary boundary. Brain Spine 4, 102835 (2024).
Kazimierska, A. et al. Analysis of the shape of intracranial pressure pulse waveform in traumatic brain injury patients. In Proc. 43rd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC) 546–549 (IEEE, 2021).
Caicedo, A. & Van Huffel, S. Weighted LS-SVM for function estimation applied to artifact removal in bio-signal processing. In Proc. 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology 988–991 (IEEE, 2010).
Cabeleira, M., Fedriga, M. & Smielewski, P. Automatic pulse classification for artefact removal using SAX strings, a CENTER-TBI study. Acta Neurochir. Suppl. 131, 231–234 (2021).
Tsien, C. L., Kohane, I. S. & McIntosh, N. Multiple signal integration by decision tree induction to detect artifacts in the neonatal intensive care unit. Artif. Intell. Med. 19, 189–202 (2000).
Son, Y. et al. Automated artifact elimination of physiological signals using a deep belief network: an application for continuously measured arterial blood pressure waveforms. Inf. Sci. 456, 145–158 (2018).
Kim, H., Lee, S.-B., Son, Y., Czosnyka, M. & Kim, D.-J. Hemodynamic instability and cardiovascular events after traumatic brain injury predict outcome after artifact removal with deep belief network analysis. J. Neurosurg. Anesthesiol. 30, 347 (2018).
Au-Yeung, W.-T. M., Sahani, A. K., Isselbacher, E. M. & Armoundas, A. A. Reduction of false alarms in the intensive care unit using an optimized machine learning based approach. npj Digit. Med. 2, 1–5 (2019).
Ghosal, P., Himavathi, S. & Srinivasan, E. Random forest based feature ranking to evaluate the effect of motion artifact on different clinical features of PPG signal. In Proc. 2023 International Conference on Intelligent Systems for Communication, IoT and Security (ICISCoIS) 562–567 (IEEE, 2023).
LeCun, Y., Bengio, Y. & Hinton, G. Deep learning. Nature 521, 436–444 (2015).
Ioffe, S. & Szegedy, C. Batch normalization: accelerating deep network training by reducing internal covariate shift. In Proc. 32nd International Conference on Machine Learning 448–456 (PMLR, 2015).
Srivastava, N., Hinton, G., Krizhevsky, A., Sutskever, I. & Salakhutdinov, R. Dropout: a simple way to prevent neural networks from overfitting. J. Mach. Learn. Res. 15, 1929–1958 (2014).
Chilamkurthy, S. et al. Deep learning algorithms for detection of critical findings in head CT scans: a retrospective study. Lancet 392, 2388–2396 (2018).
He, K., Zhang, X., Ren, S. & Sun, J. Deep residual learning for image recognition. In Proc. 2016 IEEE Conference on Computer Vision and Pattern Recognition (CVPR) 770–778 (IEEE, 2016).
Megjhani, M. et al. An active learning framework for enhancing identification of non-artifactual intracranial pressure waveforms. Physiol. Meas. 40, 015002 (2019).
Cohn, D. A., Ghahramani, Z. & Jordan, M. I. Active learning with statistical models. J. Artif. Intell. Res. 4, 129–145 (1996).
Gal, Y., Islam, R. & Ghahramani, Z. Deep Bayesian active learning with image data. In Proc. 34th International Conference on Machine Learning 1183–1192 (PMLR, 2017).
Festag, S. & Spreckelsen, C. in German Medical Data Sciences: Bringing Data to Life 118–125 (IOS Press, 2021).
Holland, A. & Asgari, S. A simple unsupervised, real-time clustering method for arterial blood pressure signal classification. In Proc. Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) 1509–1512 (IEEE, 2019).
Haule, H. et al. VAE-IF: Deep feature extraction with averaging for fully unsupervised artifact detection in routinely acquired ICU time-series. Comput. Biol. Med. 186, 109610 (2025).
Franklin, D. et al. Synchronized wearables for the detection of haemodynamic states via electrocardiography and multispectral photoplethysmography. Nat. Biomed. Eng. 7, 1229–1241 (2023).
Krishnan, R., Rajpurkar, P. & Topol, E. J. Self-supervised learning in medicine and healthcare. Nat. Biomed. Eng. 6, 1346–1352 (2022).
Edinburgh, T. et al. in Intracranial Pressure and Neuromonitoring XVII (eds Depreitere, B., Meyfroidt, G. & Güiza, F.) 235–241 (Springer International, 2021).
Chen, J. et al. Denosieformer: a transformer-based approach for single-channel EEG artifact removal. IEEE Trans. Instrum. Meas. 73, 1–16 (2024).
Nowroozilarki, Z., Mortazavi, B. J. & Jafari, R. Variational autoencoders for biomedical signal morphology clustering and noise detection. IEEE J. Biomed. Health Inform. 28, 169–180 (2024).
Sheu, M.-H. et al. Lightweight denoising autoencoder design for noise removal in electrocardiography. IEEE Access. 10, 98104–98116 (2022).
Van Breugel, B., Liu, T., Oglic, D. & Van Der Schaar, M. Synthetic data in biomedicine via generative artificial intelligence. Nat. Rev. Bioeng. 2, 991–1004 (2024).
Bhattacharyay, S. et al. Mining the contribution of intensive care clinical course to outcome after traumatic brain injury. npj Digit. Med. 6, 1–11 (2023).
Bergmann, T. et al. Artifact identification and removal methodologies for intracranial pressure signals: a systematic scoping review. Physiol. Meas. 45, 12TR01 (2024).
Deisenhofer, I. et al. Artificial intelligence for individualized treatment of persistent atrial fibrillation: a randomized controlled trial. Nat. Med. 31, 1286–1293 (2025).
Johnson, L. S. et al. Artificial intelligence for direct-to-physician reporting of ambulatory electrocardiography. Nat. Med. 31, 925–931 (2025).
Komorowski, M., Celi, L. A., Badawi, O., Gordon, A. C. & Faisal, A. A. The artificial intelligence clinician learns optimal treatment strategies for sepsis in intensive care. Nat. Med. 24, 1716–1720 (2018).
Tang, H. et al. Injectable ultrasonic sensor for wireless monitoring of intracranial signals. Nature 630, 84–90 (2024).
Zhou, S. et al. Transcranial volumetric imaging using a conformal ultrasound patch. Nature 629, 810–818 (2024).
Verma, V. et al. Brain temperature monitoring in newborn infants: current methodologies and prospects. Front. Pediatr. 10, 1008539 (2022).
Siddiquee, M. R. et al. Movement artefact removal from NIRS signal using multi-channel IMU data. Biomed. Eng. Online 17, 120 (2018).
Aviles-Espinosa, R., Dore, H. & Rendon-Morales, E. An experimental method for bio-signal denoising using unconventional sensors. Sensors 23, 3527 (2023).
Zhou, A. et al. A wireless and artefact-free 128-channel neuromodulation device for closed-loop stimulation and recording in non-human primates. Nat. Biomed. Eng. 3, 15–26 (2019).
Schalk, G. et al. Translation of neurotechnologies. Nat. Rev. Bioeng. 2, 637–652 (2024).
Sun, K., Roy, A. & Tobin, J. M. Artificial intelligence and machine learning: definition of terms and current concepts in critical care research. J. Crit. Care 82, 154792 (2024).
Ercole, A. et al. Guidelines for Data Acquisition, Quality and Curation for Observational Research Designs (DAQCORD). J. Clin. Transl. Sci. 4, 354–359 (2020).
Beqiri, E. et al. Common data elements for disorders of consciousness: recommendations from the Working Group on Physiology and Big Data. Neurocrit. Care 39, 593–599 (2023).
Nan, Y. et al. Data harmonisation for information fusion in digital healthcare: a state-of-the-art systematic review, meta-analysis and future research directions. Inf. Fusion. 82, 99–122 (2022).
Bernhardt, M., Jones, C. & Glocker, B. Potential sources of dataset bias complicate investigation of underdiagnosis by machine learning algorithms. Nat. Med. 28, 1157–1158 (2022).
Leek, J. T. et al. Tackling the widespread and critical impact of batch effects in high-throughput data. Nat. Rev. Genet. 11, 733–739 (2010).
Pivovarov, R., Albers, D. J., Sepulveda, J. L. & Elhadad, N. Identifying and mitigating biases in EHR laboratory tests. J. Biomed. Inform. 51, 24–34 (2014).
Bower, J. K., Patel, S., Rudy, J. E. & Felix, A. S. Addressing bias in electronic health record-based surveillance of cardiovascular disease risk: finding the signal through the noise. Curr. Epidemiol. Rep. 4, 346–352 (2017).
Amor, L. B., Lahyani, I., Jmaiel, M. & Drira, K. Anomaly detection and diagnosis scheme for mobile health applications. In Proc. International Conference on Advanced Information Networking and Applications (AINA) 777–784 (IEEE, 2018).
Chen, S. F., Guo, Z., Ding, C., Hu, X. & Rudin, C. Sparse learned kernels for interpretable and efficient medical time series processing. Nat. Mach. Intell. 6, 1132–1144 (2024).
Liu, Q. & Paparrizos, J. The elephant in the room: towards a reliable time-series anomaly detection benchmark. In Proc. Advances in Neural Information Processing Systems 37 108231–108261 (NeurIPS 2024).
Topol, E. J. High-performance medicine: the convergence of human and artificial intelligence. Nat. Med. 25, 44–56 (2019).
Huber, P. J. Robust estimation of a location parameter. Ann. Math. Stat. 35, 73–101 (1964).
Gulamali, F. et al. Derivation, external and clinical validation of a deep learning approach for detecting intracranial hypertension. npj Digit. Med. 7, 233 (2024).
Mascia, L. et al. The accuracy of transcranial Doppler to detect vasospasm in patients with aneurysmal subarachnoid hemorrhage. Intensive Care Med. 29, 1088–1094 (2003).
Qiu, J. et al. LLM-based agentic systems in medicine and healthcare. Nat. Mach. Intell. 6, 1418–1420 (2024).
Tang, C. et al. A roadmap for the development of human body digital twins. Nat. Rev. Electr. Eng. 1, 199–207 (2024).
Chen, X., Tang, C., Dai, Y., Li, C. & Gao, S. in Blockchain and Digital Twin for Smart Hospitals (ed. Nguyen, T. A.) Ch. 18 (Elsevier, 2025).
Sandmann, S. et al. Benchmark evaluation of DeepSeek large language models in clinical decision-making. Nat. Med. 31, 2546–2549 (2025).
Johnson, A. E. W. et al. MIMIC-III, a freely accessible critical care database. Sci. Data 3, 160035 (2016).
Yang, F. et al. Group-based trajectory modeling of intracranial pressure in patients with acute brain injury: Results from multi-center ICUs, 2008-2019. CNS Neurosci. Ther. 28, 1218–1228 (2022).
Pollard, T. J. et al. The eICU collaborative research database, a freely available multi-center database for critical care research. Sci. Data 5, 180178 (2018).
Piper, I. et al. The BrainIT group: concept and core dataset definition. Acta Neurochir. 145, 615–628 (2003).
Piper, I. et al. The Brain monitoring with Information Technology (BrainIT) collaborative network: past, present and future direction. Romanian Neurosurg. 17, 12–33 (2010).
Kim, N. et al. Trending autoregulatory indices during treatment for traumatic brain injury. J. Clin. Monit. Comput. 30, 821–831 (2016).
Maas, A. I. R. et al. Collaborative European NeuroTrauma Effectiveness Research in Traumatic Brain Injury (CENTER-TBI): a prospective longitudinal observational study. Neurosurgery 76, 67–80 (2015).
Yue, J. K. et al. Transforming research and clinical knowledge in traumatic brain injury pilot: multicenter implementation of the common data elements for traumatic brain injury. J. Neurotrauma 30, 1831–1844 (2013).
Greil, M. E. et al. External ventricular drain versus intraparenchymal pressure monitor in severe traumatic brain injury: a TRACK-TBI study. Neurosurgery https://doi.org/10.1227/neu.0000000000003594 (2025).
Usuga, F. A. C., Gissel, C. & Hernández, A. M. Motion artifact reduction in electrocardiogram signals through a redundant denoising independent component analysis method for wearable health care monitoring systems: algorithm development and validation. JMIR Med. Inform. 10, e40826 (2022).
Allan Gunn, C., Hu, X. & Vandenberghe, L. Artifact rejection and missing data imputation in cerebral blood flow velocity signals via trace norm minimization. Physiol. Meas. 41, 114003 (2020).
Ghosh, A. K., Catelli, D. S., Wilson, S., Nowlan, N. C. & Vaidyanathan, R. Multi-modal detection of fetal movements using a wearable monitor. Inf. Fusion. 103, 102124 (2024).
Deng, A. & Hooi, B. Graph neural network-based anomaly detection in multivariate time series. In Proc. 35th AAAI Conference on Artificial Intelligence (AAAI-21) 4027–4035 (AAAI Press, 2021).
Acknowledgements
X.C. thanks the Cambridge Trust and the China Scholarship Council for PhD financial support. S.G. acknowledges the National Natural Science Foundation of China (award ID: 62171014). The authors thank M. Czosnyka for valuable feedback on the structure and content of the manuscript.
Author information
Authors and Affiliations
Contributions
X.C. researched data, contributed to content discussion, and wrote and edited the manuscript. S.Y.B. and I.O. researched data and contributed to discussion of content. W.X., E.B., C.T., L.G.O., S.G. and P.S. contributed to content discussion. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Citation diversity statement
The authors acknowledge that papers authored by scholars from historically excluded groups are systematically under-cited. They have made every attempt to reference relevant papers in a manner that is equitable in terms of racial, ethnic, gender and geographical representation.
Peer review
Peer review information
Nature Reviews Bioengineering thanks Gregory Hawryluk, Sergio Brasil and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, X., Bögli, S.Y., Olakorede, I. et al. Artefacts in continuous neuromonitoring. Nat Rev Bioeng (2026). https://doi.org/10.1038/s44222-025-00378-3
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44222-025-00378-3