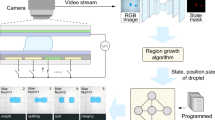
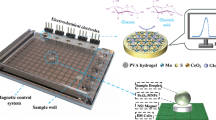
Abstract
Active-matrix digital microfluidics (AM-DMF) leverages semiconductor-derived electrode arrays to dynamically control thousands of micrometre-scale droplets and has emerged as a transformative platform for high-throughput and precise manipulation of liquid samples. This technology enables various programmable operations, such as droplet generation, transport, mixing and dilution, to be performed with unparalleled accuracy and, thereby, overcomes several limitations of conventional microchannel and passive-matrix digital microfluidics. This Review provides a critical analysis of the design principles and transformative potential of AM-DMF, focusing on its potential biomedical applications in genomics, single-cell analysis and drug discovery. Important contributions of artificial intelligence that increase the efficiency and reliability of complex AM-DMF workflows are also discussed. Despite this considerable progress, further innovation is needed to overcome ongoing challenges such as biofouling, reagent selectivity and electrode stability. This Review outlines future directions for AM-DMF as a versatile tool in life sciences and showcases its role in enabling next-generation droplet manipulation and workflow automation.
Key points
-
The transition from passive-matrix (PM) to active-matrix (AM) digital microfluidics (DMF) chip configurations notably enhances scalability, enabling both efficient and precise control of large-scale droplet arrays.
-
AM-DMF primarily uses various thin-film transistor types, printed circuit board or complementary metal oxide semiconductor processes; its driving circuits have undergone continuous evolution to enable precise droplet driving and sensing.
-
The evolution of DMF architectures from PM (DMF 1.0) through AM (DMF 2.0), gate-on-array (DMF 2.5) and integrated circuit-driven (DMF 3.0) iterations has been a key driver in advancing the commercialization of this technology.
-
Integration with multifunctional modules and artificial intelligence renders AM-DMF a promising platform for automated, multifunctional workflows.
-
AM-DMF is a transformative platform for high-throughput applications such as genomics, single-cell analysis and drug discovery.
-
Ongoing challenges such as biofouling, reagent compatibility and electrode stability remain to be addressed to promote the widespread adoption and commercialization of AM-DMF.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Sackmann, E. K., Fulton, A. L. & Beebe, D. J. The present and future role of microfluidics in biomedical research. Nature 507, 181–189 (2014).
Dou, Y. et al. Orally administrable H2S-scavenging metal–organic framework prepared by co-flow microfluidics for comprehensive restoration of intestinal milieu. Adv. Mater. 35, 2210047 (2023).
Zhang, D., Bi, H., Liu, B. & Qiao, L. Detection of pathogenic microorganisms by microfluidics based analytical methods. Anal. Chem. 90, 5512–5520 (2018).
Czerniecki, S. M. et al. High-throughput screening enhances kidney organoid differentiation from human pluripotent stem cells and enables automated multidimensional phenotyping. Cell Stem Cell 22, 929.e4–940.e4 (2018).
Cao, Y. et al. Bead-jet printing enabled sparse mesenchymal stem cell patterning augments skeletal muscle and hair follicle regeneration. Nat. Commun. 13, 7463 (2022).
Zhong, B. et al. Interindividual- and blood-correlated sweat phenylalanine multimodal analytical biochips for tracking exercise metabolism. Nat. Commun. 15, 624 (2024).
Gopinathan, K. A., Mishra, A., Mutlu, B. R., Edd, J. F. & Toner, M. A microfluidic transistor for automatic control of liquids. Nature 622, 735–741 (2023).
Peterson, V. M. et al. Multiplexed quantification of proteins and transcripts in single cells. Nat. Biotechnol. 35, 936–939 (2017).
Li, C. et al. Under oil open-channel microfluidics empowered by exclusive liquid repellency. Sci. Adv. 6, eaay9919 (2020).
Novak, R. et al. Robotic fluidic coupling and interrogation of multiple vascularized organ chips. Nat. Biomed. Eng. 4, 407–420 (2020).
Li, J., Ha, N. S., Liu, T. ', Van Dam, R. M. & Kim, C.-J. ‘CJ’ Ionic-surfactant-mediated electro-dewetting for digital microfluidics. Nature 572, 507–510 (2019).
Samiei, E., Tabrizian, M. & Hoorfar, M. A review of digital microfluidics as portable platforms for lab-on a-chip applications. Lab Chip 16, 2376–2396 (2016).
Cai, L. et al. Highly multiplexing, throughput and efficient single-cell protein analysis with digital microfluidics. Small Methods 8, e2400375 (2024).
Liang, S. et al. DMF-Bimol: counting mRNA and protein molecules in single cells with digital microfluidics. Anal. Chem. 96, 17253–17261 (2024).
Antkowiak, P. L. et al. Integrating DNA encapsulates and digital microfluidics for automated data storage in DNA. Small 18, e2107381 (2022).
Yetisen, A. K., Akram, M. S. & Lowe, C. R. Paper-based microfluidic point-of-care diagnostic devices. Lab Chip 13, 2210–2251 (2013).
Hou, Y. et al. Biosensor-based microfluidic platforms for rapid clinical detection of pathogenic bacteria. Adv. Funct. Mater. 35, 2411484 (2025).
Hu, C. et al. “Cell-on-demand” digital microfluidics for real-time low-abundance single-cell isolation and sample analysis. Small 21, e2504239 (2025).
Mazutis, L. et al. Single-cell analysis and sorting using droplet-based microfluidics. Nat. Protoc. 8, 870–891 (2013).
Scott, E. Y. et al. Integrating single-cell and spatially resolved transcriptomic strategies to survey the astrocyte response to stroke in male mice. Nat. Commun. 15, 1584 (2024).
Torabinia, M., Dakarapu, U. S., Asgari, P., Jeon, J. & Moon, H. Electrowetting-on-dielectric (EWOD) digital microfluidic device for in-line workup in organic reactions: a critical step in the drug discovery work cycle. Sens. Actuators B Chem. 330, 129252 (2021).
Zhai, J. et al. Drug screening on digital microfluidics for cancer precision medicine. Nat. Commun. 15, 4363 (2024).
Ai, Y., Xie, R., Xiong, J. & Liang, Q. Microfluidics for biosynthesizing: from droplets and vesicles to artificial cells. Small 16, e1903940 (2020).
Ngocho, K. et al. Synthetic cells from droplet-based microfluidics for biosensing and biomedical applications. Small 20, e2400086 (2024).
Lin, G. et al. Magnetofluidic platform for multidimensional magnetic and optical barcoding of droplets. Lab Chip 15, 216–224 (2015).
Ding, X. et al. Surface acoustic wave microfluidics. Lab Chip 13, 3626–3649 (2013).
Park, J. et al. Acoustothermal tweezer for droplet sorting in a disposable microfluidic chip. Lab Chip 17, 1031–1040 (2017).
Fan, Y., Wu, H., Wang, J. & Lv, J. Field-programmable topographic-morphing array for general-purpose lab-on-a-chip systems. Adv. Mater. 37, 2410604 (2025).
Yang, B. et al. Automated and label-free detection of HIV DNA via digital microfluidics-chemiluminescence analysis. Sens. Actuators B Chem. 389, 133905 (2023).
Hong, S. Y. et al. Stretchable active matrix temperature sensor array of polyaniline nanofibers for electronic skin. Adv. Mater. 28, 930–935 (2016).
Shayan, M., Bhattacharjee, S., Song, Y.-A., Chakrabarty, K. & Karri, R. Toward secure microfluidic fully programmable valve array biochips. IEEE Trans. Very Large Scale Integr. VLSI Syst. 27, 2755–2766 (2019).
Hadwen, B. et al. Programmable large area digital microfluidic array with integrated droplet sensing for bioassays. Lab Chip 12, 3305–3313 (2012). This paper presents an AM-DMF device with integrated impedance sensing and demonstrates its application in a colorimetric glucose assay of human blood serum samples.
Ma, H. et al. Large-area manufacturable active matrix digital microfluidics platform for high-throughput biosample handling. In 2020 IEEE International Electron Devices Meeting 35.5.1–35.5.4 (IEEE, 2020). This work presents an a-Si:H TFT-based AM-DMF platform that achieves precise single-pixel droplet manipulation with a volume variation of ~1%.
Yu, J. et al. Field programmable digital microfluidics chip for high-throughput droplet array manipulation. In 2023 International Electron Devices Meeting 1–4 (IEEE, 2023). This paper presents an AM-DMF chip with 640 × 280 pixels that uses 9T2C GOA and 3T1C circuits to enable high-throughput parallel droplet manipulation and single-cell operations for advanced biological applications.
Anderson, S., Hadwen, B. & Brown, C. Thin-film-transistor digital microfluidics for high value in vitro diagnostics at the point of need. Lab Chip 21, 962–975 (2021). This paper demonstrates a fully automated sample-to-answer SARS-CoV-2 RT–PCR (PCR with reverse transcription) test from saliva using a high-resolution AM-DMF chip, showcasing its potential for point-of-need molecular diagnostics.
Wang, D. et al. Active-matrix digital microfluidics design for field programmable high-throughput digitalized liquid handling. iScience 27, 109324 (2024). This article introduces a field-programmable droplet array system with a novel TFT design that can manipulate thousands of 0.5-nl droplets.
Jiang, S. et al. Droplet position sensing in TFT active-matrix digital microfluidics. IEEE Electron. Device Lett. 45, 1361–1364 (2024).
Guo, Z. et al. Deep learning-assisted label-free parallel cell sorting with digital microfluidics. Adv. Sci. 12, 2408353 (2025). This work presents a label-free cell sorting method that integrates an AM-DMF platform with a YOLOv8-based deep learning algorithm to enable real-time image recognition and morphology-based cell sorting.
Lu, G.-R. et al. Flexible droplet routing in active matrix-based digital microfluidic biochips. ACM Trans. Des. Autom. Electron. Syst. 23, 37 (2018).
Jia, Z. et al. Artificial intelligence-enabled multipurpose smart detection in active-matrix electrowetting-on-dielectric digital microfluidics. Microsyst. Nanoeng. 10, 139 (2024).
Geng, H., Feng, J., Stabryla, L. M. & Cho, S. K. Dielectrowetting manipulation for digital microfluidics: creating, transporting, splitting, and merging of droplets. Lab Chip 17, 1060–1068 (2017).
Shin, D. J., Trick, A. Y., Hsieh, Y.-H., Thomas, D. L. & Wang, T.-H. Sample-to-answer droplet magnetofluidic platform for point-of-care hepatitis C viral load quantitation. Sci. Rep. 8, 9793 (2018).
Qian, J. et al. Rapid and comprehensive detection of viral antibodies and nucleic acids via an acoustofluidic integrated molecular diagnostics chip: AIMDx. Sci. Adv. 11, eadt5464 (2025).
Lee, K. S. et al. Optofluidic Raman-activated cell sorting for targeted genome retrieval or cultivation of microbial cells with specific functions. Nat. Protoc. 16, 634–676 (2021).
Yafia, M. et al. Microfluidic chain reaction of structurally programmed capillary flow events. Nature 605, 464–469 (2022).
Abdelgawad, M., Park, P. & Wheeler, A. R. Optimization of device geometry in single-plate digital microfluidics. J. Appl. Phys. 105, 094506 (2009).
Peng, J. et al. All-in-one digital microfluidics pipeline for proteomic sample preparation and analysis. Chem. Sci. 14, 2887–2900 (2023).
Peng, C., Zhang, Z., Kim, C. J. & Ju, Y. S. EWOD (electrowetting on dielectric) digital microfluidics powered by finger actuation. Lab Chip 14, 1117–1122 (2014).
Tan, J. et al. Orbital electrowetting-on-dielectric for droplet manipulation on superhydrophobic surfaces. Adv. Mater. 36, 2314346 (2024).
Gong, J. & Kim, C. J. All-electronic droplet generation on-chip with real-time feedback control for EWOD digital microfluidics. Lab Chip 8, 898–906 (2008).
Li, J. & Kim, C. C. Current commercialization status of electrowetting-on-dielectric (EWOD) digital microfluidics. Lab Chip 20, 1705–1712 (2020).
Lippmann, M. G. Relations entre les phénomènes électriques et capillaires [French]. Ann. Chim. Phys. 5, 494–579 (1875).
Berge, B. Electrocapillarity and wetting of insulator films by water. Comptes Rendus Acad. Sci. 2b Mec. 317, 157–163 (1993).
Cho, S. K., Moon, H. & Kim, C.-J. Creating, transporting, cutting, and merging liquid droplets by electrowetting-based actuation for digital microfluidic circuits. J. Microelectromech. Syst. 12, 70–80 (2003).
Chatterjee, D., Shepherd, H. & Garrell, R. L. Electromechanical model for actuating liquids in a two-plate droplet microfluidic device. Lab Chip 9, 1219–1229 (2009).
Li, D. et al. Point-of-care blood coagulation assay enabled by printed circuit board-based digital microfluidics. Lab Chip 22, 709–716 (2022).
Vo, P. Q. N., Husser, M. C., Ahmadi, F., Sinha, H. & Shih, S. C. C. Image-based feedback and analysis system for digital microfluidics. Lab Chip 17, 3437–3446 (2017).
Jain, V. & Patrikar, R. M. A low-cost portable dynamic droplet sensing system for digital microfluidics applications. IEEE Trans. Instrum. Meas. 69, 3623–3630 (2020).
Ahmadi, F. et al. Integrating machine learning and digital microfluidics for screening experimental conditions. Lab Chip 23, 81–91 (2023).
Shen, J. et al. A low-temperature digital microfluidic system used for protein–protein interaction detection. Lab Chip 23, 4390–4399 (2023).
Li, M. et al. One-shot high-resolution melting curve analysis for KRAS point-mutation discrimination on a digital microfluidics platform. Lab Chip 22, 537–549 (2022).
Jiang, S. et al. Thin-film transistor digital microfluidics circuit design with capacitance-based droplet sensing. Sensors 24, 4789 (2024).
Wang, D. et al. Active-matrix digital microfluidics design and optimization for high-throughput droplets manipulation. IEEE J. Electron. Devices Soc. 11, 411–415 (2023).
Luo, Z. et al. Programmable high integration and resolution digital microfluidic device driven by thin film transistor arrays. IEEE Access 10, 30573–30582 (2022). This paper presents a high-resolution integrated circuit-driven AM-DMF device featuring a custom control system with droplet detection and feedback functions.
Qin, F. et al. Solution for mass production of high-throughput digital microfluidic chip based on a-Si TFT with in-pixel boost circuit. Micromachines 12, 1199 (2021).
Xu, Y. et al. Digital microfluidic lab-on-a-chip on a TFT glass substrate enabling point-of-care testing. IEEE Electron. Device Lett. 44, 1500–1503 (2023).
Hu, S. et al. Large-area electronics-enabled high-resolution digital microfluidics for parallel single-cell manipulation. Anal. Chem. 95, 6905–6914 (2023).
Brakke, K. A. The Surface Evolver. Exp. Math. 1, 141–165 (1992).
Lienemann, J., Greiner, A. & Korvink, J. G. in Design Automation Methods and Tools for Microfluidics-Based Biochips (eds Chakrabarty, K. & Zeng, J.) 53–84 (Springer, 2006).
Alistar, M., Pop, P. & Madsen, J. Synthesis of application-specific fault-tolerant digital microfluidic biochip architectures. IEEE Trans. Comput. Aided Des. Integr. Circuits Syst. 35, 764–777 (2016).
Madec, M. et al. Multiphysics simulation of biosensors involving 3D biological reaction–diffusion phenomena in a standard circuit EDA environment. IEEE Trans. Circuits Syst. Regul. Pap. 66, 2188–2197 (2019).
Zeng, J. Modeling and simulation of electrified droplets and its application to computer-aided design of digital microfluidics. IEEE Trans. Comput. Aided Des. Integr. Circuits Syst. 25, 224–233 (2006).
Lienemann, J., Greiner, A. & Korvink, J. G. Modeling, simulation, and optimization of electrowetting. IEEE Trans. Comput. Aided Des. Integr. Circuits Syst. 25, 234–247 (2006).
Fink, G., Hamidovic, M., Haselmayr, W. & Wille, R. Automatic design of droplet-based microfluidic ring networks. IEEE Trans. Comput. Aided Des. Integr. Circuits Syst. 40, 339–349 (2021).
Lee, H., Liu, Y., Ham, D. & Westervelt, R. M. Integrated cell manipulation system — CMOS/microfluidic hybrid. Lab Chip 7, 331–337 (2007).
Khorasani, M., Behnam, M., Van Den Berg, L., Backhouse, C. J. & Elliott, D. G. High-voltage CMOS controller for microfluidics. IEEE Trans. Biomed. Circuits Syst. 3, 89–96 (2009).
Noh, J. H., Noh, J., Kreit, E., Heikenfeld, J. & Rack, P. D. Toward active-matrix lab-on-a-chip: programmable electrofluidic control enabled by arrayed oxide thin film transistors. Lab Chip 12, 353–360 (2011).
Cao, W. et al. The future transistors. Nature 620, 501–515 (2023).
Wan, C. et al. Toward a brain–neuromorphics interface. Adv. Mater. 36, 2311288 (2024).
Hsiao, H., Hsu, Y., Liu, A., Hsieh, J. & Lin, Y. Ultrasensitive refractive index sensing based on the quasi-bound states in the continuum of all-dielectric metasurfaces. Adv. Opt. Mater. 10, 2200812 (2022).
Wu, T. & Suzuki, Y. Liquid dielectrophoresis on electret: a novel approach towards CMOS-driven digital microfludics. J. Adhes. Sci. Technol. 26, 2025–2045 (2012).
Chung, J., Hwang, H. Y., Chen, Y. & Lee, T. Y. Microfluidic packaging of high-density CMOS electrode array for lab-on-a-chip applications. Sens. Actuators B Chem. 254, 542–550 (2018).
Pollack, M. G. Electrowetting-Based Microactuation of Droplets for Digital Microfluidics. PhD dissertation, Duke Univ. (2001).
Khan, S. M., Gumus, A., Nassar, J. M. & Hussain, M. M. CMOS enabled microfluidic systems for healthcare based applications. Adv. Mater. 30, 1705759 (2018).
Carvalho, J. et al. Single-use microfluidic device for purification and concentration of environmental DNA from river water. Talanta 226, 122109 (2021).
Hua, Z. et al. Multiplexed real-time polymerase chain reaction on a digital microfluidic platform. Anal. Chem. 82, 2310–2316 (2010).
Geng, D. et al. Thin-film transistors for large-area electronics. Nat. Electron. 6, 963–972 (2023).
Nathan, A. et al. Amorphous silicon thin film transistor circuit integration for organic LED displays on glass and plastic. IEEE J. Solid State Circuits 39, 1477–1486 (2004).
Meng, Z., Wang, M. & Wong, M. High performance low temperature metal-induced unilaterally crystallized polycrystalline silicon thin film transistors for system-on-panel applications. IEEE Trans. Electron. Devices 47, 404–409 (2000).
Lee, S. & Nathan, A. Subthreshold Schottky-barrier thin-film transistors with ultralow power and high intrinsic gain. Science 354, 302–304 (2016).
Kohno, A., Sameshima, T., Sano, N., Sekiya, M. & Hara, M. High performance poly-Si TFTs fabricated using pulsed laser annealing and remote plasma CVD with low temperature processing. IEEE Trans. Electron. Devices 42, 251–257 (1995).
Siddik, A. B. et al. Blue laser annealed sub-micron channel p-type low temperature poly-Si TFT without kink effect for high-resolution display. IEEE Electron. Device Lett. 42, 172–175 (2021).
Sheng, J. et al. Amorphous IGZO TFT with high mobility of ∼70 cm2 /(V s) via vertical dimension control using PEALD. ACS Appl. Mater. Interfaces 11, 40300–40309 (2019).
Xing, Y. et al. A robust and scalable active-matrix driven digital microfluidic platform based on printed-circuit board technology. Lab Chip 21, 1886–1896 (2021). This work proposes a robust and scalable PCB-based AM-DMF platform and demonstrates its versatility through the on-chip synthesis of a pentapeptide.
Jiang, S., Wang, D., Ma, H., Nathan, A. & Yu, J. Low temperature polysilicon pixel circuits for active-matrix digital microfluidic chips. Displays 88, 103048 (2025).
Kwon, S. et al. Prototype of flexible digital X-ray imaging detector. IEEE Electron. Device Lett. 45, 2439–2442 (2024).
Hu, C., Jin, K. & Ma, H. A universal model for continuous “one-to-two” high-efficient droplet generation in digital microfluidics. Appl. Phys. Lett. 122, 181601 (2023).
Jin, K. et al. “One-to-three” droplet generation in digital microfluidics for parallel chemiluminescence immunoassays. Lab Chip 21, 2892–2900 (2021).
Shaik, F. A. et al. Thin-film-transistor array: an exploratory attempt for high throughput cell manipulation using electrowetting principle. J. Micromech. Microeng. 27, 054001 (2017).
Yang, Z. et al. AM-DMF-SCP: integrated single-cell proteomics analysis on an active matrix digital microfluidic chip. JACS Au 4, 1811–1823 (2024). This study presents an integrated AM-DMF platform for all-in-one single-cell proteomics processing, which minimizes sample loss and improves the sensitivity of mass spectrometry analysis.
Zhang, B. et al. Polar coordinate active-matrix digital microfluidics for high-resolution concentration gradient generation. Lab Chip 24, 2193–2201 (2024).
Wei, Y., Wang, D., Jiang, S., Ma, H. & Yu, J. A high-voltage serial-in-parallel-out shift register with amorphous silicon TFTs. IEEE J. Electron. Devices Soc. 11, 416–420 (2023).
Kalsi, S. et al. Rapid and sensitive detection of antibiotic resistance on a programmable digital microfluidic platform. Lab Chip 15, 3065–3075 (2015). This study demonstrates rapid, highly sensitive detection of an antibiotic resistance gene via AM-DMF-based recombinase polymerase amplification, which achieved a 100-fold improvement in detection limit compared to benchtop methods.
Yang, G. et al. A portable driving system for high-resolution active matrix electrowetting display based on FPGA. J. Soc. Inf. Disp. 28, 287–296 (2020).
Zhang, T. et al. Optimizing bipolar reset waveform to improve grayscale stability in active matrix electrowetting displays. Micromachines 15, 1247 (2024).
Ji, J. et al. AM-DMF-ddRPA: an all-in-one digital microfluidic platform for rapid and automatic digital nucleic acid analysis. Angew. Chem. Int. Ed. 64, e202501913 (2025). This study describes a rapid, fully automated AM-DMF-based droplet digital recombinase polymerase amplification platform, which completes sample-to-answer quantitative detection of influenza viruses within 60 min.
Fobel, R., Fobel, C. & Wheeler, A. R. DropBot: an open-source digital microfluidic control system with precise control of electrostatic driving force and instantaneous drop velocity measurement. Appl. Phys. Lett. 102, 193513 (2013).
Saha, B., Das, B. & Majumder, M. A deep-reinforcement learning approach for optimizing homogeneous droplet routing in digital microfluidic biochips. Nanotechnol. Precis. Eng. 6, 023001 (2023).
Bandodkar, A. J. et al. Battery-free, skin-interfaced microfluidic/electronic systems for simultaneous electrochemical, colorimetric, and volumetric analysis of sweat. Sci. Adv. 5, eaav3294 (2019).
Xu, Y. et al. A TFT-based on-chip optical sensor for detection of chemiluminescence in lab-on-a-chip. IEEE Electron. Device Lett. 46, 1119–1122 (2025).
Hu, C. et al. Ultra-fast electronic detection of antimicrobial resistance genes using isothermal amplification and thin film transistor sensors. Biosens. Bioelectron. 96, 281–287 (2017).
Medina-Sánchez, M., Martínez-Domingo, C., Ramon, E. & Merkoçi, A. An inkjet-printed field-effect transistor for label-free biosensing. Adv. Funct. Mater. 24, 6291–6302 (2014).
Shah, G. J., Ohta, A. T., Chiou, E. P., Wu, M. C. & Kim, C. J. EWOD-driven droplet microfluidic device integrated with optoelectronic tweezers as an automated platform for cellular isolation and analysis. Lab Chip 9, 1732–1739 (2009).
Lamanna, J. et al. Digital microfluidic isolation of single cells for -omics. Nat. Commun. 11, 5632 (2020).
Jiang, S. et al. Efficient yeast cell collection and manipulation based on dielectrophoresis-integrated digital microfluidics. Langmuir 41, 8312–8321 (2025).
Huang, B. et al. Counting low-copy number proteins in a single cell. Science 315, 81–84 (2007).
Hoshino, K. et al. Microchip-based immunomagnetic detection of circulating tumor cells. Lab Chip 11, 3449–3457 (2011).
Wang, X. et al. Positive dielectrophoresis-based Raman-activated droplet sorting for culture-free and label-free screening of enzyme function in vivo. Sci. Adv. 6, eabb3521 (2020).
Wang, Z. et al. Structure-controllable Ag aerogel optimized SERS-digital microfluidic platform for ultrasensitive and high-throughput detection of harmful substances. Sens. Actuators B Chem. 401, 134934 (2024).
Das, A., Fehse, S., Polack, M., Panneerselvam, R. & Belder, D. Surface-enhanced Raman spectroscopic probing in digital microfluidics through a microspray hole. Anal. Chem. 95, 1262–1272 (2023).
Das, A. et al. On-the-fly mass spectrometry in digital microfluidics enabled by a microspray hole: toward multidimensional reaction monitoring in automated synthesis platforms. J. Am. Chem. Soc. 144, 10353–10360 (2022).
Wang, Y. et al. Highly sensitive and automated surface enhanced Raman scattering-based immunoassay for H5N1 detection with digital microfluidics. Anal. Chem. 90, 5224–5231 (2018).
Li, B. B. et al. Cell invasion in digital microfluidic microgel systems. Sci. Adv. 6, eaba9589 (2020).
Jia, Z. et al. Intelligent single-cell manipulation: LLMs- and object detection-enhanced active-matrix digital microfluidics. Microsyst. Nanoeng. 11, 133 (2025). This research develops an AM-DMF platform that leverages large language models for automated workflow generation and advanced object detection for high-precision single-cell manipulation and identification.
Shen, R. et al. Nucleic acid analysis on electrowetting-based digital microfluidics. Trends Anal. Chem. 158, 116826 (2023).
Shamsi, M. H., Choi, K., Ng, A. H. C. & Wheeler, A. R. A digital microfluidic electrochemical immunoassay. Lab Chip 14, 547–554 (2014).
Witters, D., Knez, K., Ceyssens, F., Puers, R. & Lammertyn, J. Digital microfluidics-enabled single-molecule detection by printing and sealing single magnetic beads in femtoliter droplets. Lab Chip 13, 2047–2054 (2013).
Shih, S. C. C., Barbulovic-Nad, I., Yang, X., Fobel, R. & Wheeler, A. R. Digital microfluidics with impedance sensing for integrated cell culture and analysis. Biosens. Bioelectron. 42, 314–320 (2013).
Au, S. H., Chamberlain, M. D., Mahesh, S., Sefton, M. V. & Wheeler, A. R. Hepatic organoids for microfluidic drug screening. Lab Chip 14, 3290–3299 (2014).
Zallocchi, M. et al. Piplartine attenuates aminoglycoside-induced TRPV1 activity and protects from hearing loss in mice. Sci. Transl. Med. 16, eadn2140 (2024).
Tummino, T. A. et al. Drug-induced phospholipidosis confounds drug repurposing for SARS-CoV-2. Science 373, 541–547 (2021).
Shinozawa, T. et al. High-fidelity drug-induced liver injury screen using human pluripotent stem cell-derived organoids. Gastroenterology 160, 831.e10–846.e10 (2021).
Uijttewaal, E. C. H. CRISPR-StAR enables high-resolution genetic screening in complex in vivo models. Nat. Biotechnol. 43, 1848–1860 (2025).
Song, S. et al. A cell-based multiplex immunoassay platform using fluorescent protein-barcoded reporter cell lines. Commun. Biol. 4, 1338 (2021).
Liu, Y., Zhan, L., Qin, Z., Sackrison, J. & Bischof, J. C. Ultrasensitive and highly specific lateral flow assays for point-of-care diagnosis. ACS Nano 15, 3593–3611 (2021).
Merola, F. et al. Tomographic flow cytometry by digital holography. Light Sci. Appl. 6, e16241 (2016).
Au, S. H., Kumar, P. & Wheeler, A. R. A new angle on pluronic additives: advancing droplets and understanding in digital microfluidics. Langmuir 27, 8586–8594 (2011).
Ng, A. H. C., Li, B. B., Chamberlain, M. D. & Wheeler, A. R. Digital microfluidic cell culture. Annu. Rev. Biomed. Eng. 17, 91–112 (2015).
Chatterjee, D., Hetayothin, B., Wheeler, A. R., King, D. J. & Garrell, R. L. Droplet-based microfluidics with nonaqueous solvents and solutions. Lab Chip 6, 199–206 (2006).
Choi, K., Ng, A. H. C., Fobel, R. & Wheeler, A. R. Digital microfluidics. Annu. Rev. Anal. Chem. 5, 413–440 (2012).
Dong, C. et al. On the droplet velocity and electrode lifetime of digital microfluidics: voltage actuation techniques and comparison. Microfluid. Nanofluid. 18, 673–683 (2015).
Leipert, J. & Tholey, A. Miniaturized sample preparation on a digital microfluidics device for sensitive bottom-up microproteomics of mammalian cells using magnetic beads and mass spectrometry-compatible surfactants. Lab Chip 19, 3490–3498 (2019).
Ho, M. et al. Antifouling properties of pluronic and tetronic surfactants in digital microfluidics. ACS Appl. Mater. Interfaces 15, 6326–6337 (2023).
Acknowledgements
The research of the authors was supported by the China Academy of Engineering, Institute of Land Cooperation Consulting Project 2025-DFZD-39.
Author information
Authors and Affiliations
Contributions
All authors contributed substantially to discussions of the article content. H.M. and J.Y. researched data for the article, D.W. and S.J. wrote the first draft, and A.N., D.W., H.M. and J.Y. reviewed and/or edited the manuscript before submission.
Corresponding authors
Ethics declarations
Diversity statement
The authors acknowledge that papers authored by scholars from historically excluded groups are systematically under-cited. Every attempt has been made to reference relevant papers in a manner that is equitable in terms of racial, ethnic, gender and geographical representation.
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Electrical Engineering thanks Ulf Schlichtmann, who co-reviewed with Yushen Zhang; Jan G. Korvink; and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Alto: https://nicoyalife.com/products-overview/alto-digital-spr/
Cobas ePlex: https://diagnostics.roche.com/global/en/products/systems/cobas-eplex-system-sys-447.html
eProtein Discovery: https://www.nuclera.com/system/
VolTRAX V2: https://nanoporetech.com/document/voltrax-v2-protocol(2019)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, D., Jiang, S., Ma, H. et al. Active-matrix digital microfluidics for high-throughput, precise droplet handling. Nat Rev Electr Eng 3, 46–60 (2026). https://doi.org/10.1038/s44287-025-00230-1
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44287-025-00230-1