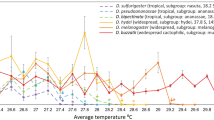
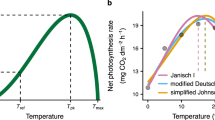
Abstract
Thermal stress reduces fertility and fecundity in animals at temperatures below lethal. Reproductive output is impaired across taxa under diverse heat-exposure regimes, with consequences for individual fitness, population persistence and ecosystem dynamics. This pattern holds across terrestrial and aquatic systems, with implications for conservation, livestock, aquaculture and human health. Yet these sublethal effects remain underrepresented in biodiversity forecasts. In this Review, we synthesize evidence for the biological mechanisms associated with thermally induced declines in fertility and fecundity, and assess how life history and exposure regime can shape thermal sensitivity. Fertility-based thermal limits can predict species distributions and extinction risk better than survival-based measures, albeit tested across a limited taxonomic range. Evolutionary responses to fertility loss under warming seem constrained but increased mutational variation, local adaptation and hybridization might increase fertility resilience. Key research priorities include broader taxonomic evaluation of both evolutionary potential and ecological outcomes under more sophisticated conditions, assessing how fertility is affected when different environmental stressors interact, and understanding how community and ecosystem dynamics will change if fertility-sensitive taxa either shift distributions or go extinct. Recognizing and addressing fertility-based vulnerability is essential for anticipating biodiversity change and designing more effective responses to climate impacts.
Key points
-
Elevated temperatures can decrease reproductive output at temperatures below those causing mortality; hence an understanding of this phenomenon is critical for predicting organismal responses to climate change, including downstream effects on humans
-
Variation in thermal vulnerability within and between species is shaped by how temperature affects the fertilization environment experienced by gametes, the interaction between the magnitude, duration and frequency of heat exposure, and by the relative sensitivity of each sex and life-history stage; additional complexity arises under multiple stressor conditions and from uncertainty as to whether phenotypic plasticity aids or exacerbates responses
-
Heat-impaired reproductive output alters predictions of climate change impacts on species distributions and population persistence; however, evidence for ecological and evolutionary consequences is taxonomically limited, and scenarios involving shifting species interactions and community-level variation in thermal fertility sensitivity are unexplored
-
The potential for adaptive increases in reproductive heat tolerance depends on heritable genetic variation, sex-specific genetic architecture and linked traits, and temperature-dependent DNA mutation; yet evidence for adaptive responses is sparse, limited by a small number of studies in few taxa
-
Recognizing fertility-based vulnerability is essential for assessing the effects of climate change on populations, species, communities and ecosystems, including subsequent ecosystem service effects; improved taxonomic coverage under more complex conditions is required to anticipate ecological and evolutionary consequences and responses across scales
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
IPCC. Climate Change 2023: Synthesis Report. Contribution of Working Groups I, II and III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change [Core Writing Team, H. Lee and J. Romero (eds.)] (IPCC, 2023).
Bennett, J. M. et al. GlobTherm, a global database on thermal tolerances for aquatic and terrestrial organisms. Sci. Data 5, 180022 (2018).
Walsh, B. S. et al. The impact of climate change on fertility. Trends Ecol. Evolution 34, 249–259 (2019).
Ørsted, M., Jørgensen, L. B. & Overgaard, J. Finding the right thermal limit: a framework to reconcile ecological, physiological and methodological aspects of CTmax in ectotherms. J. Exp. Biol. 225, jeb244514 (2022).
Parratt, S. R. et al. Temperatures that sterilize males better match global species distributions than lethal temperatures. Nat. Clim. Change 11, 481–484 (2021).
van Heerwaarden, B. & Sgrò, C. M. Male fertility thermal limits predict vulnerability to climate warming. Nat. Commun. 12, 2214 (2021).
Bretman, A. et al. Systematic approaches to assessing high-temperature limits to fertility in animals. J. Evol. Biol. 37, 471–485 (2024).
Peña, S. T. et al. Tropical summer induces DNA fragmentation in boar spermatozoa: implications for evaluating seasonal infertility. Reprod. Fertil. Dev. 31, 590–601 (2019).
McAfee, A. et al. Vulnerability of honey bee queens to heat-induced loss of fertility. Nat. Sustain. 3, 367–376 (2020).
Xiao, L. et al. Effects of temperature anomaly on sperm quality: a multi-center study of 33,234 men. Heliyon 10, e26765 (2024).
Leith, N. T., Macchiano, A., Moore, M. P. & Fowler-Finn, K. D. Temperature impacts all behavioral interactions during insect and arachnid reproduction. Curr. Opin. Insect Sci. 45, 106–114 (2021).
Neate-Clegg, M. H. C., Tonelli, B. A. & Tingley, M. W. Advances in breeding phenology outpace latitudinal and elevational shifts for North American birds tracking temperature. Nat. Ecol. Evol. 8, 2027–2036 (2024).
Wiley, E. M. & Ridley, A. R. The effects of temperature on offspring provisioning in a cooperative breeder. Anim. Behav. 117, 187–195 (2016).
Dougherty, L. R. et al. A systematic map of studies testing the relationship between temperature and animal reproduction. Ecol. Solut. Evid. 5, e12303 (2024).
Benard, M. F. Warmer winters reduce frog fecundity and shift breeding phenology, which consequently alters larval development and metamorphic timing. Glob. Change Biol. 21, 1058–1065 (2015).
Chamaillé-Jammes, S., Massot, M., Aragón, P. & Clobert, J. Global warming and positive fitness response in mountain populations of common lizards Lacerta vivipara. Glob. Change Biol. 12, 392–402 (2006).
Schou, M. F. et al. Extreme temperatures compromise male and female fertility in a large desert bird. Nat. Commun. 12, 666 (2021).
Campos, F. A. et al. Does climate variability influence the demography of wild primates? Evidence from long-term life-history data in seven species. Glob. Change Biol. 23, 4907–4921 (2017).
Dunham, A. E., Erhart, E. M. & Wright, P. C. Global climate cycles and cyclones: consequences for rainfall patterns and lemur reproduction in southeastern Madagascar: climate effects on lemur demography. Glob. Change Biol. 17, 219–227 (2011).
Van Heezik, Y., Jalme, M. S., Hémon, S. & Seddon, P. Temperature and egg-laying experience influence breeding performance of captive female houbara bustards. J. Avian Biol. 33, 63–70 (2002).
Sturup, M., Baer-Imhoof, B., Nash, D. R., Boomsma, J. J. & Baer, B. When every sperm counts: factors affecting male fertility in the honey bee Apis mellifera. Behav. Ecol. 24, 1192–1198 (2013).
Weaving, H., Terblanche, J. S. & English, S. Heatwaves are detrimental to fertility in the viviparous tsetse fly. Proc. R. Soc. B 291, 20232710 (2024).
Eads, A. R., Kennington, W. J. & Evans, J. P. Interactive effects of ocean warming and acidification on sperm motility and fertilization in the mussel Mytilus galloprovincialis. Mar. Ecol. Prog. Ser. 562, 101–111 (2016).
Armstrong, E. J., Dubousquet, V., Mills, S. C. & Stillman, J. H. Elevated temperature, but not acidification, reduces fertilization success in the small giant clam, Tridacna maxima. Mar. Biol. 167, 8 (2020).
Romano, A. et al. Shrinking body size under climate warming is not associated with selection for smaller individuals in a migratory bird. J. Anim. Ecol. 94, 958–970 (2025).
Klepsatel, P. et al. Variation in thermal performance and reaction norms among populations. Evolution 67, 3573–3587 (2013).
Rezende, E. L., Castañeda, L. E. & Santos, M. Tolerance landscapes in thermal ecology. Funct. Ecol. 28, 799–809 (2014).
Buckley, L. B., Huey, R. B. & Ma, C.-S. How damage, recovery, and repair alter the fitness impacts of thermal stress Integr. Compar. Biol. 65, 1061–1075 (2025).
Meena, A. et al. Life stage-specific effects of heat stress on spermatogenesis and oogenesis in Drosophila melanogaster. J. Therm. Biol. 125, 104001 (2024).
Reshma, R. et al. Transgenerational effects of thermal stress on reproductive physiology of fall armyworm, Spodoptera frugiperda. J. Pest. Sci. 96, 1465–1481 (2023).
Rizzoto, G., Boe-Hansen, G., Klein, C., Thundathil, J. C. & Kastelic, J. P. Acute mild heat stress alters gene expression in testes and reduces sperm quality in mice. Theriogenology 158, 375–381 (2020).
Gandara, A. C. P. & Drummond-Barbosa, D. Warm and cold temperatures have distinct germline stem cell lineage effects during Drosophila oogenesis. Development 149, dev200149 (2022).
Li, J. et al. Effects of chronic heat stress on granulosa cell apoptosis and follicular atresia in mouse ovary. J. Anim. Sci. Biotechnol. 7, 57 (2016).
Rao, M. et al. Transient scrotal hyperthermia affects human sperm DNA integrity, sperm apoptosis, and sperm protein expression. Andrology 4, 1054–1063 (2016).
Roth, Z. & Hansen, P. J. Disruption of nuclear maturation and rearrangement of cytoskeletal elements in bovine oocytes exposed to heat shock during maturation. Reproduction 129, 235–244 (2005).
Yadav, S. K. et al. The thermo-sensitive gene expression signatures of spermatogenesis. Reprod. Biol. Endocrinol. 16, 56 (2018).
Barati, F. et al. Meiotic competence and DNA damage of porcine oocytes exposed to an elevated temperature. Theriogenology 69, 767–772 (2008).
Adriaenssens, B., Damme, R., Seebacher, F. & Wilson, R. S. Sex cells in changing environments: can organisms adjust the physiological function of gametes to different temperatures? Glob. Change Biol. 18, 1797–1803 (2012).
Boni, R., Gallo, A., Montanino, M., Macina, A. & Tosti, E. Dynamic changes in the sperm quality of Mytilus galloprovincialis under continuous thermal stress. Mol. Reprod. Dev. 83, 162–173 (2016).
Sales, K. et al. Experimental heatwaves comprise sperm function and cause transgenerational damage in a model insect. Nat. Commun. 9, 4771 (2018).
Canal Domenech, B. & Fricke, C. Recovery from heat-induced infertility — a study of reproductive tissue responses and fitness consequences in male Drosophila melanogaster. Ecol. Evol. 12, e9563 (2022).
Martinet, B. et al. Mating under climate change: impact of simulated heatwaves on the reproduction of model pollinators. Funct. Ecol. 35, 739–752 (2021).
Kurhanewicz, N. A., Dinwiddie, D., Bush, Z. D. & Libuda, D. E. Elevated temperatures cause transposon-associated DNA damage in C. elegans spermatocytes. Curr. Biol. 30, 5007–5017.e5004 (2020).
Baur, J., Zwoinska, M., Koppik, M., Snook, R. R. & Berger, D. Heat stress reveals a fertility debt owing to postcopulatory sexual selection. Evol. Lett. 8, 101–113 (2024).
Meena, A. et al. Fertility loss and recovery dynamics after repeated heat stress across life stages in male Drosophila melanogaster: patterns and processes. R. Soc. Open Sci. 11, 241082 (2024).
Boni, R. Heat stress, a serious threat to reproductive function in animals and humans. Mol. Reprod. Dev. 86, 1307–1323 (2019).
Zwoinska, M. K., Rodrigues, L. R., Slate, J. & Snook, R. R. Phenotypic responses to and genetic architecture of sterility following exposure to sub-lethal temperature during development. Front. Genet. 11, 573 (2020).
Rodrigues, L. R., Zwoinska, M. K., Wiberg, R. A. W. & Snook, R. R. The genetic basis and adult reproductive consequences of developmental thermal plasticity. J. Anim. Ecol. 91, 1119–1134 (2022).
Cappucci, U. et al. The Hsp70 chaperone is a major player in stress-induced transposable element activation. Proc. Natl Acad. Sci. 116, 17943–17950 (2019).
Mojica, E. A. & Kültz, D. Physiological mechanisms of stress-induced evolution. J. Exp. Biol. 225, jeb243264 (2022).
Bodelón, A., Fablet, M., Siqueira De Oliveira, D., Vieira, C. & García Guerreiro, M. P. Impact of heat stress on transposable element expression and derived small RNAs in Drosophila subobscura. Genome Biol. Evol. 15, evad189 (2023).
Poiani, A. Complexity of seminal fluid: a review. Behav. Ecol. Sociobiol. 60, 289–310 (2006).
Rossi, N., Lopez Juri, G., Chiaraviglio, M. & Cardozo, G. Oviductal fluid counterbalances the negative effect of high temperature on sperm in an ectotherm model. Biol. Open. 10, bio058593 (2021).
Kustra, M. C. et al. Warm waters undermine cryptic female choice. Funct. Ecol. https://doi.org/10.1111/1365-2435.70266 (2026).
Walsh, B. S. et al. Female fruit flies cannot protect stored sperm from high temperature damage. J. Therm. Biol. 105, 103209 (2022).
Gandara, A. C. P. & Drummond-Barbosa, D. Chronic exposure to warm temperature causes low sperm abundance and quality in Drosophila melanogaster. Sci. Rep. 13, 12331 (2023).
Hirao, Y. & Yanagimachi, R. Temperature dependence of sperm–egg fusion and post-fusion events in hamster fertilization. J. Exp. Zool. 205, 433–437 (1978).
Sakatani, M., Yamanaka, K., Balboula, A. Z., Takenouchi, N. & Takahashi, M. Heat stress during in vitro fertilization decreases fertilization success by disrupting anti-polyspermy systems of the oocytes. Mol. Reprod. Dev. 82, 36–47 (2015).
Morimoto, R. I. The heat shock response: systems biology of proteotoxic stress in aging and disease. Cold Spring Harb. Symp. Quant. Biol. 76, 91–99 (2011).
Neuer, A. The role of heat shock proteins in reproduction. Hum. Reprod. Upd. 6, 149–159 (2000).
Gòdia, M. et al. A RNA-seq analysis to describe the boar sperm transcriptome and its seasonal changes. Front. Genet. 10, 299 (2019).
Pei, Y., Wu, Y. & Qin, Y. Effects of chronic heat stress on the expressions of heat shock proteins 60, 70, 90, A2, and HSC70 in the rabbit testis. Cell Stress Chap. 17, 81–87 (2012).
Wang, S.-H. et al. Acute heat stress induces differential gene expressions in the testes of a broiler-type strain of Taiwan country chickens. PLoS ONE 10, e0125816 (2015).
López-Galindo, L. et al. Transcriptomic analysis reveals insights on male infertility in Octopus maya under chronic thermal stress. Front. Physiol. 9, 1920 (2019).
Tang, L. et al. Negative effects of heat stress on ovarian tissue in female rabbit. Front. Veterinary Sci. 9, 9182 (2022).
Fangue, N. A., Hofmeister, M. & Schulte, P. M. Intraspecific variation in thermal tolerance and heat shock protein gene expression in common killifish, Fundulus heteroclitus. J. Exp. Biol. 209, 2859–2872 (2006).
Krebs, R. A. A comparison of Hsp70 expression and thermotolerance in adults and larvae of three Drosophila species. Cell Stress Chap. 4, 243 (1999).
Smithson, C. H., Duncan, E. J., Sait, S. M. & Bretman, A. Expression of heat shock proteins and thermal sensitivity of male fertility across six Drosophila species. J. Evol. Biol. https://doi.org/10.1093/jeb/voaf153 (2025).
Bei, M., Wang, Q., Yu, W., Han, L. & Yu, J. Effects of heat stress on ovarian development and the expression of HSP genes in mice. J. Therm. Biol. 89, 102532 (2020).
Krebs, R. A. & Loeschcke, V. Costs and benefits of activation of the heat-shock response in Drosophila melanogaster. Funct. Ecol. 8, 730 (1994).
Ware-Gilmore, F., Novelo, M., Sgrò, C. M., Hall, M. D. & McGraw, E. A. Assessing the role of family level variation and heat shock gene expression in the thermal stress response of the mosquito Aedes aegypti. Phil. Trans. R. Soc. B 378, 20220011 (2023).
Podrabsky, J. E. & Somero, G. N. Changes in gene expression associated with acclimation to constant tem peratures and fluctuating daily temperatures in an annual killifish Austrofundulus limnaeus. J. Exp. Biol. 207, 2237–2254 (2004).
Velikaneye, B. A. & Kozak, G. M. Epigenomic changes in Ostrinia moths under elevated pupal and adult temperature. Mol. Ecol. 34, e17676 (2025).
Khan, S. & Mishra, R. K. Multigenerational effect of heat stress on the Drosophila melanogaster sperm proteome. J. Prot. Res. 23, 2265–2278 (2024).
Laporta, J., Khatib, H. & Zachut, M. Review: Phenotypic and molecular evidence of inter- and trans-generational effects of heat stress in livestock mammals and humans. Animal 18, 101121 (2024).
Morosinotto, C., Gasparini, C., Pilastro, A., Rosenthal, G. G. & Breedveld, M. C. Cryptic reproductive costs of heatwaves for animal populations. Glob. Change Biol. 31, e70558 (2025).
Canal Domenech, B. & Fricke, C. Developmental heat stress interrupts spermatogenesis inducing early male sterility in Drosophila melanogaster. J. Therm. Biol. 114, 103589 (2023).
Rohmer, C., David, J. R., Moreteau, B. & Joly, D. Heat induced male sterility in Drosophila melanogaster: adaptive genetic variations among geographic populations and role of the Y chromosome. J. Exp. Biol. 207, 2735–2743 (2004).
Walsh, B. S. et al. Plastic responses of survival and fertility following heat stress in pupal and adult Drosophila virilis. Ecol. Evol. 11, 18238–18247 (2021).
Li, H. et al. The fertility recovering from heat stress and interactions of heat shock protein 20 with reproduction-related proteins in Monochamus alternatus. Insect Sci. 32, 2139–2154 (2025).
Iossa, G. Sex-specific differences in thermal fertility limits. Trends Ecol. Evol. 34, 490–492 (2019).
Clutton-Brock, T. H. & Parker, G. A. Potential reproductive rates and the operation of sexual selection. Q. Rev. Biol. 67, 437–456 (1992).
David, J. R. et al. Male sterility at extreme temperatures: a significant but neglected phenomenon for understanding Drosophila climatic adaptations. J. Evol. Biol. 18, 838–846 (2005).
Chatten, A., Grieve, I., Meligoniti, E., Hayward, C. & Pilakouta, N. Predicting the effects of climate change on the fertility of aquatic animals using a meta-analytic approach. Ecol. Lett. 28, e70054 (2025).
Baur, J., Jagusch, D., Michalak, P., Koppik, M. & Berger, D. The mating system affects the temperature sensitivity of male and female fertility. Funct. Ecol. 36, 92–106 (2022).
Parrett, J. M., Kulczak, M. & Szudarek-Trepto, N. Fertility loss under thermal stress: males have lower fertility limits but no evidence of sex differences in sensitivity. Oikos 2024, e10329 (2024).
Ørsted, M., Willot, Q., Olsen, A. K., Kongsgaard, V. & Overgaard, J. Thermal limits of survival and reproduction depend on stress duration: a case study of Drosophila suzukii. Ecol. Lett. 27, e14421 (2024).
Janowitz, S. A. & Fischer, K. Opposing effects of heat stress on male versus female reproductive success in Bicyclus anynana butterflies. J. Therm. Biol. 36, 283–287 (2011).
Santos, M. A. et al. Sex and population differences underlie variation in reproductive success in a warming environment. Evolution 77, 1842–1851 (2023).
Piyaphongkul, J., Pritchard, J. & Bale, J. Heat stress impedes development and lowers fecundity of the brown plant hopper Nilaparvata lugens (Stål). PLoS ONE 7, e47413 (2012).
Kingsolver, J. G. et al. Complex life cycles and the responses of insects to climate change. Integr. Comp. Biol. 51, 719–732 (2011).
Dahlke, F. T., Wohlrab, S., Butzin, M. & Pörtner, H.-O. Thermal bottlenecks in the life cycle define climate vulnerability of fish. Science 369, 65–70 (2020).
Zhang, W., Chang, X.-Q., Hoffmann, A., Zhang, S. & Ma, C.-S. Impact of hot events at different developmental stages of a moth: the closer to adult stage, the less reproductive output. Sci. Rep. 5, 10436 (2015).
Rebolledo, A. P., Sgrò, C. M. & Monro, K. Thermal performance curves reveal shifts in optima, limits, and breadth in early life. J. Exp. Biol. 223, jeb.233254 (2020).
Collin, R. & Chan, K. Y. K. The sea urchin Lytechinus variegatus lives close to the upper thermal limit for early development in a tropical lagoon. Ecol. Evol. 6, 5623–5634 (2016).
Rodrigues, L. R. et al. Fluctuating heat stress during development exposes reproductive costs and putative benefits. J. Anim. Ecol. 91, 391–403 (2022).
Betini, G. S., Wang, X., Avgar, T., Guzzo, M. M. & Fryxell, J. M. Food availability modulates temperature-dependent effects on growth, reproduction, and survival in Daphnia magna. Ecol. Evol. 10, 756–762 (2020).
Donelson, J. M., Munday, P. L., McCormick, M. I., Pankhurst, N. W. & Pankhurst, P. M. Effects of elevated water temperature and food availability on the reproductive performance of a coral reef fish. Mar. Ecol. Prog. Ser. 401, 233–243 (2010).
Costa, E. A. P. D. A., Santos, E. M. D. M., Correia, J. C. & Albuquerque, C. M. R. D. Impact of small variations in temperature and humidity on the reproductive activity and survival of Aedes aegypti (Diptera, Culicidae). Rev. Brasil. Entomol. 54, 488–493 (2010).
Ricupero, M. et al. Combined thermal and insecticidal stresses on the generalist predator Macrolophus pygmaeus. Sci. Total Environ. 729, 138922 (2020).
Harvey, B. P., Gwynn-Jones, D. & Moore, P. J. Meta-analysis reveals complex marine biological responses to the inter active effects of ocean acidification and warming. Ecol. Evol. 3, 1016–1030 (2013).
Parker, L. M., Ross, P. M. & O’Connor, W. A. Comparing the effect of elevated pCO2 and temperature on the fertilization and early development of two species of oysters. Mar. Biol. 157, 2435–2452 (2010).
Pedersen, S. A. & Hanssen, A. E. Ocean acidification ameliorates harmful effects of warming in primary consumer. Ecol. Evol. 8, 396–404 (2018).
Angilletta, M. J. Thermal Adaptation — A Theoretical and Empirical Synthesis (Oxford Univ. Press, 2009).
Suckling, C. C. et al. Adult acclimation to combined temperature and pH stressors significantly enhances reproductive outcomes compared to short-term exposures. J. Anim. Ecol. 84, 773–784 (2015).
Porcelli, D., Gaston, K. J., Butlin, R. K. & Snook, R. R. Local adaptation of reproductive performance during thermal stress. J. Evol. Biol. 30, 422–429 (2017).
Weaving, H., Terblanche, J. S., Pottier, P. & English, S. Meta-analysis reveals weak but pervasive plasticity in insect thermal limits. Nat. Commun. 13, 5292 (2022).
Pottier, P. et al. Developmental plasticity in thermal tolerance: ontogenetic variation, persistence, and future directions. Ecol. Lett. 25, 2245–2268 (2022).
Pottier, P., Burke, S., Drobniak, S. M., Lagisz, M. & Nakagawa, S. Sexual (in)equality? A meta-analysis of sex differences in thermal acclimation capacity across ectotherms. Funct. Ecol. 35, 2663–2678 (2021).
Donelson, J. M., Salinas, S., Munday, P. L. & Shama, L. N. S. Transgenerational plasticity and climate change experiments: where do we go from here? Glob. Change Biol. 24, 13–34 (2018).
Cavieres, G. et al. Rapid within- and transgenerational changes in thermal tolerance and fitness in variable thermal landscapes. Ecol. Evol. 10, 8105–8113 (2020).
Jarrold, M. D. et al. Life-history trade-offs and limitations associated with phenotypic adaptation under future ocean warming and elevated salinity. Phil. Trans. R. Soc. B 374, 20180428 (2019).
Buckley, L. B. et al. Characterizing biological responses to climate variability and extremes to improve biodiversity projections. PLoS Clim. 2, e0000226 (2023).
Rivera-Rincón, N. et al. A comparison of thermal stress response between Drosophila melanogaster and Drosophila pseudoobscura reveals differences between species and sexes. J. Insect Physiol. 153, 104616 (2024).
Dyck, V. A., Hendrichs, J. & Robinson, A. S. (eds) Sterile Insect Technique: Principles and Practice in Area-wide Integrated Pest Management 2nd edn (CRC Press, 2021).
Barclay, H. J. in Sterile Insect Technique: Principles and Practice in Area-wide Integrated Pest Management 2nd edn (eds Dyck, V. A. et al.) 201–244 (CRC Press, 2021).
Shuster, S. M., Pyzyna, B., Mayer, L. P. & Dyer, C. A. The opportunity for sexual selection and the evolution of non-responsiveness to pesticides, sterility inducers and contraceptives. Heliyon 4, e00943 (2018).
Chen, I. C., Hill, J. K., Ohlemüller, R., Roy, D. B. & Thomas, C. D. Rapid range shifts of species associated with high levels of climate warming. Science 333, 1024–1026 (2011).
Arce-Valdés, L. R. & Sánchez-Guillén, R. A. The evolutionary outcomes of climate-change-induced hybridization in insect populations. Curr. Opin. Insect Sci. 54, 100966 (2022).
Kyogoku, D. When does reproductive interference occur? Predictions and data. Popul. Ecol. 62, 196–206 (2020).
Matute, D. R., Novak, C. J. & Coyne, J. A. Temperature-based extrinsic reproductive isolation in two species of Drosophila. Evolution 63, 595–612 (2009).
Coyne, J. A., Kim, S. Y., Chang, A. S., Lachaise, D. & Elwyn, S. Sexual isolation between two sibling species with overlapping ranges Drosophila santomea and Drosophila yakuba. Evolution 56, 2424–2434 (2002).
Servedio, M. R. & Noor, M. A. F. The role of reinforcement in speciation: theory and data. Annu. Rev. Ecol. Evol. Syst. 34, 339–364 (2003).
Matute, D. R. Reinforcement of gametic isolation in Drosophila. PLoS Biol. 8, e1000341 (2010).
Gallegos, C., Hodgins, K. A. & Monro, K. Temperature and sex shape reproductive barriers in a climate change hotspot. Evolution 78, 906–918 (2024).
Taylor, S. A. & Larson, E. L. Insights from genomes into the evolutionary importance and prevalence of hybridization in nature. Nat. Ecol. Evol. 3, 170–177 (2019).
Griffiths, J. S., Kawji, Y. & Kelly, M. W. An experimental test of adaptive introgression in locally adapted populations of splash pool copepods. Mol. Biol. Evol. 38, 1306–1316 (2021).
Poikela, N., Snook, R. R., Kulmuni, J. & Ritchie, M. G. Adaptive introgression improves heat-tolerance despite barriers to gene flow: a case study in Drosophila flavomontana and Drosophila montana. Preprint at bioRxiv https://doi.org/10.64898/2026.01.13.699320 (2026).
Roy, H. E. et al. Curbing the major and growing threats from invasive alien species is urgent and achievable. Nat. Ecol. Evol. 8, 1216–1223 (2024).
Kelley, A. L. The role thermal physiology plays in species invasion. Conserv. Physiol. 2, cou045 (2014).
Green, L., Havenhand, J. N. & Kvarnemo, C. Evidence of rapid adaptive trait change to local salinity in the sperm of an invasive fish. Evol. Appl. 13, 533–544 (2020).
Hoffmann, A. A., Sgrò, C. M. & Van Heerwaarden, B. Testing evolutionary adaptation potential under climate change in invertebrates (mostly Drosophila): findings, limitations and directions. J. Exp. Biol. 226, jeb245749 (2023).
Cammack, K. M., Mesa, H. & Lamberson, W. R. Genetic variation in fertility of heat-stressed male mice. Theriogenology 66, 2195–2201 (2006).
Kovuri, P., Yadav, A. & Sinha, H. Role of genetic architecture in phenotypic plasticity. Trends Genet. 39, 703–714 (2023).
Santos, M. A. et al. No evidence for short-term evolutionary response to a warming environment in Drosophila. Evolution 75, 2816–2829 (2021).
Kellermann, V. & Van Heerwaarden, B. Terrestrial insects and climate change: adaptive responses in key traits. Physiol. Entomol. 44, 99–115 (2019).
Bennett, J. M. et al. The evolution of critical thermal limits of life on Earth. Nat. Commun. 12, 1198 (2021).
Sales, K., Gage, M. J. G. & Vasudeva, R. Experimental evolution reveals that males evolving within warmer thermal regimes improve reproductive performance under heatwave conditions in a model insect. J. Evol. Biol. 37, 1329–1344 (2024).
Walsh, B. & Lynch, M. Evolution and Selection of Quantitative Traits (Oxford Univ. Press, 2018).
Berger, D., Stångberg, J., Grieshop, K., Martinossi-Allibert, I. & Arnqvist, G. Temperature effects on life-history trade-offs, germline maintenance and mutation rate under simulated climate warming. Proc. R. Soc. B 284, 20171721 (2017).
Blanckenhorn, W. U. The evolution of body size: what keeps organisms small? Q. Rev. Biol. 75, 385–407 (2000).
Chakraborty, A. et al. Within-population variation in body size plasticity in response to combined nutritional and thermal stress is partially independent from variation in development time. J. Evol. Biol. 36, 264–279 (2023).
Antunes, M. A. et al. Body size decline during thermal evolution is only detected at mild temperature. Proc. R. Soc. B 291, 20241498 (2024).
Rogell, B. et al. Sex-dependent evolution of life-history traits following adaptation to climate warming. Funct. Ecol. 28, 469–478 (2014).
Gardner, J. L., Peters, A., Kearney, M. R., Joseph, L. & Heinsohn, R. Declining body size: a third universal response to warming? Trends Ecol. Evol. 26, 285–291 (2011).
Berger, D., Stångberg, J., Baur, J. & Walters, R. J. Elevated temperature increases genome-wide selection on de novo mutations. Proc. R. Soc. B 288, 20203094 (2021).
Swings, T. et al. Adaptive tuning of mutation rates allows fast response to lethal stress in Escherichia coli. eLife 6, e22939 (2017).
Hoffmann, A. A., Chown, S. L. & Clusella-Trullas, S. Upper thermal limits in terrestrial ectotherms: how constrained are they? Funct. Ecol. 27, 934–949 (2013).
Duffy, K., Gouhier, T. C. & Ganguly, A. R. Climate-mediated shifts in temperature fluctuations promote extinction risk. Nat. Clim. Change 12, 1037–1044 (2022).
Johnson, C. A., Ren, R. & Buckley, L. B. Temperature sensitivity of fitness components across life cycles drives insect responses to climate change. Am. Nat. 202, 753–766 (2023).
Svensson, E. I. et al. Heritable variation in thermal profiles is associated with reproductive success in the world’s largest bird. Evol. Lett. 8, 200–211 (2024).
Katzenberger, M., Duarte, H., Relyea, R., Beltrán, J. F. & Tejedo, M. Variation in upper thermal tolerance among 19 species from temperate wetlands. J. Therm. Biol. 96, 102856 (2021).
Diamond, S. E. et al. Heat tolerance predicts the importance of species interaction effects as the climate changes. Integr. Comp. Biol. 57, 112–120 (2017).
Toledo, I. M., Dahl, G. E. & De Vries, A. Dairy cattle management and housing for warm environments. Livestock Sci. 255, 104802 (2022).
Beck, H. E. et al. Present and future Köppen-Geiger climate classification maps at 1-km resolution. Sci. Data 5, 180214 (2018).
Gaskins, A. J. et al. Impact of ambient temperature on ovarian reserve. Fertil. Steril. 116, 1052–1060 (2021).
Fast, I. et al. Temperature-responsive miRNAs in Drosophila orchestrate adaptation to different ambient temperatures. RNA 23, 1352–1364 (2017).
Sirotkin, A. V. & Bauer, M. Heat shock proteins in porcine ovary: synthesis, accumulation and regulation by stress and hormones. Cell Stress Chap. 16, 379–387 (2011).
Gruntenko, N. E., Bownes, M., Terashima, J., Sukhanova, M. & Raushenbach, I. Y. Heat stress affects oogenesis differently in wild-type Drosophila virilis and a mutant with altered juvenile hormone and 20-hydroxyecdysone levels. Insect Mol. Biol. 12, 393–404 (2003).
Itami, N., Shirasuna, K., Kuwayama, T. & Iwata, H. Short-term heat stress induces mitochondrial degradation and biogenesis and enhances mitochondrial quality in porcine oocytes. J. Therm. Biol. 74, 256–263 (2018).
Sales, K., Vasudeva, R. & Gage, M. J. G. Fertility and mortality impacts of thermal stress from experimental heatwaves on different life stages and their recovery in a model insect. R. Soc. Open Sci. 8, 201717 (2021).
Li, Y. et al. Differential gene expression in the testes of different murine strains under normal and hyperthermic conditions. J. Androl. 30, 325–337 (2009).
Houston, B. J. et al. Heat exposure induces oxidative stress and DNA damage in the male germ line. Biol. Reprod. 98, 593–606 (2018).
Hurley, L. L., McDiarmid, C. S., Friesen, C. R., Griffith, S. C. & Rowe, M. Experimental heatwaves negatively impact sperm quality in the zebra finch. Proc. R. Soc. B 285, 20172547 (2018).
Angilletta, M. J. Estimating and comparing thermal performance curves. J. Therm. Biol. 31, 541–545 (2006).
Buckley, L. B., Huey, R. B. & Kingsolver, J. G. Asymmetry of thermal sensitivity and the thermal risk of climate change. Glob. Ecol. Biogeogr. 31, 2231–2244 (2022).
Rezende, E. L. & Bozinovic, F. Thermal performance across levels of biological organization. Phil. Trans. R. Soc. B 374, 20180549 (2019).
Jørgensen, L. B., Ørsted, M., Malte, H., Wang, T. & Overgaard, J. Extreme escalation of heat failure rates in ectotherms with global warming. Nature 611, 93–98 (2022).
Deutsch, C. A. et al. Impacts of climate warming on terrestrial ectotherms across latitude. Proc. Natl Acad. Sci. 105, 6668–6672 (2008).
Rezende, E. L., Bozinovic, F., Szilágyi, A. & Santos, M. Predicting temperature mortality and selection in natural Drosophila populations. Science 369, 1242–1245 (2020).
Verberk, W. C. E. P., Hoefnagel, K. N., Peralta-Maraver, I., Floury, M. & Rezende, E. L. Long-term forecast of thermal mortality with climate warming in riverine amphipods. Glob. Change Biol. 29, 5033–5043 (2023).
Arnold, P. A. et al. A framework for modelling thermal load sensitivity across life. Glob. Change Biol. 31, e70315 (2025).
Candolin, U. & Heuschele, J. Is sexual selection beneficial during adaptation to environmental change? Trends Ecol. Evol. 23, 446–452 (2008).
Parrett, J. M. et al. Genomic evidence that a sexually selected trait captures genome-wide variation and facilitates the purging of genetic load. Nat. Ecol. Evol. 6, 1330–1342 (2022).
Emlen, S. T. & Oring, L. W. Ecology, sexual selection, and the evolution of mating systems. Science 197, 215–223 (1977).
Willink, B., Ho, T. A. T. & Svensson, E. I. Ecology and sexual conflict drive the macroevolutionary dynamics of female-limited color polymorphisms in damselflies. Proc. Natl Acad. Sci. 122, e2503400122 (2025).
Perry, J. C. & Rowe, L. Sexual conflict in its ecological setting. Phil. Trans. R. Soc. B 373, 20170418 (2018).
García-Roa, R., Garcia-Gonzalez, F., Noble, D. W. A. & Carazo, P. Temperature as a modulator of sexual selection. Biol. Rev. 95, 1607–1629 (2020).
Walsh, B. S., Mannion, N. L. M., Price, T. A. R. & Parratt, S. R. Sex-specific sterility caused by extreme temperatures is likely to create cryptic changes to the operational sex ratio in Drosophila virilis. Curr. Zool. 67, 341–343 (2020).
Winkler, L., Eilhardt, R. & Janicke, T. Population density affects sexual selection in an insect model. Funct. Ecol. 37, 2734–2747 (2023).
Costa, S. G., Magalhães, S. & Rodrigues, L. R. Multiple mating rescues offspring sex ratio but not productivity in a haplodiploid exposed to developmental heat stress. Funct. Ecol. 37, 1291–1303 (2023).
Cook, P. A., Costello, R. A., Brodie III, E. D. & Formica, V. Population age structure shapes selection on social behaviour in a long-lived insect. Phil. Trans. R. Soc. B 379, 20230331 (2024).
Schacht, R. et al. Adult sex ratios: causes of variation and implications for animal and human societies. Commun. Biol. 5, 1273 (2022).
Svensson, E. I. Eco-evolutionary dynamics of sexual selection and sexual conflict. Funct. Ecol. 33, 60–72 (2019).
Pembury Smith, M. Q. R., Latkova, L. & Snook, R. R. Facultative polyandry under heat stress and the evolutionary potential for climate-driven shifts in mating systems. Heredity https://doi.org/10.1038/s41437-025-00795-5 (2025).
Kotiaho, J. S., Simmons, L. W. & Tomkins, J. L. Towards a resolution of the lek paradox. Nature 410, 684–686 (2001).
Pembury Smith, M. Q. R., Trojmar, D., Gotthard, K., Wiklund, C. & Snook, R. R. Sex-specific variation in thermal sensitivity has multiple negative effects on reproductive trait performance. J. Anim. Ecol. 94, 943–957 (2025).
De Nardo, A. N. et al. Experimental evolution reveals trade-offs between sexual selection and heat tolerance in Drosophila prolongata. Evolution 79, 823–836 (2025).
Koppik, M., Baur, J. & Berger, D. Increased male investment in sperm competition results in reduced maintenance of gametes. PLoS Biol. 21, e3002049 (2023).
Plesnar-Bielak, A., Skrzynecka, A. M., Prokop, Z. M. & Radwan, J. Mating system affects population performance and extinction risk under environmental challenge. Proc. R. Soc. B 279, 4661–4667 (2012).
Parrett, J. M. & Knell, R. J. The effect of sexual selection on adaptation and extinction under increasing temperatures. Proc. R. Soc. B 285, 20180303 (2018).
Iglesias-Carrasco, M. et al. Sexual selection buffers the negative consequences of population fragmentation on adaptive plastic responses to increasing temperatures. Evolution 78, 86–97 (2024).
Acknowledgements
The authors thank the European Society for Evolutionary Biology for funding the Special Topics Network (STN) on The Evolutionary Ecology of Thermal Fertility Limits and the members of the STN, whose work and collaboration on this topic we value immensely. The authors thank the Vetenskapsrådet (grants 2022-03116 and 2018-04598 to R.R.S.), the Natural Environment Research Council (grant NE/X011550/1 to L.D.), the Biotechnology and Biological Research Council (grant BB/W016753/1 to A.B. and R.R.S. and grant UKRI1927 to A.B., R.R.S. and C.F.), and a Heisenberg fellowship from the Deutsche Forschungsgemeinschaft (grant FR 2973/11-1 to C.F.) for funding support.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to all aspects of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Biodiversity thanks Nicola Hemmings and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Glossary
- Acclimation
-
Physiological adjustment to an increase in temperature, in particular when longer-term exposure to a mildly stressful temperature increases tolerance of a subsequent higher temperature.
- Adaptive introgression
-
Where genes from outcrossing to other populations or species improve fitness under novel conditions.
- Antagonistic pleiotropy
-
When a gene/allele has beneficial effects in one/some traits but negative effects for other traits.
- Critical thermal limit
-
The upper (CTmax) or lower (CTmin) temperature at which critical biological function (often measured as motor control or coordinated movement) is lost, or death occurs.
- Critical thermal maximum
-
The highest temperature at which a physiological function is lost (often measured as motor control or coordinated movement), or death occurs.
- Cryptic genetic variation
-
Genetic variation that is normally masked, having little to no effect on the phenotype, but that is exposed under stressful conditions, thereby putatively available to fuel adaptation.
- Evolutionary rescue
-
Selection for evolutionary change enabling individuals to survive under stressful or novel conditions, sufficiently rapidly to save a population from extinction.
- Fecundity
-
Number of eggs or viable offspring produced by an individual. (We note that in the medical literature the definitions of fertility and fecundity can be switched in meaning compared to those used in most non-human contexts. When fecundity is measured as the number of eggs produced, then the effect of heat treatment on offspring survival is not conflated. However, in many taxa, only offspring number is reported. In cases in which a paper reports offspring number, we included it in our review only when the parents, and not offspring, were placed under heat stress. This limits the potential confounding effect of heat stress on survival of offspring that is separate from the effect on the parents).
- Fertility
-
Ability to produce viable offspring. (We note that in the medical literature the definitions of fertility and fecundity can be switched in meaning compared to those used in most non-human contexts).
- Hardening
-
A period of elevated temperature that improves performance under subsequent higher (extreme) temperatures.
- Heat-shock proteins
-
A family of proteins found in virtually all living organisms, produced by cells in response to exposure to high or low temperatures, as part of the heat-shock response; many heat-shock proteins function as chaperone proteins, stabilizing the structure of other proteins that are sensitive to heat stress.
- Heatwave (or heat wave)
-
A meteorological term for a sustained period of abnormally warm weather that lasts for multiple days, measured relative to the normal climate for a given location and time of year. (We note that there is no universally agreed definition for the duration and severity of a heatwave, but as a rough estimate, most definitions require three to five consecutive days of temperatures 5 °C or more above the seasonal average. Many studies attempt to mimic heatwaves to make their temperature manipulations more ecologically realistic).
- Infertile
-
Inability to produce viable offspring; can specifically mean inability to produce gametes.
- Lethal temperature
-
The temperature at which there is, for example, 80% (LT80) or 50% (LT50) mortality in a population or set of experimental organisms, often measured similarly to a lethal dose response.
- Local adaptation
-
Divergent selection leading to local populations having higher relative fitness under local environmental conditions.
- Microclimate
-
A local set of atmospheric conditions that differ from those in the surrounding areas, of interest to thermal biologists because of the potential to provide protection from temperature extremes; for example, animals may be able to shelter from high temperatures in burrows or shaded areas that remain cool.
- Operational sex ratio
-
The ratio of males to females within the mating pool (those animals fertile and ready to mate).
- Phenotypic plasticity
-
The ability of a genome to produce multiple phenotypes depending on the environment.
- Polyspermy
-
Entrance of more than one sperm into the egg (typically lethal).
- Reinforcement
-
Natural selection for reduced reproductive costs for hybrid reproduction increases reproductive isolation.
- Reproductive interference
-
When reproductive isolation is incomplete and heterospecific individuals still engage in mating/reproductive activities, resulting in a fitness reduction for at least one of the interacting partners.
- Reproductive output
-
A holistic term incorporating both fertility and fecundity.
- Secondary contact
-
Populations diverge during a period of geographic isolation but then come back into contact, increasing the opportunity for gene flow.
- Sterile
-
Inability to produce viable offspring; can specifically mean inability to produce gametes.
- Thermal death time
-
The integration of stress intensity (temperature) and exposure time. (We note that through cumulative effects, organisms may suffer more from a mildly stressful high temperature experienced for a long time than from an extreme temperature experienced for a short time. Because the relationship is exponential, a small increase in temperature may lead to a large decrease in tolerance time).
- Thermal fertility limit
-
The temperature at which individuals become (at least temporarily) sterile (scored as a binary 0/1 outcome); as for lethal temperature, it is measured as TFL80 or TFL50, the temperature at which either 80% or 50% of individuals in a population or set of experimental organisms are infertile.
- Thermal performance curve
-
Graphical representation of the relationship between temperature and the value of a biological trait (for example, how activity, climbing ability or offspring production change over a range of temperatures).
- Thermal safety margin
-
The difference between the thermal limit and the maximum ambient temperature experienced in the environment.
- Thermal sensitivity of fertility
-
The relationship between temperature and reproductive output; the number of offspring produced or proxies thereof (such as sperm velocity, follicle number) are a measure of fertility rather than a binary score (0/1) of fertility.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Snook, R.R., Bretman, A., Dougherty, L.R. et al. The consequences of rising temperatures for animal fertility. Nat. Rev. Biodivers. (2026). https://doi.org/10.1038/s44358-026-00142-4
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44358-026-00142-4