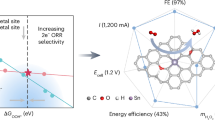
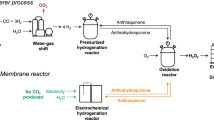
Abstract
Hydrogen peroxide (H2O2) is widely used in household and industrial applications but its synthesis is energy-intensive. An alternative to the traditional anthraquinone process is H2O2 electrosynthesis through a two-electron oxygen-reduction reaction (2e– ORR), using a membrane electrode assembly (MEA). In this Review, we overview the use of the MEA configuration for H2O2 electrosynthesis. A typical MEA cell includes a gas-diffusion electrode, an ion-exchange membrane and a flow field plate that regulate the mass transport of O2, water (reactant) and H2O2 (product) and manage the liquid–gas interactions. Depending on the ion-exchange membrane used, the H2O2 electrosynthesis systems are classified as single-membrane MEA, double-membrane solid-electrolyte MEA and membrane-free. Reducing the cell voltage or increasing the yield can be achieved through anode design strategies, including organic upgrading with low electro-oxidation potential and two-electron water oxidation that enables a theoretical full-cell H2O2 Faradaic efficiency of 200%. MEA-based H2O2 electrosynthesis coupled with downstream thermocatalytic chemical synthesis can produce value-added chemicals such as alcohols and epoxides. Current H2O2 electrosynthesis is approaching industrially relevant current densities (>300 mA cm–2), but long-term stability across diverse electrolysis environments (such as different pH conditions) requires optimization to meet the requirements of commercial applications.
Key points
-
A membrane electrode assembly (MEA) cell enables H2O2 electrosynthesis via the two-electron oxygen reduction reaction.
-
The gas-diffusion electrode, the ion-exchange membrane and the flow field plate regulate the O2, water (protons) and H2O2 (HO2–) mass transport and optimize the liquid–gas interactions.
-
Single-membrane MEA, double-membrane solid-electrolyte MEA and membrane-free cells have different architecture designs that affect the H2O2 production efficiency.
-
Alternative anode oxidation reactions, including organic upgrading and two-electron water oxidation, reduce the cell voltage and enhance the overall yield of the H2O2 electrosynthesis system, respectively.
-
Coupling MEA-based H2O2 electrosynthesis with a thermocatalytic process (such as olefin and alkane oxidation) enables in situ utilization of H2O2 and electrification transformation of traditional chemical synthesis routines.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hage, R. & Lienke, A. Applications of transition-metal catalysts to textile and wood-pulp bleaching. Angew. Chem. Int. Ed. 45, 206–222 (2006).
Brillas, E., Sirés, I. & Oturan, M. A. Electro-Fenton process and related electrochemical technologies based on Fenton’s reaction chemistry. Chem. Rev. 109, 6570–6631 (2009).
Agarwal, N. et al. Aqueous Au–Pd colloids catalyze selective CH4 oxidation to CH3OH with O2 under mild conditions. Science 358, 223–227 (2017).
Flores, M. J., Brandi, R. J., Cassano, A. E. & Labas, M. D. Chemical disinfection with H2O2 — the proposal of a reaction kinetic model. Chem. Eng. J. 198–199, 388–396 (2012).
Mazzucato, M., Facchin, A., Parnigotto, M. & Durante, C. New and revised aspects of the electrochemical synthesis of hydrogen peroxide: from model electrocatalytic systems to scalable materials. ACS Catal. 14, 6369–6403 (2024).
Murray, A. T., Voskian, S., Schreier, M., Hatton, T. A. & Surendranath, Y. Electrosynthesis of hydrogen peroxide by phase-transfer catalysis. Joule 3, 2942–2954 (2019).
Campos-Martin, J. M., Blanco-Brieva, G. & Fierro, J. L. G. Hydrogen peroxide synthesis: an outlook beyond the anthraquinone process. Angew. Chem. Int. Ed. 45, 6962–6984 (2006).
Shi, X. et al. Understanding activity trends in electrochemical water oxidation to form hydrogen peroxide. Nat. Commun. 8, 701 (2017).
Sugiyama, R. & Yamano, T. in Managing Hazardous Reactions and Compounds in Process Chemistry Ch. 5 (eds Pesti, J. A. & Abdel-Magid, A. F.) 135–166 (American Chemical Society, 2014).
Wang, Y. et al. Refining metal-free carbon nanoreactors through electronic and geometric comodification for boosted H2O2 electrosynthesis toward efficient water decontamination. Environ. Sci. Technol. 58, 21893–21903 (2024).
Wen, Y. et al. Photovoltaic-driven stable electrosynthesis of H2O2 in simulated seawater and its disinfection application. Chem. Sci. 15, 18969–18976 (2024).
Jia, Y. et al. Life cycle assessment of hydrogen peroxide produced from mainstream hydrogen sources in China. J. Clean. Prod. 352, 131655 (2022).
Lee, B.-H. et al. Supramolecular tuning of supported metal phthalocyanine catalysts for hydrogen peroxide electrosynthesis. Nat. Catal. 6, 234–243 (2023).
Xia, C., Xia, Y., Zhu, P., Fan, L. & Wang, H. Direct electrosynthesis of pure aqueous H2O2 solutions up to 20% by weight using a solid electrolyte. Science 366, 226–231 (2019).
Siahrostami, S. et al. A review on challenges and successes in atomic-scale design of catalysts for electrochemical synthesis of hydrogen peroxide. ACS Catal. 10, 7495–7511 (2020).
Zhang, X., Xia, Y., Xia, C. & Wang, H. Insights into practical-scale electrochemical H2O2 synthesis. Trends Chem. 2, 942–953 (2020).
Segets, D., Andronescu, C. & Apfel, U.-P. Accelerating CO2 electrochemical conversion towards industrial implementation. Nat. Commun. 14, 7950 (2023).
O’Brien, C. P. et al. CO2 electrolyzers. Chem. Rev. 124, 3648–3693 (2024).
Yu, X. et al. Coverage enhancement accelerates acidic CO2 electrolysis at ampere-level current with high energy and carbon efficiencies. Nat. Commun. 15, 1711 (2024).
Fang, M. et al. Hydrophobic, ultrastable Cuδ+ for robust CO2 electroreduction to C2 products at ampere-current levels. J. Am. Chem. Soc. 145, 11323–11332 (2023).
Kutz, R. B. et al. Sustainion imidazolium-functionalized polymers for carbon dioxide electrolysis. Energy Technol. 5, 929–936 (2017).
Zhang, J. et al. Tantalum-stabilized ruthenium oxide electrocatalysts for industrial water electrolysis. Science 387, 48–55 (2025).
Endrődi, B. et al. Multilayer electrolyzer stack converts carbon dioxide to gas products at high pressure with high efficiency. ACS Energy Lett. 4, 1770–1777 (2019).
Wei, P. et al. Coverage-driven selectivity switch from ethylene to acetate in high-rate CO2/CO electrolysis. Nat. Nanotechnol. 18, 299–306 (2023).
Wen, G. et al. Continuous CO2 electrolysis using a CO2 exsolution-induced flow cell. Nat. Energy 7, 978–988 (2022).
Yang, C. et al. Electro-driven cycling Fenton catalysis through two-dimensional electroresponsive metal–organic frameworks for water purification. Nat. Water 2, 793–802 (2024).
Zhang, J. et al. Suppressing the hydrogen bonding interaction with *OOH toward efficient H2O2 electrosynthesis via remote electronic tuning of Co–N4. Appl. Catal. B 358, 124448 (2024).
Xu, B. et al. Local O2 concentrating boosts the electro-Fenton process for energy-efficient water remediation. Proc. Natl Acad. Sci. USA 121, e2317702121 (2024).
Jiang, Q., Ji, Y., Zheng, T., Li, X. & Xia, C. The nexus of innovation: electrochemically synthesizing H2O2 and Its integration with downstream reactions. ACS Mater. Au 4, 133–147 (2024).
Zou, H. et al. Pushing the limit of atomically dispersed Au catalysts for electrochemical H2O2 production by precise electronic perturbation of the active site. Chem. Catal. 3, 100583 (2023).
Li, H. et al. Scalable neutral H2O2 electrosynthesis by platinum diphosphide nanocrystals by regulating oxygen reduction reaction pathways. Nat. Commun. 11, 3928 (2020).
Xu, Y., Ruban, A. V. & Mavrikakis, M. Adsorption and dissociation of O2 on Pt−Co and Pt−Fe alloys. J. Am. Chem. Soc. 126, 4717–4725 (2004).
Groß, A., Eichler, A., Hafner, J., Mehl, M. J. & Papaconstantopoulos, D. A. Unified picture of the molecular adsorption process: O2/Pt(111). Surf. Sci. 539, L542–L548 (2003).
Yang, X. et al. Tuning two-electron oxygen-reduction pathways for H2O2 electrosynthesis via engineering atomically dispersed single metal site catalysts. Adv. Mater. 34, 2107954 (2022).
He, C. et al. Rational design of oxygen species adsorption on nonnoble metal catalysts for two-electron oxygen reduction. Adv. Energy Mater. 14, 2303233 (2024).
Choi, C. H. et al. Hydrogen peroxide synthesis via enhanced two-electron oxygen reduction pathway on carbon-coated Pt surface. J. Phys. Chem. C 118, 30063–30070 (2014).
Deng, Z., Choi, S. J., Li, G. & Wang, X. Advancing H2O2 electrosynthesis: enhancing electrochemical systems, unveiling emerging applications, and seizing opportunities. Chem. Soc. Rev. 53, 8137–8181 (2024).
Yang, S. et al. Toward the decentralized electrochemical production of H2O2: a focus on the catalysis. ACS Catal. 8, 4064–4081 (2018).
Ge, X. et al. Oxygen reduction in alkaline media: from mechanisms to recent advances of catalysts. ACS Catal. 5, 4643–4667 (2015).
Nie, Y., Li, L. & Wei, Z. Recent advancements in Pt and Pt-free catalysts for oxygen reduction reaction. Chem. Soc. Rev. 44, 2168–2201 (2015).
Siahrostami, S. H2O2 electrosynthesis and emerging applications, challenges, and opportunities: a computational perspective. Chem. Catal. 3, 100568 (2023).
Siahrostami, S. et al. Enabling direct H2O2 production through rational electrocatalyst design. Nat. Mater. 12, 1137–1143 (2013).
Chang, Q. et al. Promoting H2O2 production via 2-electron oxygen reduction by coordinating partially oxidized Pd with defect carbon. Nat. Commun. 11, 2178 (2020).
Yu, Z. et al. Selective and durable H2O2 electrosynthesis catalyst in acid by selenization induced straining and phasing. Nat. Commun. 15, 9346 (2024).
Ross, R. D. et al. Stable pentagonal layered palladium diselenide enables rapid electrosynthesis of hydrogen peroxide. J. Am. Chem. Soc. 146, 15718–15729 (2024).
Xiao, C. et al. Super-coordinated nickel N4Ni1O2 site single‐atom catalyst for selective H2O2 electrosynthesis at high current densities. Angew. Chem. Int. Ed. 61, e202206544 (2022).
Chen, S. et al. Identification of the highly active Co-N4 coordination motif for selective oxygen reduction to hydrogen peroxide. J. Am. Chem. Soc. 144, 14505–14516 (2022).
Chen, S. et al. Tuning proton affinity on Co-N-C atomic interface to disentangle activity-selectivity trade-off in acidic oxygen reduction to H2O2. Angew. Chem. Int. Ed. 64, e202418713 (2025).
Zhang, C. et al. A pentagonal defect-rich metal-free carbon electrocatalyst for boosting acidic O2 reduction to H2O2 production. J. Am. Chem. Soc. 145, 11589–11598 (2023).
Wu, Q. et al. Unveiling the dynamic active site of defective carbon-based electrocatalysts for hydrogen peroxide production. Nat. Commun. 14, 6275 (2023).
Park, J., Nabae, Y., Hayakawa, T. & Kakimoto, M.-a Highly selective two-electron oxygen reduction catalyzed by mesoporous nitrogen-doped carbon. ACS Catal. 4, 3749–3754 (2014).
Adžić, R. R., Tripković, A. V. & Marković, N. M. Structural effects in electrocatalysis: oxidation of formic acid and oxygen reduction on single-crystal electrodes and the effects of foreign metal adatoms. J. Electroanal. Chem. Interfacial Electrochem. 150, 79–88 (1983).
Ricciardulli, T. et al. Effect of Pd coordination and isolation on the catalytic reduction of O2 to H2O2 over PdAu bimetallic nanoparticles. J. Am. Chem. Soc. 143, 5445–5464 (2021).
Hannagan, R. T., Giannakakis, G., Flytzani-Stephanopoulos, M. & Sykes, E. C. H. Single-atom alloy catalysis. Chem. Rev. 120, 12044–12088 (2020).
Verdaguer-Casadevall, A. et al. Trends in the electrochemical synthesis of H2O2: enhancing activity and selectivity by electrocatalytic site engineering. Nano Lett. 14, 1603–1608 (2014).
Choi, C. H. et al. Tuning selectivity of electrochemical reactions by atomically dispersed platinum catalyst. Nat. Commun. 7, 10922 (2016).
Hammer, B. & Nørskov, J. K. Theoretical surface science and catalysis — calculations and concepts. Adv. Catal. 45, 71–129 (2000).
Sun, Y. et al. Activity–selectivity trends in the electrochemical production of hydrogen peroxide over single-site metal–nitrogen–carbon catalysts. J. Am. Chem. Soc. 141, 12372–12381 (2019).
Bonakdarpour, A. et al. Preparation and electrochemical studies of metal–carbon composite catalysts for small-scale electrosynthesis of H2O2. Electrochim. Acta 56, 9074–9081 (2011).
Li, J. et al. Atomically dispersed manganese catalysts for oxygen reduction in proton-exchange membrane fuel cells. Nat. Catal. 1, 935–945 (2018).
Liu, W. et al. A generalized coordination engineering strategy for single-atom catalysts toward efficient hydrogen peroxide electrosynthesis. Adv. Mater. 36, 2406403 (2024).
Li, Y. et al. Single-atom iron catalyst with biomimetic active center to accelerate proton spillover for medical-level electrosynthesis of H2O2 disinfectant. Angew. Chem. Int. Ed. 62, e202306491 (2023).
Chen, Y. et al. Oxygen functional groups regulate cobalt-porphyrin molecular electrocatalyst for acidic H2O2 electrosynthesis at industrial-level current. Angew. Chem. Int. Ed. 63, e202407163 (2024).
Song, X. et al. Graphene-supported single nickel atom catalyst for highly selective and efficient hydrogen peroxide production. ACS Appl. Mater. Interfaces 12, 17519–17527 (2020).
Jiao, Y., Zheng, Y., Jaroniec, M. & Qiao, S. Z. Design of electrocatalysts for oxygen- and hydrogen-involving energy conversion reactions. Chem. Soc. Rev. 44, 2060–2086 (2015).
Xia, Y. et al. Highly active and selective oxygen reduction to H2O2 on boron-doped carbon for high production rates. Nat. Commun. 12, 4225 (2021).
Li, L. et al. Tailoring selectivity of electrochemical hydrogen peroxide generation by tunable pyrrolic–nitrogen–carbon. Adv. Energy Mater. 10, 2000789 (2020).
Long, Y. et al. Tailoring the atomic-local environment of carbon nanotube tips for selective H2O2 electrosynthesis at high current densities. Adv. Mater. 35, 2303905 (2023).
Lu, Z. et al. High-efficiency oxygen reduction to hydrogen peroxide catalysed by oxidized carbon materials. Nat. Catal. 1, 156–162 (2018).
Kim, H. W. et al. Efficient hydrogen peroxide generation using reduced graphene oxide-based oxygen reduction electrocatalysts. Nat. Catal. 1, 282–290 (2018).
Melchionna, M., Fornasiero, P. & Prato, M. The rise of hydrogen peroxide as the main product by metal-free catalysis in oxygen reductions. Adv. Mater. 31, 1802920 (2019).
Tang, C. et al. Topological defects in metal-free nanocarbon for oxygen electrocatalysis. Adv. Mater. 28, 6845–6851 (2016).
Zhai, Q. et al. Recent advances on carbon-based metal-free electrocatalysts for energy and chemical conversions. Adv. Mater. 36, 2405664 (2024).
Chen, S. et al. Defective carbon-based materials for the electrochemical synthesis of hydrogen peroxide. ACS Sustain. Chem. Eng. 6, 311–317 (2018).
Jiang, Y. et al. Significant contribution of intrinsic carbon defects to oxygen reduction activity. ACS Catal. 5, 6707–6712 (2015).
Jing, L. et al. Dual-engineering of porous structure and carbon edge enables highly selective H2O2 electrosynthesis. Adv. Funct. Mater. 33, 2305795 (2023).
Zhu, J. & Mu, S. Defect engineering in carbon-based electrocatalysts: insight into intrinsic carbon defects. Adv. Funct. Mater. 30, 2001097 (2020).
Li, W. et al. Defect engineering for fuel-cell electrocatalysts. Adv. Mater. 32, 1907879 (2020).
Chen, J. et al. Recent progress and perspective of electrochemical CO2 reduction towards C2–C5 products over non-precious metal heterogeneous electrocatalysts. Nano Res. 14, 3188–3207 (2021).
Tian, Q. et al. Hydrogen peroxide electrosynthesis via selective oxygen reduction reactions through interfacial reaction microenvironment engineering. Adv. Mater. 37, 2414490 (2025).
Jung, E. et al. Atomic-level tuning of Co–N–C catalyst for high-performance electrochemical H2O2 production. Nat. Mater. 19, 436–442 (2020).
Wen, Y. et al. Electrochemical reactors for continuous decentralized H2O2 production. Angew. Chem. Int. Ed. 61, e202205972 (2022).
Rabiee, H. et al. Gas diffusion electrodes (GDEs) for electrochemical reduction of carbon dioxide, carbon monoxide, and dinitrogen to value-added products: a review. Energy Environ. Sci. 14, 1959–2008 (2021).
Park, S., Wijaya, D. T., Na, J. & Lee, C. W. Towards the large-scale electrochemical reduction of carbon dioxide. Catalysts 11, 253 (2021).
Fan, W. et al. Rational design of heterogenized molecular phthalocyanine hybrid single-atom electrocatalyst towards two-electron oxygen reduction. Nat. Commun. 14, 1426 (2023).
Hu, X. et al. Boosting industrial-level CO2 electroreduction of N-doped carbon nanofibers with confined tin–nitrogen active sites via accelerating proton transport kinetics. Adv. Funct. Mater. 33, 2208781 (2023).
Chen, J. et al. Promoting electrochemical CO2 reduction via boosting activation of adsorbed intermediates on iron single-atom catalyst. Adv. Funct. Mater. 32, 2110174 (2022).
Chen, J. et al. Accelerated transfer and spillover of carbon monoxide through tandem catalysis for kinetics-boosted ethylene electrosynthesis. Angew. Chem. Int. Ed. 62, e202215406 (2023).
Wu, Y., Rabiee, H., Zhao, X. S., Wang, G. & Jiang, Y. Insights into electrolyte flooding in flexible gas diffusion electrodes for CO2 electrolysis: from mechanisms to effective mitigation strategies. J. Mater. Chem. A 12, 14206–14228 (2024).
Zhao, E., Wang, S., Yu, G. & Wang, Y. Insight into the deterioration mechanism of gas diffusion electrode during long-term electrosynthesis of hydrogen peroxide under industrially relevant current densities. Electrochim. Acta 502, 144835 (2024).
Cui, L. et al. An anti-electrowetting carbon film electrode with self-sustained aeration for industrial H2O2 electrosynthesis. Energy Environ. Sci. 17, 655–667 (2024).
Yan, J., Xia, J. & Wang, K. Low-cell-voltage electrosynthesis of hydrogen peroxide. Green Chem. 26, 5869–5878 (2024).
Joensen, B. Ó. et al. Unveiling transport mechanisms of cesium and water in operando zero-gap CO2 electrolyzers. Joule 8, 1754–1771 (2024).
Xiao, F. et al. Recent advances in electrocatalysts for proton exchange membrane fuel cells and alkaline membrane fuel cells. Adv. Mater. 33, 2006292 (2021).
Zhang, Z. et al. Membrane electrode assembly for electrocatalytic CO2 reduction: principle and application. Angew. Chem. Int. Ed. 62, e202302789 (2023).
Zhang, S.-K. et al. Interfacial electrochemical–chemical reaction coupling for efficient olefin oxidation to glycols. Joule 7, 1887–1901 (2023).
Perry, S. C. et al. Electrochemical synthesis of hydrogen peroxide from water and oxygen. Nat. Rev. Chem. 3, 442–458 (2019).
Ni, C., Xu, W., Deng, N. & Huang, X. Challenges in scaling up H2O2 electrosynthesis: addressing Joule heating effects. J. Clean. Prod. 480, 144066 (2024).
Yang, C. et al. Interfacial O2 accumulation affects microenvironment in carbon-based electrocatalysts for H2O2 production. ACS Energy Lett. 7, 4398–4407 (2022).
Wood, D., Davey, J., Atanassov, P. & Borup, R. PEMFC component characterization and its relationship to mass-transport overpotentials during long-term testing. ECS Trans. 3, 753 (2006).
Weber, A. Z. & Newman, J. Effects of microporous layers in polymer electrolyte fuel cells. J. Electrochem. Soc. 152, A677 (2005).
Kim, B., Hillman, F., Ariyoshi, M., Fujikawa, S. & Kenis, P. J. A. Effects of composition of the micro porous layer and the substrate on performance in the electrochemical reduction of CO2 to CO. J. Power Sources 312, 192–198 (2016).
Bashir, S. M. & Gyenge, E. L. Efficient oxygen reduction to alkaline peroxide at current densities up to 500 mA cm−2 on a gas diffusion electrode with hydrophobic carbon microporous layer: influence of fluid dynamics and reactor operation on scale-up. Chem. Eng. J. 494, 152854 (2024).
Tang, J. et al. Selective hydrogen peroxide conversion tailored by surface, interface, and device engineering. Joule 5, 1432–1461 (2021).
Pérez, J. F. et al. Improving the efficiency of carbon cloth for the electrogeneration of H2O2: role of polytetrafluoroethylene and carbon black loading. Ind. Eng. Chem. Res. 56, 12588–12595 (2017).
Cui, L. et al. Species mass transfer governs the selectivity of gas diffusion electrodes toward H2O2 electrosynthesis. Nat. Commun. 15, 10632 (2024).
Möller, T. et al. The product selectivity zones in gas diffusion electrodes during the electrocatalytic reduction of CO2. Energy Environ. Sci. 14, 5995–6006 (2021).
Uchida, M., Aoyama, Y., Eda, N. & Ohta, A. New preparation method for polymer‐electrolyte fuel cells. J. Electrochem. Soc. 142, 463 (1995).
Peng, X. et al. Electrochemical C–N coupling of CO2 and nitrogenous small molecules for the electrosynthesis of organonitrogen compounds. Chem. Soc. Rev. 52, 2193–2237 (2023).
Mack, F. et al. Influence of the polytetrafluoroethylene content on the performance of high-temperature polymer electrolyte membrane fuel cell electrodes. Int. J. Hydrog. Energy 41, 7475–7483 (2016).
Nwabara, U. O. et al. Binder-focused approaches to improve the stability of cathodes for CO2 electroreduction. ACS Appl. Energy Mater. 4, 5175–5186 (2021).
Park, J. O. et al. Role of binders in high temperature PEMFC electrode. J. Electrochem. Soc. 158, B675 (2011).
Xing, Z., Hu, L., Ripatti, D. S., Hu, X. & Feng, X. Enhancing carbon dioxide gas-diffusion electrolysis by creating a hydrophobic catalyst microenvironment. Nat. Commun. 12, 136 (2021).
Sheng, X., Ge, W., Jiang, H. & Li, C. Engineering the Ni-N-C catalyst microenvironment enabling CO2 electroreduction with nearly 100% CO selectivity in acid. Adv. Mater. 34, 2201295 (2022).
Xu, W. et al. Fast and stable electrochemical production of H2O2 by electrode architecture engineering. ACS Sustain. Chem. Eng. 9, 7120–7129 (2021).
Chao, G., Tang, H., Ju, Q., Li, N. & Geng, K. Fluorinated polybenzimidazole as binders for high-temperature proton exchange membrane fuel cells. J. Power Sources 556, 232473 (2023).
Zhang, Q. et al. Highly efficient electrosynthesis of hydrogen peroxide on a superhydrophobic three-phase interface by natural air diffusion. Nat. Commun. 11, 1731 (2020).
Liang, R. et al. Novel photoelectrocatalytic system of oxygen vacancy-rich black TiO2-x nanocones photoanode and natural air diffusion cathode for efficient water purification and simultaneous H2O2 production. Appl. Catal. B 352, 124042 (2024).
Zhang, Q. et al. Dual strategies to enhance mineralization efficiency in innovative electrochemical advanced oxidation processes using natural air diffusion electrode: Improving both H2O2 production and utilization efficiency. Chem. Eng. J. 413, 127564 (2021).
Zhu, Y. et al. Designing a self-breathing electrode modulated by spatial hydrophobic microenvironments with stabilized H2O2 generation for wastewater treatment. Appl. Catal. B 351, 123973 (2024).
Lippmann, G. Relations entre les phénomènes électriques et capillaires. Ann. Chim. Phys. 5, 494–549 (1875).
Li, M. et al. The role of electrode wettability in electrochemical reduction of carbon dioxide. J. Mater. Chem. A 9, 19369–19409 (2021).
Liu, G. et al. Wetting-regulated gas-involving (photo)electrocatalysis: biomimetics in energy conversion. Chem. Soc. Rev. 50, 10674–10699 (2021).
Kong, Y. et al. Cracks as efficient tools to mitigate flooding in gas diffusion electrodes used for the electrochemical reduction of carbon dioxide. Small Methods 6, 2200369 (2022).
Hu, H. et al. Effective perspiration is essential to uphold the stability of zero-gap MEA-based cathodes used in CO2 electrolysers. J. Mater. Chem. A 11, 5083–5094 (2023).
Jeanty, P. et al. Upscaling and continuous operation of electrochemical CO2 to CO conversion in aqueous solutions on silver gas diffusion electrodes. J. CO2 Util. 24, 454–462 (2018).
Wu, W. et al. Addressing the carbonate issue: electrocatalysts for acidic CO2 reduction reaction. Adv. Mater. 37, 2312894 (2025).
Gu, J. et al. Modulating electric field distribution by alkali cations for CO2 electroreduction in strongly acidic medium. Nat. Catal. 5, 268–276 (2022).
Zhang, X. et al. Electrochemical oxygen reduction to hydrogen peroxide at practical rates in strong acidic media. Nat. Commun. 13, 2880 (2022).
Cao, P. et al. Highly efficient acidic electrosynthesis of hydrogen peroxide at industrial-level current densities promoted by alkali metal cations. Angew. Chem. Int. Ed. 63, e202406452 (2024).
Monteiro, M. C. O. et al. Absence of CO2 electroreduction on copper, gold and silver electrodes without metal cations in solution. Nat. Catal. 4, 654–662 (2021).
Ni, W. et al. Molecular engineering of cation solvation structure for highly selective carbon dioxide electroreduction. Angew. Chem. Int. Ed. 62, e202303233 (2023).
Fan, Y. et al. Mechanistic insights into surfactant-modulated electrode-electrolyte interface for steering H2O2 electrosynthesis. J. Am. Chem. Soc. 146, 7575–7583 (2024).
Zhong, Y. et al. An artificial electrode/electrolyte interface for CO2 electroreduction by cation surfactant self-assembly. Angew. Chem. Int. Ed. 59, 19095–19101 (2020).
Gyenge, E. L. & Oloman, C. W. Influence of surfactants on the electroreduction of oxygen to hydrogen peroxide in acid and alkaline electrolytes. J. Appl. Electrochem. 31, 233–243 (2001).
Nian, Q. et al. Designing electrolyte structure to suppress hydrogen evolution reaction in aqueous batteries. ACS Energy Lett. 6, 2174–2180 (2021).
Ge, W. et al. Dynamically formed surfactant assembly at the electrified electrode–electrolyte interface boosting CO2 electroreduction. J. Am. Chem. Soc. 144, 6613–6622 (2022).
Fang, Y. et al. Boosting hydrogen peroxide electrosynthesis via modulating the interfacial hydrogen‐bond environment. Angew. Chem. Int. Ed. 62, e202304413 (2023).
Chen, Q. et al. Electrocatalytic CO2 reduction to C2+ products in flow cells. Adv. Mater. 36, 2303902 (2023).
Geise, G. M., Hickner, M. A. & Logan, B. E. Ionic resistance and permselectivity tradeoffs in anion exchange membranes. ACS Appl. Mater. Interfaces 5, 10294–10301 (2013).
Olsson, J. S., Pham, T. H. & Jannasch, P. Poly(arylene piperidinium) hydroxide ion exchange membranes: synthesis, alkaline stability, and conductivity. Adv. Funct. Mater. 28, 1702758 (2018).
Ghielmi, A., Vaccarono, P., Troglia, C. & Arcella, V. Proton exchange membranes based on the short-side-chain perfluorinated ionomer. J. Power Sources 145, 108–115 (2005).
Hickner, M. A. & Pivovar, B. S. The chemical and structural nature of proton exchange membrane fuel cell properties. Fuel Cell 5, 213–229 (2005).
Pavel, C. C. et al. Highly efficient platinum group metal free based membrane-electrode assembly for anion exchange membrane water electrolysis. Angew. Chem. Int. Ed. 53, 1378–1381 (2014).
Slade, S., Campbell, S. A., Ralph, T. R. & Walsh, F. C. Ionic conductivity of an extruded Nafion 1100 EW series of membranes. J. Electrochem. Soc. 149, A1556 (2002).
Paddison, S. J. The modeling of molecular structure and ion transport in sulfonic acid based ionomer membranes. J. N. Mater. Electrochem. Syst. 4, 197–208 (2001).
Lees, E. W., Mowbray, B. A. W., Parlane, F. G. L. & Berlinguette, C. P. Gas diffusion electrodes and membranes for CO2 reduction electrolysers. Nat. Rev. Mater. 7, 55–64 (2021).
Wang, J. et al. Poly(aryl piperidinium) membranes and ionomers for hydroxide exchange membrane fuel cells. Nat. Energy 4, 392–398 (2019).
Desousa, R. C., Li, J. H. & Essig, A. Flux ratios and isotope interaction in an ion exchange membrane. Nature 231, 44–45 (1971).
Weekes, D. M., Salvatore, D. A., Reyes, A., Huang, A. & Berlinguette, C. P. Electrolytic CO2 reduction in a flow cell. Acc. Chem. Res. 51, 910–918 (2018).
Habibzadeh, F. et al. Ion exchange membranes in electrochemical CO2 reduction processes. Electrochem. Energy Rev. 6, 26 (2023).
Zhao, Y. et al. Conversion of CO2 to multicarbon products in strong acid by controlling the catalyst microenvironment. Nat. Synth. 2, 403–412 (2023).
Huang, J. E. et al. CO2 electrolysis to multicarbon products in strong acid. Science 372, 1074–1078 (2021).
Ma, W. et al. Electrocatalytic reduction of CO2 to ethylene and ethanol through hydrogen-assisted C–C coupling over fluorine-modified copper. Nat. Catal. 3, 478–487 (2020).
Resasco, J. et al. Promoter effects of alkali metal cations on the electrochemical reduction of carbon dioxide. J. Am. Chem. Soc. 139, 11277–11287 (2017).
Ringe, S. et al. Understanding cation effects in electrochemical CO2 reduction. Energy Environ. Sci. 12, 3001–3014 (2019).
Bohra, D., Chaudhry, J. H., Burdyny, T., Pidko, E. A. & Smith, W. A. Modeling the electrical double layer to understand the reaction environment in a CO2 electrocatalytic system. Energy Environ. Sci. 12, 3380–3389 (2019).
Li, C.-Y. et al. Unconventional interfacial water structure of highly concentrated aqueous electrolytes at negative electrode polarizations. Nat. Commun. 13, 5330 (2022).
Shen, L.-f et al. Interfacial structure of water as a new descriptor of the hydrogen evolution reaction. Angew. Chem. Int. Ed. 59, 22397–22402 (2020).
Wang, X. et al. Site-selective protonation enables efficient carbon monoxide electroreduction to acetate. Nat. Commun. 15, 616 (2024).
Oh, D. et al. Unassisted electrochemical H2O2 production coupled to glycerol oxidation. Nat. Synth. https://doi.org/10.1038/s44160-025-00774-y (2025).
Reyes, A. et al. Managing hydration at the cathode enables efficient CO2 electrolysis at commercially relevant current densities. ACS Energy Lett. 5, 1612–1618 (2020).
Meek, K. M. et al. High-throughput anion exchange membrane characterization at NREL. ECS Trans. 92, 723 (2019).
Tian, Q. et al. Mesoporous carbon spheres with programmable interiors as efficient nanoreactors for H2O2 electrosynthesis. Nat. Commun. 15, 983 (2024).
Bui, J. C., Corpus, K. R. M., Bell, A. T. & Weber, A. Z. On the nature of field-enhanced water dissociation in bipolar membranes. J. Phys. Chem. C 125, 24974–24987 (2021).
Blommaert, M. A. et al. Insights and challenges for applying bipolar membranes in advanced electrochemical energy systems. ACS Energy Lett. 6, 2539–2548 (2021).
Oener, S. Z., Foster, M. J. & Boettcher, S. W. Accelerating water dissociation in bipolar membranes and for electrocatalysis. Science 369, 1099–1103 (2020).
Fan, L. et al. Selective production of ethylene glycol at high rate via cascade catalysis. Nat. Catal. 6, 585–595 (2023).
Balster, J. et al. Tailoring the interface layer of the bipolar membrane. J. Membr. Sci. 365, 389–398 (2010).
Rajesh, A. M., Chakrabarty, T., Prakash, S. & Shahi, V. K. Effects of metal alkoxides on electro-assisted water dissociation across bipolar membranes. Electrochim. Acta 66, 325–331 (2012).
Chen, L., Xu, Q., Oener, S. Z., Fabrizio, K. & Boettcher, S. W. Design principles for water dissociation catalysts in high-performance bipolar membranes. Nat. Commun. 13, 3846 (2022).
Sasmal, S. et al. Materials descriptors for advanced water dissociation catalysts in bipolar membranes. Nat. Mater. 23, 1421–1427 (2024).
Kang, M.-S., Choi, Y.-J. & Moon, S.-H. Effects of inorganic substances on water splitting in ion-exchange membranes. II. Optimal contents of inorganic substances in preparing bipolar membranes. J. Colloid Interface Sci. 273, 533–539 (2004).
Hamilton, P. J. & Pollet, B. G. Polymer electrolyte membrane fuel cell (PEMFC) flow field plate: design, materials and characterisation. Fuel Cell 10, 489–509 (2010).
Lu, S. et al. Mass transfer effect to electrochemical reduction of CO2: electrode, electrocatalyst and electrolyte. J. Energy Storage 52, 104764 (2022).
Gao, G., Obasanjo, C. A., Crane, J. & Dinh, C.-T. Comparative analysis of electrolyzers for electrochemical carbon dioxide conversion. Catal. Today 423, 114284 (2023).
Yuan, S. et al. Flow field design matters for high current density zero-gap CO2 electrolyzers. ACS Energy Lett. 9, 5945–5954 (2024).
Nesbitt, N. T. et al. Liquid–solid boundaries dominate activity of CO2 reduction on gas-diffusion electrodes. ACS Catal. 10, 14093–14106 (2020).
Sauermoser, M., Kizilova, N., Pollet, B. G. & Kjelstrup, S. Flow field patterns for proton exchange membrane fuel cells. Front. Energy Res. 8, 13 (2020).
Wu, D., Jiao, F. & Lu, Q. Progress and understanding of CO2/CO electroreduction in flow electrolyzers. ACS Catal. 12, 12993–13020 (2022).
Maharudrayya, S., Jayanti, S. & Deshpande, A. P. Pressure losses in laminar flow through serpentine channels in fuel cell stacks. J. Power Sources 138, 1–13 (2004).
Liu, Z. et al. Numerical investigation of PEMFC performance based on different multistage serpentine flow field designs. Chem. Eng. J. 500, 156951 (2024).
Wang, Y. et al. Review of flow field designs for polymer electrolyte membrane fuel cells. Energies 16, 4207 (2023).
Hontañón, E., Escudero, M. J., Bautista, C., Garcı́a-Ybarra, P. L. & Daza, L. Optimisation of flow-field in polymer electrolyte membrane fuel cells using computational fluid dynamics techniques. J. Power Sources 86, 363–368 (2000).
Rosli, R. E. et al. A review of high-temperature proton exchange membrane fuel cell (HT-PEMFC) system. Int. J. Hydrogen Energy 42, 9293–9314 (2017).
Zhang, Y., Tao, Y. & Shao, J. Application of porous materials for the flow field in polymer electrolyte membrane fuel cells. J. Power Sources 492, 229664 (2021).
Belsa, B. et al. Materials challenges on the path to gigatonne CO2 electrolysis. Nat. Rev. Mater. 9, 535–549 (2024).
Jiang, H. et al. The anolyte matters: towards highly efficient electrochemical CO2 reduction. Chem. Eng. J. 422, 129923 (2021).
Cheng, F. et al. Accelerated water activation and stabilized metal-organic framework via constructing triangular active-regions for ampere-level current density hydrogen production. Nat. Commun. 13, 6486 (2022).
Li, Y. et al. Accelerated proton-coupled electron transfer via engineering palladium sub-nanoclusters for scalable electrosynthesis of hydrogen peroxide. Angew. Chem. Int. Ed. 64, e202413159 (2024).
Xia, R. et al. Electrochemical oxidation of nitric oxide to concentrated nitric acid with carbon-based catalysts at near-ambient conditions. Nat. Catal. 8, 328–337 (2025).
Li, W., Bonakdarpour, A., Gyenge, E. & Wilkinson, D. P. Drinking water purification by electrosynthesis of hydrogen peroxide in a power-producing PEM fuel cell. ChemSusChem 6, 2137–2143 (2013).
Li, W., Bonakdarpour, A., Gyenge, E. & Wilkinson, D. P. Production of hydrogen peroxide for drinking water treatment in a proton exchange membrane electrolyzer at near-neutral pH. J. Electrochem. Soc. 167, 044502 (2020).
Ge, L. et al. Electrochemical CO2 reduction in membrane-electrode assemblies. Chem 8, 663–692 (2022).
Adler, Z. et al. Hydrogen peroxide electrosynthesis in a strong acidic environment using cationic surfactants. Precis. Chem. 2, 129–137 (2024).
Cao, Y. et al. Surface hydroxide promotes CO2 electrolysis to ethylene in acidic conditions. Nat. Commun. 14, 2387 (2023).
Zhang, M. D., Huang, J. R., Liang, C. P., Chen, X. M. & Liao, P. Q. Continuous electrosynthesis of pure H2O2 solution with medical-grade concentration by a conductive Ni-phthalocyanine-based covalent organic framework. J. Am. Chem. Soc. 146, 31034–31041 (2024).
Lin, S. et al. Electrochemical pilot H2O2 production by solid-state electrolyte reactor: insights from a hybrid catalyst for 2-electron oxygen reduction reaction. Angew. Chem. Int. Ed. 64, e202502144 (2025).
Ruggiero, B. N. et al. Local reaction microenvironment impacts on H2O2 electrosynthesis in a dual membrane electrode assembly solid electrolyte electrolyzer. Chem. Eng. J. 486, 150246 (2024).
Huang, X. et al. Recent advances of electrocatalyst and cell design for hydrogen peroxide production. Nano-Micro Lett. 15, 86 (2023).
Chen, F.-Y. et al. Electrochemical nitrate reduction to ammonia with cation shuttling in a solid electrolyte reactor. Nat. Catal. 7, 1032–1043 (2024).
Salvatore, D. A. et al. Designing anion exchange membranes for CO2 electrolysers. Nat. Energy 6, 339–348 (2021).
Sabri Rawah, B., Albloushi, M. & Li, W. Electro-synthesis of pure aqueous H2O2 on nitrogen-doped carbon in a solid electrolyte flow cell without using anion exchange membrane. Chem. Eng. J. 466, 143282 (2023).
Chen, Z. et al. Development of a reactor with carbon catalysts for modular-scale, low-cost electrochemical generation of H2O2. React. Chem. Eng. 2, 239–245 (2017).
Zhang, Q. et al. Direct insights into the role of epoxy groups on cobalt sites for acidic H2O2 production. Nat. Commun. 11, 4181 (2020).
Jung, E., Shin, H., Hooch Antink, W., Sung, Y.-E. & Hyeon, T. Recent advances in electrochemical oxygen reduction to H2O2: catalyst and cell design. ACS Energy Lett. 5, 1881–1892 (2020).
Xia, C. et al. Confined local oxygen gas promotes electrochemical water oxidation to hydrogen peroxide. Nat. Catal. 3, 125–134 (2020).
Farinazzo Bergamo Dias Martins, P., Plazl, I., Strmcnik, D. & Genorio, B. Prospect of microfluidic devices for on-site electrochemical production of hydrogen peroxide. Curr. Opin. Electrochem. 38, 101223 (2023).
Pérez, J. F. et al. Towards the scale up of a pressurized-jet microfluidic flow-through reactor for cost-effective electro-generation of H2O2. J. Clean. Prod. 211, 1259–1267 (2019).
Esposito, D. V. Membraneless electrolyzers for low-cost hydrogen production in a renewable energy future. Joule 1, 651–658 (2017).
Talabi, O. O., Dorfi, A. E., O’Neil, G. D. & Esposito, D. V. Membraneless electrolyzers for the simultaneous production of acid and base. Chem. Commun. 53, 8006–8009 (2017).
Pang, X., Davis, J. T., Harvey, A. D. & Esposito, D. V. Framework for evaluating the performance limits of membraneless electrolyzers. Energy Environ. Sci. 13, 3663–3678 (2020).
Davis, J. T., Brown, D. E., Pang, X. & Esposito, D. V. High speed video investigation of bubble dynamics and current density distributions in membraneless electrolyzers. J. Electrochem. Soc. 166, F312–F321 (2019).
Bajdich, M., García-Mota, M., Vojvodic, A., Nørskov, J. K. & Bell, A. T. Theoretical investigation of the activity of cobalt oxides for the electrochemical oxidation of water. J. Am. Chem. Soc. 135, 13521–13530 (2013).
Lyu, S. et al. Exceptional catalytic activity of oxygen evolution reaction via two-dimensional graphene multilayer confined metal-organic frameworks. Nat. Commun. 13, 6171 (2022).
Wang, K. et al. Highly active ruthenium sites stabilized by modulating electron-feeding for sustainable acidic oxygen-evolution electrocatalysis. Energy Environ. Sci. 15, 2356–2365 (2022).
Wang, K. et al. Supramolecular engineering of vinylene-linked covalent organic framework – ruthenium oxide hybrids for highly active proton exchange membrane water electrolysis. Adv. Mater. 37, 2417374 (2025).
Meng, Y. et al. Highly active oxygen evolution integrated with efficient CO2 to CO electroreduction. Proc. Natl Acad. Sci. USA 116, 23915–23922 (2019).
Verma, S., Lu, S. & Kenis, P. J. A. Co-electrolysis of CO2 and glycerol as a pathway to carbon chemicals with improved technoeconomics due to low electricity consumption. Nat. Energy 4, 466–474 (2019).
Qi, J. et al. Energy-saving and product-oriented hydrogen peroxide electrosynthesis enabled by electrochemistry pairing and product engineering. Nat. Commun. 14, 6263 (2023).
Na, J. et al. General technoeconomic analysis for electrochemical coproduction coupling carbon dioxide reduction with organic oxidation. Nat. Commun. 10, 5193 (2019).
Yan, D., Mebrahtu, C., Wang, S. & Palkovits, R. Innovative electrochemical strategies for hydrogen production: from electricity input to electricity output. Angew. Chem. Int. Ed. 62, e202214333 (2023).
Mettler, M. S., Vlachos, D. G. & Dauenhauer, P. J. Top ten fundamental challenges of biomass pyrolysis for biofuels. Energy Environ. Sci. 5, 7797–7809 (2012).
Román, A. M., Hasse, J. C., Medlin, J. W. & Holewinski, A. Elucidating acidic electro-oxidation pathways of furfural on platinum. ACS Catal. 9, 10305–10316 (2019).
Sun, Y., Wang, J., Qi, Y., Li, W. & Wang, C. Efficient electrooxidation of 5-hydroxymethylfurfural using co-doped Ni3S2 catalyst: promising for H2 production under industrial-level current density. Adv. Sci. 9, 2200957 (2022).
Zhang, Z. & Deng, K. Recent advances in the catalytic synthesis of 2,5-furandicarboxylic acid and its derivatives. ACS Catal. 5, 6529–6544 (2015).
Li, Y., Wei, X., Chen, L., Shi, J. & He, M. Nickel-molybdenum nitride nanoplate electrocatalysts for concurrent electrolytic hydrogen and formate productions. Nat. Commun. 10, 5335 (2019).
Wu, D., Hao, J., Song, Z., Fu, X.-Z. & Luo, J.-L. All roads lead to Rome: an energy-saving integrated electrocatalytic CO2 reduction system for concurrent value-added formate production. Chem. Eng. J. 412, 127893 (2021).
Lin, L. et al. Engineered 2D transition metal dichalcogenides — a vision of viable hydrogen evolution reaction catalysis. Adv. Energy Mater. 10, 1903870 (2020).
Li, J., Xie, W., Zhou, H., Li, Z. & Shao, M. Techno-economic analysis of electrochemical hydrogen production coupled with alternative oxidation. Chem. Eng. Sci. 298, 120322 (2024).
Wang, J. et al. Electro-reforming polyethylene terephthalate plastic to co-produce valued chemicals and green hydrogen. J. Phys. Chem. Lett. 13, 622–627 (2022).
Wang, J. et al. Electrocatalytic valorization of poly(ethylene terephthalate) plastic and CO2 for simultaneous production of formic acid. ACS Catal. 12, 6722–6728 (2022).
Zhou, H. et al. Electrocatalytic upcycling of polyethylene terephthalate to commodity chemicals and H2 fuel. Nat. Commun. 12, 4679 (2021).
Zhou, Y. et al. Modulating O-H activation of methanol oxidation on nickel-organic frameworks for overall CO2 electrolysis. ACS Catal. 13, 2039–2046 (2023).
Medvedeva, X. V., Medvedev, J. J., Tatarchuk, S. W., Choueiri, R. M. & Klinkova, A. Sustainable at both ends: electrochemical CO2 utilization paired with electrochemical treatment of nitrogenous waste. Green Chem. 22, 4456–4462 (2020).
Hu, C. et al. Near- and long-range electronic modulation of single metal sites to boost CO2 electrocatalytic reduction. Adv. Mater. 35, 2209298 (2023).
Xi, W. et al. Electrochemical CO2 reduction coupled with alternative oxidation reactions: electrocatalysts, electrolytes, and electrolyzers. Appl. Catal. B 341, 123291 (2024).
Li, R., Xiang, K., Peng, Z., Zou, Y. & Wang, S. Recent advances on electrolysis for simultaneous generation of valuable chemicals at both anode and cathode. Adv. Energy Mater. 11, 2102292 (2021).
Chen, H., Liu, Z., Zhou, H., Yang, X. & Lin, W. Screening potential anodic chemistry in lieu of the oxygen evolution reaction in electrolysis systems: the road to practical application. Energy Environ. Sci. 16, 5771–5791 (2023).
Jiang, H. et al. Oxygen-incorporated NiMoP nanotube arrays as efficient bifunctional electrocatalysts for urea-assisted energy-saving hydrogen production in alkaline electrolyte. Adv. Funct. Mater. 31, 2104951 (2021).
Li, C. et al. Local charge distribution engineered by Schottky heterojunctions toward urea electrolysis. Adv. Energy Mater. 8, 1801775 (2018).
Choi, S. et al. Mechanistic investigation of biomass oxidation using nickel oxide nanoparticles in a CO2-saturated electrolyte for paired electrolysis. J. Phys. Chem. Lett. 11, 2941–2948 (2020).
Chen, W. et al. Activated Ni–OH bonds in a catalyst facilitates the nucleophile oxidation reaction. Adv. Mater. 34, 2105320 (2022).
Liu, X. et al. Electrochemical hydrogen generation by oxygen evolution reaction-alternative anodic oxidation reactions. Adv. Energy Sust. Res. 3, 2200005 (2022).
Cao, H., Chen, G., Yan, Y. & Wang, D. Advances in two-electron water oxidation reaction for hydrogen peroxide production: catalyst design and interface engineering. ChemSusChem 18, e202401100 (2024).
Wang, Q. et al. Recent progress on the catalysts and device designs for (photo)electrochemical on-site H2O2 production. Adv. Energy Mater. 13, 2301543 (2023).
Lin, L. et al. Recent advances in electrocatalytic two-electron water oxidation for green H2O2 production. Acta Phys. Chim. Sin. 40, 2305019 (2024).
Izgorodin, A., Izgorodina, E. & MacFarlane, D. R. Low overpotential water oxidation to hydrogen peroxide on a MnOx catalyst. Energy Environ. Sci. 5, 9496–9501 (2012).
Siahrostami, S., Li, G.-L., Viswanathan, V. & Nørskov, J. K. One- or two-electron water oxidation, hydroxyl radical, or H2O2 evolution. J. Phys. Chem. Lett. 8, 1157–1160 (2017).
Park, S. Y. et al. CaSnO3: an electrocatalyst for two-electron water oxidation reaction to form H2O2. ACS Energy Lett. 4, 352–357 (2019).
Zhang, Q. et al. Bifunctional oxygen-defect bismuth catalyst toward concerted production of H2O2 with over 150% cell faradaic efficiency in continuously flowing paired-electrosynthesis system. Adv. Mater. 36, e2408341 (2024).
Bravo-Suárez, J. J. et al. Transient technique for identification of true reaction intermediates: hydroperoxide species in propylene epoxidation on gold/titanosilicate catalysts by X-ray absorption fine structure spectroscopy. J. Phys. Chem. C 112, 1115–1123 (2008).
Lewis, R. J. et al. Highly efficient catalytic production of oximes from ketones using in situ generated H2O2. Science 376, 615–620 (2022).
Guan, M. H. et al. Boosting selective oxidation of ethylene to ethylene glycol assisted by in situ generated H2O2 from O2 electroreduction. Angew. Chem. Int. Ed. 62, e202302466 (2023).
Xu, H., Xu, H. & Cheng, D. Resolving the reaction mechanism for oxidative hydration of ethylene toward ethylene glycol by titanosilicate catalysts. ACS Catal. 12, 9446–9457 (2022).
Liu, X.-C. et al. Efficient conversion of propylene to propylene glycol by coupling H2O2 electrosynthesis and TS-1 thermocatalysis. Electrochem. Commun. 151, 107510 (2023).
Chen, Y. et al. Efficient multicarbon formation in acidic CO2 reduction via tandem electrocatalysis. Nat. Nanotechnol. 19, 311–318 (2023).
Zheng, W. et al. Designs of tandem catalysts and cascade catalytic systems for CO2 upgrading. Angew. Chem. Int. Ed. 62, e202307283 (2023).
Dong, K. et al. Epoxidation of olefins enabled by an electro-organic system. Green Chem. 24, 8264–8269 (2022).
Tian, H. et al. Selective radical oxidation of cyclohexane to adipic acid at electrochemical interfaces. ChemCatChem 15, e202201220 (2022).
Hibino, T. et al. Electrochemical extraction of methanol from lignin under mild conditions. Appl. Catal. B 341, 123328 (2024).
Dummer, N. F. et al. Methane oxidation to methanol. Chem. Rev. 123, 6359–6411 (2022).
Schwach, P., Pan, X. & Bao, X. Direct conversion of methane to value-added chemicals over heterogeneous catalysts: challenges and prospects. Chem. Rev. 117, 8497–8520 (2017).
Xie, J. et al. Highly selective oxidation of methane to methanol at ambient conditions by titanium dioxide-supported iron species. Nat. Catal. 1, 889–896 (2018).
Kim, J. et al. Electro-assisted methane oxidation to formic acid via in-situ cathodically generated H2O2 under ambient conditions. Nat. Commun. 14, 4704 (2023).
Wang, Z., Hu, N., Wang, L., Zhao, H. & Zhao, G. In situ production of hydroxyl radicals via three-electron oxygen reduction: opportunities for water treatment. Angew. Chem. Int. Ed. 63, e202407628 (2024).
Pignatello, J. J., Oliveros, E. & MacKay, A. Advanced oxidation processes for organic contaminant destruction based on the fenton reaction and related chemistry. Crit. Rev. Environ. Sci. Technol. 36, 1–84 (2006).
Song, Y. et al. High-pressure electro-fenton driving CH4 conversion by O2 at room temperature. J. Am. Chem. Soc. 146, 5834–5842 (2024).
Aasberg-Petersen, K., Stenby, E. & Fredenslund, A. Prediction of high-pressure gas solubilities in aqueous mixtures of electrolytes. Ind. Eng. Chem. Res. 30, 2180–2185 (1991).
Brown, S. S., Talukdar, R. K. & Ravishankara, A. R. Reconsideration of the rate constant for the reaction of hydroxyl radicals with nitric acid. J. Phys. Chem. A 103, 3031–3037 (1999).
Sheng, H. et al. Linear paired electrochemical valorization of glycerol enabled by the electro-Fenton process using a stable NiSe2 cathode. Nat. Catal. 5, 716–725 (2022).
Cao, P. et al. High-efficiency electrocatalysis of molecular oxygen toward hydroxyl radicals enabled by an atomically dispersed iron catalyst. Environ. Sci. Technol. 54, 12662–12672 (2020).
Wang, J., Li, S., Qin, Q. & Peng, C. Sustainable and feasible reagent-free electro-Fenton via sequential dual-cathode electrocatalysis. Proc. Natl Acad. Sci. USA 118, e2108573118 (2021).
Pignatello, J. J., Esther, O. & MacKay, A. Advanced oxidation processes for organic contaminant destruction based on the fenton reaction and related chemistry. Crit. Rev. Environ. Sci. Technol. 36, 1–84 (2006).
Xie, L. et al. The strong metal-support interactions induced electrocatalytic three-electron oxygen reduction to hydroxyl radicals for water treatment. Proc. Natl Acad. Sci. USA 120, e2307989120 (2023).
Xiao, F. et al. Selective electrocatalytic reduction of oxygen to hydroxyl radicals via 3-electron pathway with FeCo alloy encapsulated carbon aerogel for fast and complete removing pollutants. Angew. Chem. Int. Ed. 60, 10375–10383 (2021).
Shen, X. et al. In situ-formed PdFe nanoalloy and carbon defects in cathode for synergic reduction–oxidation of chlorinated pollutants in electro-fenton process. Environ. Sci. Technol. 54, 4564–4572 (2020).
Otsuka, K. & Yamanaka, I. Electrochemical cells as reactors for selective oxygenation of hydrocarbons at low temperature. Catal. Today 41, 311–325 (1998).
Li, Y., Miller, C. J., Wu, L. & Waite, T. D. Hydroxyl radical production via a reaction of electrochemically generated hydrogen peroxide and atomic hydrogen: an effective process for contaminant oxidation? Environ. Sci. Technol. 56, 5820–5829 (2022).
Jing, Y. & Chaplin, B. P. Mechanistic study of the validity of using hydroxyl radical probes to characterize electrochemical advanced oxidation processes. Environ. Sci. Technol. 51, 2355–2365 (2017).
Huang, Q. et al. Single-zinc vacancy unlocks high-rate H2O2 electrosynthesis from mixed dioxygen beyond Le Chatelier principle. Nat. Commun. 15, 4157 (2024).
Ding, S. et al. An abnormal size effect enables ampere-level O2 electroreduction to hydrogen peroxide in neutral electrolytes. Energy Environ. Sci. 16, 3363–3372 (2023).
Ni, B. et al. Second-shell N dopants regulate acidic O2 reduction pathways on isolated Pt sites. J. Am. Chem. Soc. 146, 11181–11192 (2024).
US Department of Energy. DOE announces goal to cut solar costs by more than half by 2030. DOE https://www.energy.gov/articles/doe-announces-goal-cut-solar-costs-more-half-2030 (2021).
US Department of Energy. Renewable energy pillar. DOE https://www.energy.gov/eere/renewable-energy#:~:text=Renewable%20Energy%20at%20Home,Incentives%20for%20Renewables%20and%20Efficiency%20 (2024).
US Environmental Protection Agency. eGRID with 2023 data. EPA https://www.epa.gov/egrid/download-data (2025).
Joshi, D. R. & Adhikari, N. An overview on common organic solvents and their toxicity. J. Pharm. Res. Int. 28, 1–18 (2019).
Malaj, E. et al. Organic chemicals jeopardize the health of freshwater ecosystems on the continental scale. Proc. Natl Acad. Sci. USA 111, 9549–9554 (2014).
Jenkin, M. E., Derwent, R. G. & Wallington, T. J. Photochemical ozone creation potentials for volatile organic compounds: rationalization and estimation. Atmos. Environ. 163, 128–137 (2017).
Wang, G. et al. Electrocatalysis for CO2 conversion: from fundamentals to value-added products. Chem. Soc. Rev. 50, 4993–5061 (2021).
Verma, S., Kim, B., Jhong, H.-R. M., Ma, S. & Kenis, P. J. A. A gross-margin model for defining technoeconomic benchmarks in the electroreduction of CO2. ChemSusChem 9, 1972–1979 (2016).
Zhao, E. et al. Optimization and scaling-up of porous solid electrolyte electrochemical reactors for hydrogen peroxide electrosynthesis. Nat. Commun. 16, 3212 (2025).
Grigoropoulou, G., Clark, J. H. & Elings, J. A. Recent developments on the epoxidation of alkenes using hydrogen peroxide as an oxidant. Green Chem. 5, 1–7 (2003).
Lai, W., Qiao, Y., Zhang, J., Lin, Z. & Huang, H. Design strategies for markedly enhancing energy efficiency in the electrocatalytic CO2 reduction reaction. Energy Environ. Sci. 15, 3603–3629 (2022).
Acknowledgements
The authors appreciate financial assistance from the National Natural Science Foundation of China (grants 22425805, U22A20432, 22278364, 22211530045, 22178308 and 22238008), the development projects Jianbing and Lingyan from Zhejiang province (grant 2023C01226), the National Key Research and Development Program of China (grant 2022YFB4002100), the Fundamental Research Funds for the Central Universities (grant 226-2024-00060), the Science Foundation of Donghai Laboratory (grant DH-2022ZY0009) and the Key Technology Breakthrough Program of Ningbo ‘Science and Innovation Yongjiang 2035’ (grant 2024H024). L.D. thanks the Australian Research Council for partial financial support (grant CE230100032).
Author information
Authors and Affiliations
Contributions
All authors contributed substantially to discussion of the content of this paper. X.Z. and Y.H. drafted the initial manuscript. All authors were involved in reviewing and editing the manuscript prior to submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Clean Technology thanks Elod Gyenge, who co-reviewed with Shahid Bashir; and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, X., Yang, X., Su, B. et al. Membrane electrode assembly for hydrogen peroxide electrosynthesis. Nat. Rev. Clean Technol. 1, 413–431 (2025). https://doi.org/10.1038/s44359-025-00069-7
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44359-025-00069-7