Abstract
Estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor-2 (HER2) status provide clinical utility in guiding therapeutic decision-making in metastatic breast cancer (BC). Increasing data have shown substantial differences between the receptor profiles of primary BCs and their paired metastases. In this study, we provide a large single center cohort to assess the frequency of receptor conversion in metastatic BC. The overall discordant rates were 18.3%, 40.3%, and 13.7% for ER, PR, and HER2, respectively. The discordance was significantly higher for PR when compared with ER and HER2. The conversion occurred significantly as a switch from positive to negative receptor status when compared with that from negative to positive for all three receptors. Semiquantitative analyses revealed a significantly decreased expression of both ER (25%) and PR (57%) in the metastases. There was a higher rate of PR discordance in bone metastases when comparing to other common organs of relapse. Furthermore, in the subset of patients with a single primary and multiple distant metastases, the discordant rates among the distant sites were 27.5%, 39.4%, and 14.3% for ER, PR, and HER2, respectively. A positive ER status, be it in primary or metastatic BC, was associated with a prolonged metastasis-free survival when compared with ER-negative primary tumors without conversion. Furthermore, a positive ER status in metastatic BC regardless of primary was associated with a superior overall survival when compared with an ER-negative tumor without conversion. Thus, receptor conversion is a frequent event in the course of BC progression, and can also be seen between different metastatic sites. Moreover, some conversions are of prognostic significance. The findings may reflect tumor heterogeneity, sampling or treatment effect, but may also indicate alteration in tumor biology. Repeat biomarker testing is warranted in making appropriate treatment plans in the pursuit of precision medicine.
Similar content being viewed by others
Introduction
Breast cancer (BC) is the most common cancer and the second leading cause of cancer death in women in the United States, with projected 276,480 new cases and 42,170 cancer deaths in 2020, respectively [1]. BC mortality is largely the consequence of distant organ metastases that are resistant to systemic therapy. Despite the recent advancements in early detection and aggressive treatment, distant metastasis remains a clinical challenge. The risk of distant recurrence ranges from 10 to 41%, depending on the pathologic characteristics and nodal status of the original tumor [2, 3]. Moreover, 6–10% of patients are diagnosed with metastatic BC at initial presentation, also known as de novo metastatic BC [4].
Systemic treatment for primary and metastatic BC is primarily based on the status of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor-2 (HER2). In metastatic BC, endocrine or HER2-targeted therapy decision-making has been historically directed at the biomarker status of the primary tumor, as routine biopsies of metastatic lesions and reassessment of receptor status was not part of the standard workup. However, increasing data have shown substantial differences between the receptor profiles of primary BCs and their distant metastases [5,6,7,8,9], and the discrepancy is also seen at the genomic and transcriptomic level [7]. The change (gain or loss) of hormone receptor or HER2 status between primary and metastatic tumors within the same patient, also known as “receptor conversion”, could potentially lead to inappropriate endocrine or HER2-targeted therapies in patients with metastatic BC. Therefore, the current National Comprehensive Cancer Network Panel and American Society of Clinical Oncology (ASCO) Clinical Practice Guideline recommends biopsy confirmation of disease process and retesting biomarkers in patients with accessible metastases, and to preferentially use the receptor status of the metastasis to direct therapy if supported by the clinical scenario and patient’s goals for care [10, 11]. In addition, the European School of Oncology International Consensus Guidelines for advanced BC also recommend to biopsy a metastatic lesion to confirm the diagnosis, particularly when metastasis is diagnosed for the first time [12].
There has been considerable debate about the incidence of receptor conversion. The reported rates for discordance between primary and metastatic BCs varied from 0 to 67% for ER, 0 to 78% for PR, and 0 to 44% for HER2 [5,6,7,8,9]. There are several inherent limitations in the earlier studies, of which small sample size is clearly the most notable shortcoming [5]. Other potential limitations include using a ligand-binding assay, inclusion of decalcified specimens from bone metastases, and possible sampling bias introduced by use of tissue arrays as described in an earlier study [13]. Moreover, the discrepancy of receptor status across different distant sites of relapse in the patients with multiple metastases has only been rarely examined [14]. In the present study, we assessed the frequency of receptor conversions and performed semiquantitative analyses for hormonal receptors using 390 paired primary and metastatic breast carcinomas, the largest cohort of its kind from a single institution. The association between receptor conversion and prognostic outcomes in these patients was also analyzed.
Materials and methods
After approval by the Institutional Review Board, the Surgical Pathology database of the authors’ institution was searched to identify matched primary and metastatic BC cases with receptor studies available between 1998 and 2019. Locoregional recurrence, de novo metastatic BCs, and metastases to ipsilateral regional lymph nodes were excluded. Patients who did not receive systemic therapy were not included. Given that decalcification may potentially affect the analyses of receptor studies, bone biopsy specimens were not decalcified if a possible metastasis was clinically indicated per the institutional protocol. This led to a total of 390 paired primary and metastatic breast carcinomas meeting the inclusion criteria, including 40 patients with metastases to more than one distant sites. Five patients had more than one ipsilateral primary tumor or bilateral carcinomas that showed different receptor profiles, the receptor status in the metastasis was not regarded as discordant if it matched any of the primaries in these patients.
The tissue specimens were fixed for variable times but were typically within the 6–72 h window before commencing the immunohistochemistry protocol. The deparaffinized sections were incubated in sodium citrate buffer for 60 min at 100 °C for antigen retrieval. Hydrogen peroxide was applied for 4 min at 37 °C to block endogenous peroxidase. After rinsing, ER (clone 6F11 [until 2008] and clone SP1 [after 2008], prediluted; Ventana), PR (clone 1A6 [until 2008] and clone 1E2 [after 2008], prediluted; Ventana), or HER2 (clone SP3, 1:100, Thermo Scientific [until 2016] and clone 4B5, prediluted, Ventana [after 2016]) antibody was then applied for 36 min at 37 °C, followed by a biotinylated secondary antibody for 8 min. Sections were then rinsed, visualized with diaminobenzoic acid, counterstained with hematoxylin, and then taken offline to be dehydrated and coverslipped. ER and PR interpretation was performed following the American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP) guideline recommendations [15]. The staining intensity was scored into 0 (negative), 1+ (weak), 2+ (moderate), or 3+ (strong). Semiquantitative ER and PR protein expression was evaluated, when available, using an H-score as determined by multiplying the intensity by the percentage of positive staining cells (extracted from the original pathology reports or by review of slides), giving a range of 0–300. HER2 overexpression/gene amplification was defined as either a 3+ immunohistochemistry score (uniform and intense membrane staining of >30% of tumor cells) or as a positive in situ hybridization result following the ASCO/CAP guideline recommendations [16,17,18].
The categorical data obtained were statistically evaluated using either the χ2-test, while continuous data were evaluated using the Student t test. Metastasis-free survival (calculated from the date of diagnosis to the date of metastasis) and overall survival (from the date of diagnosis to the date of death) were mapped on Kaplan–Meier curves. Patients who survived or were lost to follow-up were considered as censored data in the analysis. The univariate log-rank test was used to compare groups. A P value of <0.05 was considered statistically significant. The statistical analyses were conducted using the GraphPad Prism 6.0 software and VassarStats (http://vassarstats.net/).
Results
There were a total of 390 paired primary and metastatic breast carcinomas from 348 patients with ER and/or HER2 studies available in the study period. Forty patients had more than one distant metastases, ranging from 2 to 5 sites. Collectively there were 387, 345, and 321 paired tumors meeting the inclusion criteria for ER, PR, and HER2 analyses, respectively. The most common organs of relapse were bone, liver, lung (including pleura), and brain in keeping with early studies [19, 20]. Other distant sites of metastasis largely included skin/soft tissue, abdominal and retroperitoneal locations (i.e., gastrointestinal organs and adrenal glands), gynecologic organs, head and neck, and distant (i.e., thoracic) lymph nodes.
The overall discordant rate for ER was 18.3% (n = 71). The conversion occurred significantly as a switch from positive to negative receptor status (61/295, 20.7%) when compared with that from negative to positive (10/92, 10.9%; P = 0.04) (Table 1). Semiquantitative analyses using the Student’s t test revealed a significantly decreased expression (25%) of ER in the metastases (mean H-score and 95% confidence interval: 129 ± 17) when compared to the primary tumors (172 ± 16; P < 0.0001) (Fig. 1).
Dot plot (top panel) and line graph (bottom panel) of ER and PR expression in paired primary and metastatic breast cancer. There was a significantly decreased expression of both ER and PR in the metastases (mean H-score and 95% confidence interval: 172 ± 16 vs. 129 ± 17, P < 0.0001 and 77 ± 13 vs. 33 ± 9, P < 0.0001, respectively).
There was a significantly higher conversion rate of PR (139/345, 40.3%) when compared with that of ER (P < 0.0001). Similar to ER, PR converted twice as frequent from positive to negative than vice versa (107/214, 50% vs. 32/131, 24.4%; P < 0.0001). Furthermore, there was an average of 57% reduction of PR expression in metastatic tumors when compared to the primaries (77 ± 13 vs. 33 ± 9; P < 0.0001) (Fig. 1).
Receptor conversion for HER2 occurred in 13.7% of metastatic carcinomas (44/320). Once again, a HER2 status change from positive to negative was much more common than vice versa (25/87, 28.7% vs. 19/234, 8.1%; P < 0.0001). The overall conversion rate for HER2 was significantly lower than that of PR (P < 0.0001) but not different from that of ER (P = 0.15).
To further elucidate if the receptor conversion demonstrates metastatic site-specific differences, we next analyzed the receptor conversion at the most common organs of relapse, including bone, liver, lung/pleura, and brain. To that end, there was a slightly higher yet insignificant conversion rate for ER in brain metastases when comparing to other organs. Approximately a half of bone metastases demonstrated PR discordance, although it did not reach a statistical significance when compared with other sites. A similar conversion rate for HER2 was observed across all these organs (Table 2).
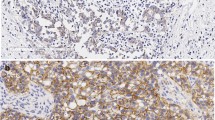
We next assessed to what extent the receptor status differs between metastatic sites. To that end, the discrepancy of receptor status was examined in the patients with single primary breast carcinomas and multiple distant metastases. The discordant rates between the distant sites of relapse were 27.5% (11/40), 39.4% (13/33) and 14.3% (5/35) for ER, PR and HER2, respectively (Table 3). Interestingly, discordance in the receptor status between separate lesions within the same organ was also observed, as exemplified in Fig. 2.
Biopsies of two liver lesions (top and bottom panels) from the same patient demonstrated metastatic breast carcinomas with similar histomorphology. While the primary tumor had an ER+/PR+/HER2+ phenotype, the two metastatic lesions demonstrated ER+/PR−/HER2− and ER+ (low)/PR+/HER2+ phenotypes, respectively. Original magnifications ×100 (H&E, ER, and PR) and ×400 (HER2 Dual ISH).
We next turned to analyze the association between receptor conversion and time to metastasis, if any. With regard to ER status, the median survival times for positive to positive (+/+), positive to negative (+/−), negative to positive (−/+), and negative to negative (−/−) conversion were 1457, 1099, 2860, and 790 days, respectively. The −/− ER status in primary and metastatic tumors was associated with a significantly worse metastasis-free survival when compared with +/+ (hazard ratio [95% confidence interval] 0.5667 [0.3643, 0.6697]; P < 0.0001), +/− (0.6013 [0.4136, 0.8090]; P = 0.002) and −/+ (0.4798 [0.3118, 0.8686]; P = 0.02) ER statuses, respectively (Fig. 3). The median survival times for +/+, +/−, −/+ and −/− PR conversion were 1417, 1223, 1332 and 879 days, respectively. Similarly, the +/+ (0.6485 [0.4647, 0.8373]; P = 0.002) and +/− (0.7288 [0.5360, 0.9532]; P = 0.038) PR conversion were both associated with a superior metastasis-free survival when compared with the −/− PR status. A trend of favorable prognosis was associated with −/+ PR conversion although it did not reach statistical significance. The median survival times for +/+, +/−, −/+, and −/− HER2 conversion were 1064, 1076, 1587, and 1063 days, respectively. No significant association for HER2 conversion was observed. As expected, a +/+ HER2 status was associated with a worse metastasis-free survival when compared with the −/− HER2 status (1.339 [1.012, 1.907]; P = 0.045) (Fig. 3).
Comparison of overall survival was also performed between patients with and without receptor conversion. To that end, the median survival times for +/+, +/−, −/+, and −/− ER statuses were 4177, 2791, 4050, and 2420 days, respectively. The −/− ER status was associated with a significantly inferior overall survival when compared with the +/+ (0.4349 [0.1902, 0.5585]; P < 0.0001) and −/+ (0.2534 [0.1646, 0.9067]; P = 0.035) ER statuses. The median survival times for +/+, +/−, −/+, and −/− PR statuses were 5877, 3196, 3636, and 2859 days, respectively. The +/+ PR status was significantly associated with a favorable overall survival when compared to the +/− (0.5143 [0.3128, 0.811]); P = 0.005) and −/− (0.4581 [0.2595, 0.7077]; P = 0.001) PR statuses, respectively. The median survival times for +/+, +/−, −/+, and −/− HER2 statuses were 3414, 3464, 9790, and 3636 days, respectively. No significant difference was found between any HER2 conversion statuses although a trend of prolonged overall survival was seen with −/+ the HER2 conversion (Fig. 3).
Discussion
The discordance of receptor status between primary breast carcinomas and their matched metastases is a phenomenon initially recognized in the late 1970s and has been subsequently further studied [21]. Yet, early investigations have resulted in considerable debates as to the incidence of discordance, as there have been highly variable conversion rates, ranging from 0 to 78% in the literature [5,6,7,8,9]. Small sample size is thought to be the principle significant influential factor for such extent of variations. A systemic review analysis revealed that 81% (22/27), 79% (19/24), and 86% (30/35) of previous studies contained less than 100 cases for ER, PR, and HER2 conversion, respectively [5]. Most of the larger-scaled studies were pooled analyses of published studies. In the current study, we presented the largest cohort from a single academic center in order to provide additional data and more meaningful insights into this critical matter. The advantage of single institution analyses is the ability to provide more detailed and specific data that is usually not possible with a large pooled literature review. The overall discordant rates between primary and metastatic breast carcinomas in this cohort were ~18%, 40%, and 14% for ER, PR and HER2, respectively. Moreover, discrepant receptor profiles across different sites of metastases were found in a significant proportion of patients with single primary carcinomas and multi-organ relapses.
A number of other factors may have attributed to the extent of variation in the literature. First, the heterogeneity of tissue sampling is an inherent bias in clinical practice. The receptor studies can be performed on a core biopsy, fine-needle aspiration or surgical resection for either primary or metastatic carcinomas, thus resulting in a false-negative receptor status, as intratumoral heterogeneity of receptor expression is well-characterized in BC. Further, some previous studies included regional lymph node metastases and local relapses [22]. Given that a clinically significant difference has been perceived between nodal metastasis/local relapse and distant metastases [9, 23], the former was not included in the present study.
Second, pre-analytical and analytical alterations may be an important contributor to such variation. A number of previous studies were performed when dextran-coated charcoal assay was utilized for hormonal receptor studies [9]. Immunohistochemistry was introduced for ER and PR analysis and approved for routine clinical practice in the late 1990’s [24]. We thus collected cases from 1998 to minimize the variation potentially caused by such assay methods. Decalcification of bone specimens using strong acidic solutions may have an impact on immunohistochemical analysis, and also precludes the specimen from being evaluated for HER2 by in situ hybridization analysis. To prevent such problems, bone biopsy specimens were not decalcified if a possible metastasis was clinically suspected per our institutional protocol. Furthermore, any change of antibodies and testing platforms over the study period should have resulted in more sensitive and consistent testing results in all studies including ours. Moreover, possible inter-assay variability is another potential confounder with such tests, especially before the establishment of guidelines. All assays in our laboratory were performed with appropriately reactive positive and negative controls. This, along with our participation in the CAP Survey and CAP Performance Improvement Program, may have helped to maintain the quality assurance and quality control systems and to minimize any variations over the time.
Third, the guidelines for interpretation of ER, PR, and HER2 have changed over the years. A 10% cutoff was used for ER and PR interpretation in the early years whereas this cutoff was revised to 1% following the ASCO/CAP guideline recommendations in 2010 [15]. To that end, semiquantitative analyses for ER and PR using an H-score were performed for all cases included in the present study to overcome, at least in part, the undesirable effect caused by the difference in the cutoff values. Further, wide implementation of the ASCO/CAP guidelines for HER2 testing was not available until 2007 [16], and the testing criteria defining HER2-positive status was revised in 2013 [17]. While this revision may result in a change of HER2 status, reclassification of the HER2 status from negative to positive reportedly occurred in only ~1% of patients [25]. Moreover, HER2 analyses by immunohistochemistry and in situ hybridization have been simultaneously performed at the authors’ institution since 2008 to further minimize the potential discrepancy between the testing methods. Thus, the criteria change was thought to have a minimal, if any, impact in the receptor conversion. Furthermore, the 2018 ASCO/CAP HER2 guideline focused update was thought to have minimal, if any, impact to the analyses in this cohort [18].
While tissue sampling and assay techniques may contribute, to some extent, to the variation in the testing results, discordance rates reportedly did not statistically differ between the testing methodologies, thus receptor conversion in metastatic BC has been thought a true biological phenomenon and not solely the result of limited accuracy of the receptor assays per se [5, 26]. The majority of conversions were from a positive to negative status, thus may be largely attributed to selective killing of BC cells overexpressing ER or HER2 by endocrine or HER2-targeted therapy. However, the reverse does occur as confirmed by some studies including ours, thus reflecting the complex biology of receptor conversion in metastatic BC. Metastasis as part of tumor progression is a clonally selective process, a hypothesis supported by experimental and clinical studies [27]. Direct proof has been provided by early studies that genetically different individual cells coexist within a tumor, differing in their metastatic capacity, and that the subpopulations within a tumor may have widely variant metastatic proclivities and expression profiles [28, 29]. This theory has been further supported by the fact that receptor status were unstable throughout tumor progression, as demonstrated by the discordance in receptor profiles among different sites of distant relapse in a subset of patients with multi-organ metastases. Such discordances in the present study were 27.5%, 39.4%, and 14.3% for ER, PR, and HER2, respectively, and were also perceived in separate lesions within the same organ. An early study showed an alteration of ER, PR, and HER2 status in 33.6%, 32.0%, and 15.7% of patients with multiple consecutive relapses, respectively, although a significant proportion of the cohort were local relapses [22]. Another previous study using exclusively distant relapses similar to our cohort showed discordant rates of 10.9%, 30.9%, and 3.6% for ER, PR, and HER2 status, respectively, across different distant metastases [14].
Another important finding in this cohort was that the extent of decreased expression was more dramatic for PR levels when compared to ER (57% vs. 25%), thus it is not surprising that PR conversion from positive to negative was significantly more frequent than that of ER, an observation in keeping with a large body of previous data using different cutoffs [5]. As an ER-dependent gene product, PR loss in the metastasis may be a key hallmark of response failure to endocrine therapy [8, 30], thus necessitating further investigation.
Location-specific discordance between primary BC and distant metastasis is of substantial clinical interest. There have been a number of studies in this regard, mostly using a small number of cases, with variable results. One study including 233 patients with non-bone metastases showed that the discordance was more frequent in brain, liver, and gastrointestinal metastases, especially for PR [13]. A pooled analysis demonstrated a significant higher ER discordant rate in bone relapses when compared with brain metastases, and a significant higher PR discordant rate in bone and liver relapses when compared with brain metastases, although those studies used different methods (including tissue microarrays) and cutoffs [5]. A significant difference in discordant rate for ER or PR between most common distant sites of relapse was not observed in our cohort, although a trend of higher PR discordance in bone relapse was seen. Furthermore, no significantly different HER2 conversion rates between metastatic sites were identified, in keeping with most early studies.
The association between receptor conversion and time to metastasis is of further interest. Our findings showed that a positive ER status, be it in primary or metastatic tumor (+/+, +/− or −/+ conversion), was associated with a prolonged metastasis-free survival when compared with ER-negative tumors without conversion (−/−). A similar observation was also found for PR status, with exception of negative to positive conversion, although such a trend was also seen. No significant association for HER2 conversion was observed. With regard to the relationship between receptor conversion and overall survival, a positive ER status in metastatic tumor regardless of primary (+/+ or −/+ conversion) was associated with a superior overall survival when compared with an ER-negative tumor without conversion (−/−). Conversely, a negative PR status in the metastatic tumor regardless of primary (+/− or −/− conversion) was associated with a worse prognosis when compared with a PR-positive tumor without conversion (+/+). These findings further reflect the biologic mechanism of PR acting as a surrogate marker for functional ER activation largely influencing BC cell growth in the metastatic setting [31]. Systemic therapy may have a confounding effect in comparing survival outcomes in the different receptor conversion categories, an inherent nature of such studies. Nonetheless, all patients in this cohort received standard of care treatment at the time of diagnosis,
In summary, receptor conversion is a frequent event in the course of BC progression, and can be a positive to negative or negative to positive conversion. Both conversions are clinically relevant, as the patient may be at risk of ineffective treatment or miss the potentially effective targeted therapy in case of loss or gain of receptor in the metastasis, respectively. Moreover, discordance in receptor status between different metastatic sites was observed in a significant proportion of metastatic BC patients. Despite the mechanism of receptor conversion, repeat biomarker studies are warranted in making appropriate treatment plans in the era of precision medicine. Furthermore, large prospective studies are needed on survival outcomes in patients with a change in systemic therapeutic decision based on receptor conversion.
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70:7–30.
Pan H, Gray R, Braybrooke J, Davies C, Taylor C, McGale P, et al. 20-year risks of breast-cancer recurrence after stopping endocrine therapy at 5 years. N Engl J Med. 2017;377:1836–46.
Radosa JC, Eaton A, Stempel M, Khander A, Liedtke C, Solomayer EF, et al. Evaluation of local and distant recurrence patterns in patients with triple-negative breast cancer according to age. Ann Surg Oncol. 2017;24:698–704.
Howlader NNA, Krapcho M, Miller D, Bishop K, Kosary CL, Yu M, et al. SEER Cancer Statistics Review, 1975–2014. https://seer.cancer.gov/csr/1975_2014. 2017.
Schrijver W, Suijkerbuijk KPM, van Gils CH, van der Wall E, Moelans CB, van Diest PJ. Receptor conversion in distant breast cancer metastases: a systematic review and meta-analysis. J Natl Cancer Inst. 2018;110:568–80.
Turner NH, Di Leo A. HER2 discordance between primary and metastatic breast cancer: assessing the clinical impact. Cancer Treat Rev. 2013;39:947–57.
Kroigard AB, Larsen MJ, Thomassen M, Kruse TA. Molecular concordance between primary breast cancer and matched metastases. Breast J. 2016;22:420–30.
Cui X, Schiff R, Arpino G, Osborne CK, Lee AV. Biology of progesterone receptor loss in breast cancer and its implications for endocrine therapy. J Clin Oncol. 2005;23:7721–35.
Aurilio G, Disalvatore D, Pruneri G, Bagnardi V, Viale G, Curigliano G, et al. A meta-analysis of oestrogen receptor, progesterone receptor and human epidermal growth factor receptor 2 discordance between primary breast cancer and metastases. Eur J Cancer. 2014;50:277–89.
NCCN (1AD). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) Breast Cancer. Version 1 2015. 1–187. https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf.
Van Poznak C, Somerfield MR, Bast RC, Cristofanilli M, Goetz MP, Gonzalez-Angulo AM, et al. Use of biomarkers to guide decisions on systemic therapy for women with metastatic breast cancer: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2015;33:2695–704.
Cardoso F, Costa A, Norton L, Senkus E, Aapro M, Andre F, et al. ESO-ESMO 2nd international consensus guidelines for advanced breast cancer (ABC2)dagger. Ann Oncol. 2014;25:1871–88.
Hoefnagel LD, van de Vijver MJ, van Slooten HJ, Wesseling P, Wesseling J, Westenend PJ, et al. Receptor conversion in distant breast cancer metastases. Breast Cancer Res. 2010;12:R75.
Hoefnagel LD, van der Groep P, van de Vijver MJ, Boers JE, Wesseling P, Wesseling J, et al. Discordance in ERalpha, PR and HER2 receptor status across different distant breast cancer metastases within the same patient. Ann Oncol. 2013;24:3017–23.
Hammond ME, Hayes DF, Dowsett M, Allred DC, Hagerty KL, Badve S, et al. American Society of Clinical Oncology/College Of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Clin Oncol. 2010;28:2784–95.
Wolff AC, Hammond ME, Schwartz JN, Hagerty KL, Allred DC, Cote RJ, et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. J Clin Oncol. 2007;25:118–45.
Wolff AC, Hammond ME, Hicks DG, Dowsett M, McShane LM, Allison KH, et al. Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. J Clin Oncol. 2013;31:3997–4013.
Wolff AC, Hammond MEH, Allison KH, Harvey BE, Mangu PB, Bartlett JMS, et al. Human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Focused Update. Arch Pathol Lab Med. 2018;142:1364–82.
Soni A, Ren Z, Hameed O, Chanda D, Morgan CJ, Siegal GP, et al. Breast cancer subtypes predispose the site of distant metastases. Am J Clin Pathol. 2015;143:471–8.
Wei S, Siegal GP. Surviving at a distant site: the organotropism of metastatic breast cancer. Semin Diagn Pathol. 2018;35:108–11.
Rosen PP, Menendez-Botet CJ, Urban JA, Fracchia A, Schwartz MK. Estrogen receptor protein (ERP) in multiple tumor specimens from individual patients with breast cancer. Cancer. 1977;39:2194–200.
Lindstrom LS, Karlsson E, Wilking UM, Johansson U, Hartman J, Lidbrink EK, et al. Clinically used breast cancer markers such as estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2 are unstable throughout tumor progression. J Clin Oncol. 2012;30:2601–8.
Houssami N, Macaskill P, Balleine RL, Bilous M, Pegram MD. HER2 discordance between primary breast cancer and its paired metastasis: tumor biology or test artefact? Insights through meta-analysis. Breast Cancer Res Treat. 2011;129:659–74.
Fitzgibbons PL, Page DL, Weaver D, Thor AD, Allred DC, Clark GM, et al. Prognostic factors in breast cancer. College of American Pathologists Consensus Statement 1999. Arch Pathol Lab Med. 2000;124:966–78.
Lim TH, Lim AS, Thike AA, Tien SL, Tan PH. Implications of the updated 2013 American Society of Clinical Oncology/College of American Pathologists Guideline recommendations on human epidermal growth factor receptor 2 gene testing using immunohistochemistry and fluorescence in situ hybridization for breast cancer. Arch Pathol Lab Med. 2016;140:140–7.
Pusztai L, Viale G, Kelly CM, Hudis CA. Estrogen and HER-2 receptor discordance between primary breast cancer and metastasis. Oncologist. 2010;15:1164–8.
Talmadge JE. Clonal selection of metastasis within the life history of a tumor. Cancer Res. 2007;67:11471–5.
Suzuki M, Tarin D. Gene expression profiling of human lymph node metastases and matched primary breast carcinomas: clinical implications. Mol Oncol. 2007;1:172–80.
Urquidi V, Sloan D, Kawai K, Agarwal D, Woodman AC, Tarin D, et al. Contrasting expression of thrombospondin-1 and osteopontin correlates with absence or presence of metastatic phenotype in an isogenic model of spontaneous human breast cancer metastasis. Clin Cancer Res. 2002;8:61–74.
Bardou VJ, Arpino G, Elledge RM, Osborne CK, Clark GM. Progesterone receptor status significantly improves outcome prediction over estrogen receptor status alone for adjuvant endocrine therapy in two large breast cancer databases. J Clin Oncol. 2003;21:1973–9.
Shen T, Brandwein-Gensler M, Hameed O, Siegal GP, Wei S. Characterization of estrogen receptor-negative/progesterone receptor-positive breast cancer. Hum Pathol. 2015;46:1776–84.
Acknowledgements
This study was presented in part at the 109th Annual Meeting of United States and Canadian Academy of Pathology on March 3, 2020, Los Angeles, CA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, R., Qarmali, M., Siegal, G.P. et al. Receptor conversion in metastatic breast cancer: analysis of 390 cases from a single institution. Mod Pathol 33, 2499–2506 (2020). https://doi.org/10.1038/s41379-020-0615-z
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41379-020-0615-z
This article is cited by
-
Breast cancer liver metastases and the impact of receptor expression on survival
Clinical & Experimental Metastasis (2026)
-
The association between young age at metastatic breast cancer diagnosis and overall survival in the EMBRACE study
npj Breast Cancer (2025)
-
AI-driven virtual cell models in preclinical research: technical pathways, validation mechanisms, and clinical translation potential
npj Digital Medicine (2025)
-
HER2 expression in cervical squamous cell carcinoma: high prevalence of HER2-low/ultralow and spatiotemporal heterogeneity across tumor evolution
npj Precision Oncology (2025)
-
Multidisciplinary discussion on phenotypical characterization of metastatic breast cancer and patient pathway optimization from an expert panel across South Italian regions
Scientific Reports (2025)