Abstract
The present study examines the safety profile of intravenous onasemnogene abeparvovec gene therapy in a real-world setting, both alone or in combination with intrathecal antisense oligonucleotide nusinersen therapy in two cohorts of patients with spinal muscular atrophy (SMA). The first cohort included eight presymptomatic infants treated solely with onasemnogene abeparvovec, while the second cohort comprised six symptomatic infants receiving onasemnogene abeparvovec and nusinersen co-therapy. All patients received the corticosteroid prednisolone coincident with gene therapy. Circulating alanine aminotransferase (ALT) and aspartate transaminase (AST) levels were measured to determine potential hepatoxicity, the primary focus of this study. Elevated ALT and AST levels were observed in one pre-symptomatic and three symptomatic patients post-treatment. However, all values returned to normal levels within 3 months of onasemnogene abeparvovec injection. Nusinersen treatment received previously or coincident with gene therapy did not impact the transient elevation of liver transaminases. This study highlights the importance of early intervention with molecular treatments for SMA and indicates that prior or coincident treatment with nusinersen is unlikely to impact safety of onasemnogene apoparvovec and could theoretically improve clinical outcomes in symptomatic infants or in those with gene therapy delayed beyond the immediate neonatal period.
Similar content being viewed by others
Introduction
Molecular therapies have demonstrated remarkable clinical benefits when administered during the pre-symptomatic or early symptomatic stages of spinal muscular atrophy (SMA), significantly improving survival rates even among the most severely affected infants [1]. In 2016, nusinersen (Spinraza, Biogen) became the first molecular therapy approved for SMA, and numerous studies have since showcased its efficacy and safety across various SMA populations [2, 3]. In 2019, the U.S. Food and Drug Administration (FDA) approved onasemnogene abeparvovec (Zolgensma, Novartis/AVXS-101, AveXis), a gene therapy using an adeno-associated virus vector to deliver the SMN1 gene through a single intravenous injection in newborns and infants with SMA. The current treatment approach for SMA in the U.S. is to choose one therapy and then only consider adding another therapy if the first therapy proves to have insufficient benefit. However, despite limited safety data on combination approaches, there is an increasing number of infants receiving dual or sequential therapy with gene therapy and nusinersen: the lack of safety studies has yet to be adequately addressed in published clinical trials or studies.
The most common adverse effect reported with onasemnogene abeparvovec gene therapy is elevated liver enzyme levels, which can indicate acute liver injury [4, 5]. In clinical practice, these elevated levels are usually managed effectively with co-administration of prednisolone, a corticosteroid hormone [5, 6]. Clinical trials have demonstrated that initiating prednisolone 48 h prior to gene therapy dosing and adjusting as needed for 2–3 months while closely monitoring liver function can successfully avoid serious medical consequences [7]. However, further longitudinal research is needed to fully understand the implications and risk of serious consequences at the extremes of age and weight not included in clinical trials.
Combination therapies for SMA patients could be used more strategically to facilitate maximum rescue of at-risk motor neurons and other tissues [8]. In newborns identified by newborn screening, timing of gene therapy initiation is variable, but not infrequently delayed several weeks. In an infant with two SMN2 copies, this could lead to substantial progression of motor neuron denervation. Nusinersen loading has been associated with a dramatic and persistent drop in neurofilament levels in two copy infants, and has a long half-life that could persist through the gene therapy dosing and transduction process. Thus, in a presymptomatic clinical cohort of infants with two SMN2 copies, we hypothesize that sequential administration of nusinersen and onasemnogene abeparvovec could present a synergistic effect in these vulnerable SMA infants, without the need to continue long-term combination therapy. In already symptomatic two copy infants, near simultaneous dosing at the earliest opportunity, as demonstrated in the cohort included in this study, could maximize survival of the greatest number of at-risk motor neurons and help ensure maximal transduction efficacy. Here, we report a comprehensive safety dataset including the levels of circulating liver enzymes from SMA cases receiving treatment with only onasemnogene abeparvovec, and SMA cases receiving combination therapy with nusinersen and onasemnogene abeparvovec early in life.
Methods
This is a longitudinal cohort study including a total of 14 SMA newborns or infants treated in the first months of life at the Massachusetts General Hospital (MGH), including eight infants who received treatment with onasemnogene abeparvovec and six infants who received combination treatment with nusinersen and then onasemnogene abeparvovec. Intravenous dosage for onasemnogene abeparvovec was 1.1 x 10e14 vg/kg of body weight. Nusinersen followed the normal dosing schedule with intrathecal injection at 12 mg each time. Patients 571, 832, 846, and 809 received 8, 2, 4, and 3 maintenance doses respectively, during the course of this study. All patients received prednisolone 48 h prior to onasemnogene abeparvovec dosing and continued for 2–3 months post dosing as per standard of care. Dosage was 1 mg/kg body weight administered daily as a 3 mg/mL oral solution. The dose was increased to 1.2–1.5 mg/kg body weight per day in cases with transaminase elevation. No patients received risdiplam (Evrysdi, Roche). Of note, all patients receiving only onasemnogene abeparvovec were treated early in life at the pre-symptomatic stage, while all patients receiving co-therapy with nusinersen and onasemnogene abeparvovec in this study demonstrated clinical symptoms with generalized weakness of skeletal, bulbar, and respiratory muscles at dosing. Of note, subject ID 571 was treated under an individual IND as part of an access program before FDA approval. The primary outcomes of this study are circulating alanine aminotransferase (ALT) and aspartate transaminase (AST) levels measured as biomarkers of potential hepatotoxicity. In addition to the absolute values, the fold change from the baseline (i.e., measurements taken before injection) ALT and AST levels were also calculated for each individual patient and presented. Circulating gamma-glutamyl transferase, troponin T-hs Gen 5 (Troponin T), white blood counts, platelets, and albumin levels were also determined as secondary outcomes along with any clinical adverse effect manifestation. Reference normal ranges were determined based on MGH laboratory references. Reference values from the Mayo Clinic and the University of California San Francisco (UCSF) Children’s are also provided. SMN1 and SMN2 copy numbers were determined using quantitative polymerase chain reaction as previously described [9]. All other outcomes were obtained from the clinical laboratory in association with research visits in the SPOT SMA Longitudinal Pediatric Data Repository (LPDR) housed within the Research Electronic Data Capture Web Application at the Newborn Screening Translational Research Network [10]. Written informed parental consent was obtained from all participants under the Institutional Ethics Review Board at the MGH (protocol #2016-P000469).
Results
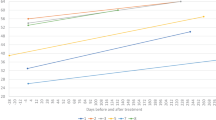
We determined circulating ALT and AST levels in two cohorts of SMA patients receiving gene therapy early in life (Tables 1 and 2). Distribution of age, sex, race, and ethnicity is presented in Table 1. The first cohort included 8 SMA infants ages 20 –190 days old treated at the pre-symptomatic stage. They received only onasemnogene abeparvovec treatment (Fig. 1A–H). The second cohort included 6 SMA infants and 1 child who received both nusinersen (dosed at 10–91 days old) and onasemnogene abeparvovec (dosed at 29–940 days old) (Fig. 2A–F). Only 1 of 8 presymptomatic infants dosed with onasemnogene abeparvovec had elevated circulating ALT and AST levels, occurring within a few days post injection (Fig. 1B), showing a 20-fold change in ALT and a 9-fold change in AST (Fig. 3A, B, E, F). This specific case was a subject with 3 SMN2 copies dosed at 20 days old. Importantly, increased ALT and AST levels returned to normal within 3 months after the injection (Figs. 1B and 3A, B, E, F). For the second cohort, including all symptomatic patients with 2 SMN2 copies, we observed three out of six cases with elevated circulating ALT and AST levels post onasemnogene abeparvovec injection (Fig. 2A-F). Two patients showed similar changes, both displaying about a 4.5-fold increase in ALT and a 4-to-7-fold increase in AST. Another patient showed a 28-fold change in ALT and a 6-fold change in AST from baseline (Fig. 3C, D, E, F). Of note, this patient was also the oldest dosed patient at 940 days old with onasemnogene abeparvovec. Increased ALT and AST levels also returned to normal levels around 3 months after the injection and were maintained at normal ranges during follow-up (Figs. 2A, C, E and 3C, D, E, F). Taken together, these data closely parallel previously published clinical trial data demonstrating that some infants with SMA experience transient hepatotoxicity after gene therapy even when prednisolone is co-administered, but these effects are transient and typically return to normal by three months. Importantly, all enrolled patients had normal albumin levels and no indications of malnutrition or chronic liver disease (Table 2 and Supplementary Table 1).
A–F Cohort receiving co-therapy. Blue line indicates the day of onasemnogene abeparvovec injection. Red line indicates the initial nusinersen dose (initial of loading phase) for each case, while red arrows indicate nusinersen maintenance doses with dosage number in text above. Colored background area indicates normal ALT and AST concentrations expected in sex-matched controls. Note: Case #571 has initiated nusinersen treatment more than 125 days before the onasemnogene abeparvovec dose.
As complementary analysis, we observed increased levels of troponin T and platelets in most of the SMA infants and children (Table 2 and Supplementary File 1), indicating potential myocardial injury and thrombocytosis. These increased levels are most likely part of the natural course of SMA pathophysiology since they were not observed as a direct consequence of any therapy. Other measurements such as circulating white blood counts and platelets also demonstrated some specific values out of normal range during this longitudinal follow-up study but did not clearly correlate with treatment (Table 2 and Supplementary File 1). Moreover, we observed multiple mild or moderate clinical events during the follow-up of this study, but only four patients treated with either gene therapy or co-therapy showed at least one severe adverse event related to a cardiac, respiratory, or nutritional issue (Table 3). These adverse events were managed in hospital visits. They were not clearly associated with treatment response and were considered most likely related to the primary underlying diagnosis. Altogether, these findings validate previous evidence showing that patients with SMA need to be closely monitored after transformative gene therapy is administered in the real-world clinical setting.
Discussion
Recent clinical trials have validated the effectiveness of onasemnogene abeparvovec in both pre-symptomatic and symptomatic infants, showing significant motor function improvements [11,12,13]. Over the past three years, onasemnogene abeparvove has largely become the treatment of choice for monotherapy in newborns with homozygous SMN1 deletion, regardless of SMN2 copy number. While gene therapy is a transformative treatment for SMA, varying adverse events have been observed: most commonly categorized as mild or moderate in severity, these adverse events need to be closely monitored [11,12,13]. There are infants receiving dual or sequential therapy with gene therapy and nusinersen or risdiplam. One of the potentially largest adverse impacts on the long-term efficacy of gene therapy treatment in newborns with 2 SMN2 copies is the delay inherent in obtaining preauthorization, approval, and required preliminary safety testing prior to the administration of AAV9-mediated gene therapy. For a proportion of newborns, because of their catabolic state, variable transition to appropriate weight gain, and social factors in the critical postpartum period, immediate dosing post-birth may be neither ideal nor feasible. Thus, adequate processes for rapid initiation of an alternative molecular therapy need to be standardized and would ideally include overlapping coverage through the duration of the viral transduction process post-dosing with AAV9-mediated intravenously delivered gene therapy.
Taking advantage of our longitudinal database (MGH SPOT SMA LPDR) that includes SMA cases treated early in life with onasemnogene abeparvovec, we were able to evaluate several markers related to the safety of gene therapy. Our findings indicate that some SMA patients can present hepatotoxicity within a few days after receiving the gene therapy. More drastic changes in ALT and AST values were notably observed in patients dosed with gene therapy at an older age in the second cohort in contrast to the first cohort, whose age at dosing was younger on average, reemphasizing the importance of initiating therapy in two SMN2 copy infants at the earliest opportunity. These effects are transient and return to normal levels within three months post-injection. Corroborating these single-center observations, a study combining data from 5 previous clinical trials has observed liver-related adverse events in 34% of SMA patients treated with gene therapy [4]. Variable liver response to the gene therapy was also clearly observed in the original trial establishing the single-dose gene therapy for SMA and emphasized the importance of co-treatment with oral prednisolone to mitigate immune-mediated hepatotoxicity [5]. Prednisolone doses can be adjusted when necessary in each specific SMA case [5]. Here, we observed a total of four SMA cases with transient liver injury in spite of prednisolone treatment, but without any evident long-lasting consequences. We speculate that the older age and symptomatic disease status in three of six patients receiving co-therapy made it more likely for them to manifest transient liver injury post onasemnogene dosing due to (1) a higher dose of vector based on increased size and weight at the time of dosing, (2) a more mature immune system, and (3) the presence of systemic metabolic dysfunction related to generalized muscular atrophy and severely diminished lean body mass. However, we cannot entirely exclude a potential synergistic toxic effect of co-therapy.
We report longitudinal safety data from six symptomatic SMA cases with only two SMN2 copies that have been treated with co-therapy with onasemnogene abeparvovec and nusinersen. Each therapy has a different mechanism of action and their specific limitations. Nusinersen is administered through intrathecal injections and works by modifying the splicing of SMN2 to increase SMN protein production. As a monotherapy in its current formulation, nusinersen requires lifelong-intermittent intrathecal injections at 4 month intervals post-loading phase and does not address the underlying genetic defect in the SMN1 gene. Gene therapy (onasemnogene abeparvovec) is a one-time intravenous gene therapy delivering a functional copy of SMN1 to the episome to increase SMN protein production. Despite the robustness of this therapy overall, particularly in the setting of newborn screening for SMA, the potential variable percentage of motor neurons transduced, the inherent delays in dosing associated with the process, and the variable outcomes in a significant subset of newborns with two SMN2 copies are at least as important as the safety concerns. Thus, alternative combination approaches that leverage the strengths of the existing therapies and maximize their benefit in the most vulnerable cohorts of SMA patients, including newborns with two SMN2 copies as well as already symptomatic infants, are warranted.
Symptomatic SMA cases who received dual therapy in this study are achieving motor milestones that were not expected in the natural history of the disease [10] and the present data also suggest that side effects of these therapies are transient and can be controlled by closely monitoring each patient. These results demonstrate the safety for sequential and coincident nusinersen therapy during the induction process for gene therapy. Nusinersen treatment administered pre-gene therapy treatment theoretically provides an advantage over risdiplam, given its prolonged treatment effect following an initial loading period. However, we did not examine the impact or safety of sequential or coincident treatment with risdiplam in this study, and thus our conclusions are limited. In line with our findings, another recent study including two SMA cases that received co-therapy with nusinersen and onasemnogene abeparvovec also reported no apparent increased burden of adverse effects [8]. This possibility of combining therapies for SMA patients has presented itself as particularly interesting for the SMA community. Based on our current data, we advocate for additional studies analyzing different outcomes in SMA patients receiving co-therapies and an open discussion about the advantages and disadvantages of combining these treatments. We hypothesize that there is limited to no benefit in continuing a second therapy once gene therapy has been delivered in a truly presymptomatic infant with two copies with regard to motor neuron rescue. However, there may be in an infant with some evident denervation and an emerging clinical phenotype. In our experience, progressive denervation can sometimes occur quite precipitously in two copy infants between ordering and dosing gene therapy. Thus, biomarkers such as CMAP, MUNE, or robust serum biomarkers must be integrated into the current clinical paradigm since clinical exams and even functional motor tests remain insufficient for this purpose. In addition, this study does not address the potential value of long term dual therapy on emergence of other systemic manifestations in infants with 2 SMN2 copies.
We observed increased troponin T and variable platelet levels in SMA patients, which are markers of increased risk for myocardial injury and thrombocytosis. While damage in motor neurons accounts for the earliest manifestations in SMA infants, evidence indicates that other tissues contribute to the SMA pathophysiology due to the direct effects of SMN deficiency in tissues other than motoneurons or indirect effects due to the severe denervation status [14, 15]. Thus, elevated troponin T levels could be a marker of defects in heart tissues playing a role in the SMA pathophysiology and all elevated troponin T levels were not related to the treatment status [14, 16]. However, additional studies have revealed that troponin T could also be expressed in the skeletal muscle [17,18,19]. Therefore, we cannot exclude the possibility of elevated troponin T circulating levels as an additional marker of muscle denervation, which is expected during SMA progression. One alternative would be to measure the levels of troponin I, which could be more reliable as a marker of myocardial injury [20], but unfortunately we did not collect these data in this cohort study or perform electrocardiogram, magnetic resonance imaging, or echocardiogram to test potential cardiac damage. A multidisciplinary approach is critical to monitor and provide the best care for SMA patients. Markers assessed in the current study corroborate the premise of systemic disease manifestation and highlight the importance of close monitoring for the emergence of disease phenotypes beyond muscle denervation.
In summary, this case series analysis study highlights the importance of early intervention with gene therapy and other molecular treatments for SMA and the need for close monitoring of treatment responses beyond muscle denervation in the real-world clinical setting.
Data availability
All data relevant to this study are contained within the manuscript.
References
Groen EJN, Talbot K, Gillingwater TH. Advances in therapy for spinal muscular atrophy: promises and challenges. Nat Rev Neurol. 2018;14:214–24.
Finkel RS, Mercuri E, Darras BT, Connolly AM, Kuntz NL, Kirschner J, et al. Nusinersen versus sham control in infantile-onset spinal muscular atrophy. N Engl J Med. 2017;377:1723–32.
Hagenacker T, Wurster CD, Günther R, Schreiber-Katz O, Osmanovic A, Petri S, et al. Nusinersen in adults with 5q spinal muscular atrophy: a non-interventional, multicentre, observational cohort study. Lancet Neurol. 2020;19:317–25.
Chand D, Mohr F, McMillan H, Tukov FF, Montgomery K, Kleyn A, et al. Hepatotoxicity following administration of onasemnogene abeparvovec (AVXS-101) for the treatment of spinal muscular atrophy. J Hepatol. 2021;74:560–6.
Mendell JR, Al-Zaidy S, Shell R, Arnold WD, Rodino-Klapac LR, Prior TW, et al. Single-dose gene-replacement therapy for spinal muscular atrophy. N Engl J Med. 2017;377:1713–22.
Al-Zaidy SA, Mendell JR. From clinical trials to clinical practice: practical considerations for gene replacement therapy in SMA type 1. Pediatr Neurol. 2019;100:3–11.
Mendell JR, Al-Zaidy SA, Lehman KJ, McColly M, Lowes LP, Alfano LN, et al. Five-year extension results of the phase 1 START trial of onasemnogene abeparvovec in spinal muscular atrophy. JAMA Neurol. 2021;78:834.
Lee BH, Collins E, Lewis L, Guntrum D, Eichinger K, Voter K, et al. Combination therapy with nusinersen and AVXS-101 in SMA type 1. Neurology. 2019;93:640–1.
Prior TW, Krainer AR, Hua Y, Swoboda KJ, Snyder PC, Bridgeman SJ, et al. A positive modifier of spinal muscular atrophy in the SMN2 gene. Am J Hum Genet. 2009;85:408–13.
Alves CRR, Petrillo M, Spellman R, Garner R, Zhang R, Kiefer M, et al. Implications of circulating neurofilaments for spinal muscular atrophy treatment early in life: a case series. Mol Ther Methods Clin Dev. 2021;23:524–38.
Strauss KA, Farrar MA, Muntoni F, Saito K, Mendell JR, Servais L, et al. Onasemnogene abeparvovec for presymptomatic infants with two copies of SMN2 at risk for spinal muscular atrophy type 1: the phase III SPR1NT trial. Nat Med. 2022;28:1381–9.
Strauss KA, Farrar MA, Muntoni F, Saito K, Mendell JR, Servais L, et al. Onasemnogene abeparvovec for presymptomatic infants with three copies of SMN2 at risk for spinal muscular atrophy: the Phase III SPR1NT trial. Nat Med. 2022;28:1390–7.
Day JW, Finkel RS, Chiriboga CA, Connolly AM, Crawford TO, Darras BT, et al. Onasemnogene abeparvovec gene therapy for symptomatic infantile-onset spinal muscular atrophy in patients with two copies of SMN2 (STR1VE): an open-label, single-arm, multicentre, phase 3 trial. Lancet Neurol. 2021;20:284–93.
Lipnick SL, Agniel DM, Aggarwal R, Makhortova NR, Finlayson SG, Brocato A, et al. Systemic nature of spinal muscular atrophy revealed by studying insurance claims. PLOS ONE. 2019;14:e0213680.
Nery FC, Siranosian JJ, Rosales I, Deguise M, Sharma A, Muhtaseb AW, et al. Impaired kidney structure and function in spinal muscular atrophy. Neurol Genet. 2019;5.e353.
Wijngaarde CA, Blank AC, Stam M, Wadman RI, van den Berg LH, van der Pol WL. Cardiac pathology in spinal muscular atrophy: a systematic review. Orphanet J Rare Dis. 2017;12. 67.
Saggin L, Gorza L, Ausoni S, Schiaffino S. Cardiac troponin T in developing, regenerating and denervated rat skeletal muscle. Development. 1990;110:547–54.
Bodor GS, Survant L, Voss EM, Smith S, Porterfield D, Apple FS. Cardiac troponin T composition in normal and regenerating human skeletal muscle. Clin Chem. 1997;43:476–84.
Jende JME, Groener JB, Kender Z, Hahn A, Morgenstern J, Heiland S, et al. Troponin T parallels structural nerve damage in type 2 diabetes: a cross-sectional study using magnetic resonance neurography. Diabetes. 2020;69:713–23.
McLaurin MD, Apple FS, Voss EM, Herzog CA, Sharkey SW. Cardiac troponin I, cardiac troponin T, and creatine kinase MB in dialysis patients without ischemic heart disease: evidence of cardiac troponin T expression in skeletal muscle. Clin Chem. 1997;43:976–82.
Acknowledgements
We are grateful to all the patients and families who participated in this study.
Funding
CRRA received a fellowship from the MGH ECOR. KJS was funded by NIH NICHD R01HD054599, NIH NINDS R21NS108015, Biogen, and Cure SMA.
Author information
Authors and Affiliations
Contributions
KJS and CRRA directed the research project. RGS, SDL, EAA, ER, and KJS collected clinical data. RGS, LLH, SDL, EAA, ER, KJS, and CRRA analyzed the data and participated in the data interpretation. LLH, KJS, and CRRA drafted the manuscript. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
CRRA and KJS are inventors on a patent filed by Mass General Brigham that describes genome engineering technologies to treat SMA. KJS was a recipient of a grant from Biogen and received clinical trial funding from AveXis and Biogen. CRRA is a consultant for Biogen and holds stocks in publicly traded companies developing gene therapies. RGS is a current employee and holds stock/stock options in Voyager therapeutics. The other authors declare no competing interests.
Ethical approval
Ethical approval and written informed parental consent were obtained from all participants under Institutional Ethics Review Boards at the Massachusetts General Hospital (protocol 2016P000469).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Spellman, R.G., Ha, L.L., Da Silva Duarte Lepez, S. et al. Early life safety profiling of gene therapy for spinal muscular atrophy. Gene Ther (2025). https://doi.org/10.1038/s41434-025-00529-6
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41434-025-00529-6