Abstract
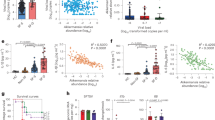
Bile acids are microbial metabolites that can impact infection of enteric and hepatitis viruses, but their functions during systemic viral infection remain unclear. Here we show that elevated levels of the secondary bile acid taurolithocholic acid (TLCA) are associated with reduced fatality rates and suppressed viraemia in patients infected with severe fever with thrombocytopenia syndrome virus (SFTSV), an emerging tick-borne haemorrhagic fever virus. TLCA inhibits viral replication and mitigates host inflammation during SFTSV infection in vitro, and indirectly suppresses SFTSV-mediated induction of ferroptosis by upregulating fatty acid desaturase 2 via the TGR5–PI3K/AKT–SREBP2 axis. High iron and ferritin serum levels during early infection were correlated with decreased TLCA levels and fatal outcomes in SFTSV-infected patients, indicating potential biomarkers. Furthermore, treatment with either ferroptosis inhibitors or TLCA protected mice from lethal SFTSV infection. Our findings highlight the therapeutic potential of bile acids to treat haemorrhagic fever viral infection.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in the Article. RNA-seq data are available in the SRA under BioProject accession numbers PRJNA1086175 and PRJNA1086126. Source data are provided with this paper.
References
de Aguiar Vallim, T. Q., Tarling, E. J. & Edwards, P. A. Pleiotropic roles of bile acids in metabolism. Cell Metab. 17, 657–669 (2013).
Fiorucci, S., Distrutti, E., Carino, A., Zampella, A. & Biagioli, M. Bile acids and their receptors in metabolic disorders. Prog. Lipid Res. 82, 101094 (2021).
Cai, J., Sun, L. & Gonzalez, F. J. Gut microbiota-derived bile acids in intestinal immunity, inflammation, and tumorigenesis. Cell Host Microbe 30, 289–300 (2022).
Kuipers, F., Bloks, V. W. & Groen, A. K. Beyond intestinal soap—bile acids in metabolic control. Nat. Rev. Endocrinol. 10, 488–498 (2014).
Chang, K.-O. & George, D. W. Bile acids promote the expression of hepatitis C virus in replicon-harboring cells. J. Virol. 81, 9633–9640 (2007).
Murakami, K. et al. Bile acids and ceramide overcome the entry restriction for GII.3 human norovirus replication in human intestinal enteroids. Proc. Natl Acad. Sci. USA 117, 1700–1710 (2020).
Honke, N. et al. Farnesoid X receptor in mice prevents severe liver immunopathology during lymphocytic choriomeningitis virus infection. Cell. Physiol. Biochem. 41, 323–338 (2017).
Lowjaga, K. A. A. T. et al. Long-term trans-inhibition of the hepatitis B and D virus receptor NTCP by taurolithocholic acid. Am. J. Physiol. Gastrointest. Liver Physiol. 320, G66–G80 (2021).
Yan, H. et al. Viral entry of hepatitis B and D viruses and bile salts transportation share common molecular determinants on sodium taurocholate cotransporting polypeptide. J. Virol. 88, 3273–3284 (2014).
Brevini, T. et al. FXR inhibition may protect from SARS-CoV-2 infection by reducing ACE2. Nature 615, 134–142 (2023).
Hu, M.-M. et al. Virus-induced accumulation of intracellular bile acids activates the TGR5-β–arrestin–SRC axis to enable innate antiviral immunity. Cell Res. 29, 193–205 (2019).
Winkler, E. S. et al. The intestinal microbiome restricts alphavirus infection and dissemination through a bile acid–type I IFN signaling axis. Cell 182, 901–918.e18 (2020).
Jang, J. Y., Im, E., Choi, Y. H. & Kim, N. D. Mechanism of bile acid-induced programmed cell death and drug discovery against cancer: a review. Int. J. Mol. Sci. 23, 7184 (2022).
Tschuck, J. et al. Farnesoid X receptor activation by bile acids suppresses lipid peroxidation and ferroptosis. Nat. Commun. 14, 6908 (2023).
Rex, D. A. B., Keshava Prasad, T. S. & Kandasamy, R. K. Revisiting regulated cell death responses in viral infections. Int. J. Mol. Sci. 23, 7023 (2022).
Dixon, S. J. et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149, 1060–1072 (2012).
Han, Y. et al. SARS-CoV-2 infection induces ferroptosis of sinoatrial node pacemaker cells. Circ. Res. 130, 963–977 (2022).
Xiao, Q. et al. Metabolism-dependent ferroptosis promotes mitochondrial dysfunction and inflammation in CD4+ T lymphocytes in HIV-infected immune non-responders. EBioMedicine 86, 104382 (2022).
Kim, Y. R. et al. Severe fever with thrombocytopenia syndrome virus infection, South Korea, 2010. Emerg. Infect. Dis. 24, 2103–2105 (2018).
Li, H. et al. Epidemiological and clinical features of laboratory-diagnosed severe fever with thrombocytopenia syndrome in China, 2011–17: a prospective observational study. Lancet Infect. Dis. 18, 1127–1137 (2018).
Takahashi, T. et al. The first identification and retrospective study of severe fever with thrombocytopenia syndrome in Japan. J. Infect. Dis. 209, 816–827 (2014).
Tran, X. C. et al. Endemic severe fever with thrombocytopenia syndrome, Vietnam. Emerg. Infect. Dis. 25, 1029–1031 (2019).
Win, A. M. et al. Genotypic heterogeneity of Orientia tsutsugamushi in scrub typhus patients and thrombocytopenia syndrome co-infection, Myanmar. Emerg. Infect. Dis. 26, 1878–1881 (2020).
Yu, X.-J. et al. Fever with thrombocytopenia associated with a novel bunyavirus in China. N. Engl. J. Med. 364, 1523–1532 (2011).
Miao, D. et al. Mapping the global potential transmission hotspots for severe fever with thrombocytopenia syndrome by machine learning methods. Emerg. Microbes Infect. 9, 817–826 (2020).
Wu, Y.-X. et al. Human-to-human transmission of severe fever with thrombocytopenia syndrome virus through potential ocular exposure to infectious blood. Int. J. Infect. Dis. 123, 80–83 (2022).
Xie, J. et al. Akkermansia muciniphila protects mice against an emerging tick-borne viral pathogen. Nat. Microbiol. 8, 91–106 (2023).
Li, H. et al. Calcium channel blockers reduce severe fever with thrombocytopenia syndrome virus (SFTSV) related fatality. Cell Res. 29, 739–753 (2019).
Perino, A. & Schoonjans, K. TGR5 and immunometabolism: insights from physiology and pharmacology. Trends Pharmacol. Sci. 36, 847–857 (2015).
Triki, M. et al. mTOR signaling and SREBP activity increase FADS2 expression and can activate sapienate biosynthesis. Cell Rep. 31, 107806 (2020).
Yi, J., Zhu, J., Wu, J., Thompson, C. B. & Jiang, X. Oncogenic activation of PI3K–AKT–mTOR signaling suppresses ferroptosis via SREBP-mediated lipogenesis. Proc. Natl Acad. Sci. USA 117, 31189–31197 (2020).
Jiang, Y. et al. EGLN1/c-Myc induced lymphoid-specific helicase inhibits ferroptosis through lipid metabolic gene expression changes. Theranostics 7, 3293–3305 (2017).
Xuan, Y. et al. SCD1/FADS2 fatty acid desaturases equipoise lipid metabolic activity and redox-driven ferroptosis in ascites-derived ovarian cancer cells. Theranostics 12, 3534–3552 (2022).
Jiang, X., Stockwell, B. R. & Conrad, M. Ferroptosis: mechanisms, biology and role in disease. Nat. Rev. Mol. Cell Biol. 22, 266–282 (2021).
Kapralov, A. A. et al. Redox lipid reprogramming commands susceptibility of macrophages and microglia to ferroptotic death. Nat. Chem. Biol. 16, 278–290 (2020).
Wang, H. et al. Characterization of ferroptosis in murine models of hemochromatosis. Hepatology 66, 449–465 (2017).
Zhong, H. & Yin, H. Role of lipid peroxidation derived 4-hydroxynonenal (4-HNE) in cancer: focusing on mitochondria. Redox Biol. 4, 193–199 (2015).
Hassannia, B., Vandenabeele, P. & Vanden Berghe, T. Targeting ferroptosis to iron out cancer. Cancer Cell 35, 830–849 (2019).
Oh, S.-J., Ikeda, M., Ide, T., Hur, K. Y. & Lee, M.-S. Mitochondrial event as an ultimate step in ferroptosis. Cell Death Discov. 8, 414 (2022).
McMullan, L. K. et al. A new phlebovirus associated with severe febrile illness in Missouri. N. Engl. J. Med. 367, 834–841 (2012).
Zhang, D.-L. et al. Hepcidin regulates ferroportin expression and intracellular iron homeostasis of erythroblasts. Blood 118, 2868–2877 (2011).
Kim, Y. & Chang, K.-O. Inhibitory effects of bile acids and synthetic farnesoid X receptor agonists on rotavirus replication. J. Virol. 85, 12570–12577 (2011).
Zhao, J. et al. SIK2 enhances synthesis of fatty acid and cholesterol in ovarian cancer cells and tumor growth through PI3K/Akt signaling pathway. Cell Death Dis. 11, 25 (2020).
DeBose-Boyd, R. A. et al. Transport-dependent proteolysis of SREBP: relocation of site-1 protease from Golgi to ER obviates the need for SREBP transport to Golgi. Cell 99, 703–712 (1999).
Namkoong, S. et al. The integral membrane protein ITM2A, a transcriptional target of PKA-CREB, regulates autophagic flux via interaction with the vacuolar ATPase. Autophagy 11, 756–768 (2015).
Yan, J.-M. et al. Bunyavirus SFTSV exploits autophagic flux for viral assembly and egress. Autophagy 18, 1599–1612 (2022).
Vila, I. K. et al. STING orchestrates the crosstalk between polyunsaturated fatty acid metabolism and inflammatory responses. Cell Metab. 34, 125–139.e8 (2022).
Xie, J., Su, M., Dang, Y. & Zhao, L. Prognostic value of serum ferritin for patients with severe fever with thrombocytopenia syndrome: a single-center retrospective cohort study. Infect. Dis. Ther. 12, 979–988 (2023).
Chen, K. et al. Ferritin and procalcitonin serve as discriminative inflammatory biomarkers and can predict the prognosis of severe fever with thrombocytopenia syndrome in its early stages. Front. Microbiol. 14, 1168381 (2023).
Drakesmith, H. & Prentice, A. Viral infection and iron metabolism. Nat. Rev. Microbiol. 6, 541–552 (2008).
Wang, M.-P., Joshua, B., Jin, N.-Y., Du, S.-W. & Li, C. Ferroptosis in viral infection: the unexplored possibility. Acta Pharmacol. Sin. 43, 1905–1915 (2022).
Cheng, J., Tao, J., Li, B., Shi, Y. & Liu, H. Swine influenza virus triggers ferroptosis in A549 cells to enhance virus replication. Virol. J. 19, 104 (2022).
Xia, H., Zhang, Z. & You, F. Inhibiting ACSL1-related ferroptosis restrains murine coronavirus infection. Viruses 13, 2383 (2021).
Hayashi, Y. et al. Ablation of fatty acid desaturase 2 (FADS2) exacerbates hepatic triacylglycerol and cholesterol accumulation in polyunsaturated fatty acid-depleted mice. FEBS Lett. 595, 1920–1932 (2021).
Yamane, D. et al. FADS2-dependent fatty acid desaturation dictates cellular sensitivity to ferroptosis and permissiveness for hepatitis C virus replication. Cell Chem. Biol. 29, 799–810.e4 (2022).
Zhou, C., Chen, Y., Ji, Y., He, X. & Xue, D. Increased serum levels of hepcidin and ferritin are associated with severity of COVID-19. Med. Sci. Monit. 26, e926178 (2020).
Wang, J. et al. Correlation of serum hepcidin levels with disease progression in hepatitis B virus-related disease assessed by nanopore film based assay. Sci. Rep. 6, 34252 (2016).
Li, H. et al. Single-cell landscape of peripheral immune responses to fatal SFTS. Cell Rep. 37, 110039 (2021).
Schmidt, P. J. Regulation of iron metabolism by hepcidin under conditions of inflammation. J. Biol. Chem. 290, 18975–18983 (2015).
Abobaker, A. Can iron chelation as an adjunct treatment of COVID-19 improve the clinical outcome? Eur. J. Clin. Pharmacol. 76, 1619–1620 (2020).
Chang, M.-L. et al. Evolution of ferritin levels in hepatitis C patients treated with antivirals. Sci. Rep. 10, 19744 (2020).
Gómez-Pastora, J. et al. Hyperferritinemia in critically ill COVID-19 patients—is ferritin the product of inflammation or a pathogenic mediator? Clin. Chim. Acta 509, 249–251 (2020).
Kakizaki, S. et al. Iron enhances hepatitis C virus replication in cultured human hepatocytes. Liver 20, 125–128 (2000).
Traoré, H. N. & Meyer, D. The effect of iron overload on in vitro HIV-1 infection. J. Clin. Virol. 31, S92–S98 (2004).
Colafrancesco, S., Alessandri, C., Conti, F. & Priori, R. COVID-19 gone bad: a new character in the spectrum of the hyperferritinemic syndrome? Autoimmun. Rev. 19, 102573 (2020).
Guo, C. et al. Bile acids control inflammation and metabolic disorder through inhibition of NLRP3 inflammasome. Immunity 45, 944 (2016).
Beuers, U. et al. Taurolithocholic acid exerts cholestatic effects via phosphatidylinositol 3-kinase-dependent mechanisms in perfused rat livers and rat hepatocyte couplets. J. Biol. Chem. 278, 17810–17818 (2003).
Woolbright, B. L. et al. Lithocholic acid feeding results in direct hepatotoxicity independent of neutrophil function in mice. Toxicol. Lett. 228, 56–66 (2014).
Zhu, S. J. & Wei, L. Akkermansia muciniphila protects mice against an emerging tick-borne viral pathogen. Figshare https://doi.org/10.6084/m9.figshare.21334422.v1 (2023).
Zhang, L. et al. CCR2 is a host entry receptor for severe fever with thrombocytopenia syndrome virus. Sci. Adv. 9, eadg6856 (2023).
Li, S. et al. SFTSV infection induces BAK/BAX-dependent mitochondrial DNA release to trigger NLRP3 inflammasome activation. Cell Rep. 30, 4370–4385.e7 (2020).
Acknowledgements
The study was supported by the National Key Research and Development Plan of China (2021YFC2300200-02 to W.L. and 2022YFC2303300 to H. Li) and the National Natural Science Foundation of China (82172270 to H.L. and 81825019 to W.L.) through funding provisions.
Author information
Authors and Affiliations
Contributions
W.L. and H. Li developed the research plan and experimental strategy. X. Zheng, Y.Z. and L.Z. performed the in vitro experiments. Y.Z., L.Z. and X.W. performed the animal experiments. T.Y., F.Z., X. Zhang, N.C. and H. Lv collected clinical samples and performed related laboratory tests. X. Zheng, Y.Z. and H. Li performed statistical analysis. S.J.Z. provided expertise in in vitro experiments. H. Li, X. Zheng, Y.Z., L.Z. and W.L. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Microbiology thanks Juan Carlos de la Torre and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 TLCA inhibits SFTSV infection in vitro.
a-c, BMDMs infected with SFTSV (MOI = 1) were treated with TLCA at the indicated concentrations for 24 h. Intracellular SFTSV RNA levels (a), NP levels (b) and supernatant viral titers (c) were measured; n = 4 biologically independent samples. d, Viability of TLCA treated BMDMs was measured at 24 h by using CCK-8; n = 6 biologically independent samples. e, Gating strategy to detect the SFTSV infection rate present on Fig. 2g, h. Debris were excluded based on cell size (FSC-H/SSC-H), double adhesions were excluded through FSC-A and FSC-H, and single cells were sorted to define the population of SFTSV NP + . f, THP-1 cells were infected with SFTSV (MOI = 5) for 2 h, and then treated with TLCA at the indicated concentrations. Intracellular and supernatant SFTSV RNAs were measured with RT-qPCR at 16 h post infection; n = 4 biologically independent samples. Data were presented as mean ± s.d. The two-sided p values were examined using One-way ANOVA followed by Tukey’s multiple comparisons test for comparison of continuous variables among multiple groups (a,c,d), or Student’s t test for comparison of variables between two groups (f). Representative images of three independent replicates were shown in immunoblotting analysis (b).
Extended Data Fig. 2 TLCA inhibits LPS triggered inflammatory response in THP-1 cells.
a-c, THP-1 cells were co-treated with LPS (1 μg/mL) and TLCA at the indicated concentrations for 24 h. Intracellular mRNA levels of IL-1β (a), mature IL-1β levels in supernatants (b), and indicated proteins in cell lysates (c) were measured; n = 4 biologically independent samples. d-f, THP-1 cells were co-treated with LPS (1 μg/mL) and TLCA (200 μM) and SBI-115 (TGR5 inhibitor; 25 μM) for 24 h. Intracellular mRNA of IL-1β (d), mature IL-1β in supernatants (e), and indicated proteins in cell lysates (f) were analyzed; n = 4 biologically independent samples. g, TGR5 knockdown THP-1 cells were treated with LPS (1 μg/mL) and TLCA (200 μM) for 24 h. Cell lysates were analyzed by immunoblotting with the indicated antibodies. Data were presented as mean ± s.d. The two-sided p values were examined using One-way ANOVA followed by Tukey’s multiple comparisons test for comparison of continuous variables among multiple groups (a,b,d,e). Representative images of three independent replicates were shown in immunoblotting analysis (c,f,g).
Extended Data Fig. 3 Effect analysis of TLCA on the IFN response in SFTSV-infected cells.
a-c, SFTSV-infected THP-1 cells were treated with TLCA, CDCA or DCA for 24 h. The levels of intracellular SFTSV RNA (a), IFNB mRNA (b), and ISG15 mRNA (c), were analyzed by RT-qPCR. d, Dot plot depicts differentially expressed genes (DEGs) in type I and III interferons pathway between groups. Symbols are labeled and sorted by adjusted p values. e,f, SFTSV-infected THP-1 cells were treated with TLCA, CDCA or DCA for 24 h. The mRNA levels of IFNL1 (e) and IFNG (f) were analyzed by RT-qPCR. Data were presented as mean ± s.d. The two-sided p values were examined using One-way ANOVA followed by Tukey’s multiple comparisons test for comparison of continuous variables among multiple groups (a-c,e,f).
Extended Data Fig. 4 TLCA depends on TGR5-PI3K/AKT-SREBP2 axis to block SFTSV replication.
a, Enhancing effect of TGR5 (INT-777) and Akt (sc79) agonists on the expression of SREBP2 and FADS2. Indicated protein in cells post-treated with INT-777 or sc79 at different time were measured. b,c, SFTSV-infected cells were treated with TGR5 (INT-777) and Akt (sc79) agonists at indicated concentrations for 24 h. Intracellular SFTSV RNA levels (b) and supernatant viral titers (c) were measured, respectively; n = 4 biologically independent samples. d, FADS2 knockdown THP-1 cells were infected with SFTSV and treated with INT-777 or sc79 for 24 h. Intracellular SFTSV RNA levels were examined by RT-qPCR; n = 4 biologically independent samples. e,f, SFTSV-infected THP-1 cells were treated with TLCA (200 μM) plus SBI-115 (TGR5 inhibitor; 20 μM) or TLCA plus T-βMCA (FXR inhibitor; 10 μM) for 24 h. Intracellular SFTSV RNA levels (e) and supernatant viral titers (f) were measured, respectively; n = 3 biologically independent samples. Data were presented as mean ± s.d. The two-sided p values were examined using One-way ANOVA followed by Tukey’s multiple comparisons test for comparison of continuous variables among multiple groups (b-d), or Student’s t test for comparison of variables between two groups (e,f). Representative images of three independent replicates were shown in immunoblotting analysis (a).
Extended Data Fig. 5 SFTSV induces ferroptosis in THP-1 cells and human PBMCs.
a,b, Levels of intracellular Fe2+ detected in SFTSV-infected THP-1 cells (a; n = 3 biologically independent samples) and hPBMCs (b; n = 6 biologically independent samples). c, Gating strategies for Fe2+ detection presented on Fig. 4a, m, Extended Data Figs. 5a, b and 8a. d, e, Levels of lipid ROS detected in SFTSV-infected THP-1 cells (d; n = 3 biologically independent samples) and hPBMCs (e; n = 6 biologically independent samples). f, Gating strategies for lipid ROS detection presented on Figs. 4c, n, p, 5a, c, e, g, i, k, l, Extended Data Figs. 5d, e, s–v, 7c, d, f, g and 8c. g–j, The mRNA levels of ferroptosis and iron metabolism related genes analyzed using RNA-seq data from THP-1 cells; n = 4 biologically independent samples. k,l, The mRNA levels of ferroptosis and iron metabolism related genes measured in THP-1 cells (k) and hPBMCs (l); n = 3 biologically independent samples. m,n, Lelves of mtROS detected in THP-1 cells (m; n = 3 biologically independent samples) and hPBMCs (n; n = 6 biologically independent samples) post infection with SFTSV (MOI = 1). o, Gating strategies for mitochondrial staining presented on Fig. 4i, j, l, and Extended Data Fig. 5m, n, p, q. p, q, The mitochondrial mass measured in THP-1 cells (p, n = 3 biologically independent samples) and hPBMCs (q, n = 6 biologically independent samples) post infection with SFTSV (MOI = 1). r, Gating strategies for mitochondrial mass detection presented on Fig. 4k. The change in fluorescence between (2) and (1) indicates the continuous activation of mitochondrial permeability transition pores. s,t, SFTSV-infected THP-1 cells (s, n = 3 biologically independent biologically independent samples) and hPBMCs (t, n = 6 biologically independent biologically independent samples) were incubated with Lip-1 (4 μM) or DFO (25 μM) for 72 h, and the lipid ROS was determined as above. u,v, SFTSV-infected THP-1 cells (u) and hPBMCs (v) were incubated with rotenone (10 nM) for 72 h, and the lipid ROS was determined as above; n = 3 biologically independent samples. Data were presented as mean ± s.d. The two-sided p values were examined using One-way ANOVA followed by Tukey’s multiple comparisons test for comparison of continuous variables among multiple groups (a,b,d,e,k,l,s-v), or Student’s t test for comparison of variables between two groups (g-j,m,n,p,q).
Extended Data Fig. 6 Transcriptomic analysis reveals the activation of oxidative cell death related pathway and cellular iron homeostasis pathway in SFTSV infected BMDMs.
a, GSEA analysis of the oxidative cell death pathway and cellular iron homeostasis pathway in SFTSV-infected BMDMs. b, Heatmap profiling of SFTSV-regulated genes related to ferroptosis and iron metabolism in BMDMs. Heatmap color code indicates variations of the log2 fold-change (log2FC) between each sample of two groups.
Extended Data Fig. 7 TLCA alleviates ferroptosis via FADS2 to inhibit SFTSV replication in THP-1 cells.
a, SFTSV-infected THP-1 cells were treated with Fe2+ (500 μM), RSL-3 (1 μM), DFO (20 μM) or Lip-1 (20 μM) for 24 h. Intracellular SFTSV RNA levels were measured with RT-qPCR; n = 3 biologically independent samples. b,c, THP-1 cells were infected with SFTSV and then treated with TLCA (200 μM) for 72 h. Levels of LDH release (b) and lipid ROS (c) were examined; n = 3 biologically independent samples. d,e, THP-1 cells treated with TLCA (100 μM) were incubated with Fe2+ (500 μM) or RSL-3 (100 nM) for 24 h. Lipid peroxidation (d) was assessed by flow cytometry after C11-BODIPY staining. Levels of cell death were analyzed via LDH release in the supernatants (e); n = 3 biologically independent samples. f-i, THP-1 cells treated with TLCA (100 μM) and sc-26196 (10 μM) were incubated with Fe2+ (500 μM) or RSL-3 (100 nM) for 24 h. Lipid peroxidation (f,g) and LDH release (h,i) were measured as above; n = 3 biologically independent samples. Data were presented as mean ± s.d. The two-sided p values were examined using One-way ANOVA followed by Tukey’s multiple comparisons test for comparison of continuous variables among multiple groups (a-i).
Extended Data Fig. 8 Assessment of Heartland virus infection mediated induction of ferroptosis and effects of TLCA and ferroptosis inhibitors on multiplication of Heartland virus.
a-c, THP-1 cells were infected with HRTV for indicated time. Levels of intracellular Fe2+ (a) were detected using flow cytometry, lipid peroxidation (b) was assessed by flow cytometry after C11-BODIPY staining, and levels of cell death were analyzed via LDH release in the supernatants (c); n = 3 biologically independent samples. d,e, HRTV-infected THP-1 cells were treated with different dose of TLCA for 24 h. Intracellular (d) and supernatant (e) SFTSV RNAs were measured with RT-qPCR; n = 3 biologically independent samples. f, HRTV-infected THP-1 cells were treated with RSL-3 (1 μM), DFO (20 μM) or Lip-1 (20 μM) for 24 h. Intracellular SFTSV RNA levels were measured with RT-qPCR; n = 3 biologically independent samples. Data were presented as mean ± s.d. The two-sided p values were examined using Student’s t test for comparison of variables between two groups (a-c), or One-way ANOVA followed by Tukey’s multiple comparisons test for comparison of continuous variables among multiple groups (d-f). R2 was estimated by a linear regression model (d,e).
Extended Data Fig. 9 Assessment of serum iron and ferritin levels as prognostic biomarkers for SFTSV infection.
a, Comparison of levels of various trace metals among 139 SFTS patients recruited in 2019, including surviving SFTS patients at acute phase of infection (Acute. Sur, n = 72) and recovery phase of infection (Recover. Sur, n = 26) and deceased SFTS patients at acute phase of infection (Acute. Dea, n = 41). b,c, Dynamic levels of aspartate transaminase (AST; b) and lactate dehydrogenase (LDH; c) were monitored in 165 SFTS patients recruited in 2022. A total of 817 serum samples serially collected at <3, 4–6, 7–9, 10–12, 13–15, 16–18, and 19–21 days since symptom onset in surviving (SF. Sur: n = 38, 225, 232, 123, 54, 21, and 8, respectively) and deceased (SF. Dea: n = 8, 60, 41, and 7, respectively) patients with SFTS. Data were presented as mean ± s.d. The two-sided p values were examined using Student’s t test for comparison of variables between two groups. d-f, Linear correlation between iron levels and ferritin (d), AST (e), and LDH (f), levels in serum in the cohort comprising 165 SFTS patients recruited in 2022. R2 was estimated by a linear regression model. g,h, Receiver operator characteristic curve (ROC) of ferritin, iron, AST, and LDH levels tested within 4–6 (g) and 7–9 (h) days after symptom onset in predicting fatal outcome of SFTS.
Extended Data Fig. 10 Assessment of SFTSV infection mediated induction of ferroptosis in mice and treatment effect of ferroptosis inhibitor and TLCA against SFTSV infection.
a,b, Serum levels of iron (a) and ferritin (b) at 5 dpi from uninfected or SFTSV-infected C57BL/6 J mice that were pretreated with anti-IFNAR1 IgG antibody (n = 6 per group). c,d, Transmission electron microscopy of mitochondria (c) and comparison of cristae numbers per mitochondria (d) in spleens from uninfected or SFTSV-infected mice at 5 dpi. The presented visual field is representative of two independent experiments, and the mitochondrial cristae number is calculated from four microscopic fields of each experiment. Yellow arrows indicate mitochondria with normal or abnormal morphology of crista. e, Representative hematoxylin and eosin (H&E) images from three biologically independent samples of lung, liver, and spleen sections from uninfected and SFTSV-challenged C57BL/6 J mice with (Lip-1 group) or without (Vehicle group) Lip-1 treatment at 5 dpi. f, Levels of ferroptosis and iron metabolism related proteins and SFTSV NP in spleens from uninfected and SFTSV-challenged C57BL/6 J mice with or without Lip-1 treatment at 5 dpi (n = 3 biologically independent samples). These proteins were not all run on the same blot, but samples derived from the same experiment and that blots were processed in parallel. g, MDA levels in splenic lysates from uninfected and SFTSV- challenged C57BL/6 J mice with or without Lip-1 treatment at 5 dpi (n = 6 per group). h, Levels of IL-1β production in spleens from uninfected and SFTSV-challenged C57BL/6 J mice with or without TLCA treatment determined with ELISA (n = 5 per group). i, Spleens from uninfected and TLCA- or vehicle-treated C57BL/6 J mice were analyzed by immunoblotting with the indicated antibodies (n = 3 biologically independent samples). These proteins were not all run on the same blot, but samples derived from the same experiment and that blots were processed in parallel. j, Viral titers in spleens from SFTSV-infected wild-type C57BL/6 J mice with or without TLCA treatment at 5 dpi (n = 5 per group). k, Representative images from three biologically independent samples of spleen sections from uninfected and SFTSV-challenged wild-type C57BL/6 J mice with or without TLCA treatment, stained with H&E or a rabbit polyclonal antibody against SFTSV NP. Data were presented as mean ± s.d. The two-sided p values were examined using Student’s t test for comparison of variables between two groups (a,b,d,j,), or One-way ANOVA followed by Tukey’s multiple comparisons test for comparison of continuous variables among multiple groups (g,h). The samples derived from the same experiment and the blots were processed in parallel (f,i).
Supplementary information
Supplementary Information
Supplementary Figs. 1–5, Supplementary Tables 1–3 and references.
Supplementary Data 1
Source data for Supplementary Fig. 2.
Supplementary Data 2
Source data for Supplementary Fig. 4.
Supplementary Data 3
Source data for Supplementary Fig. 5.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 2
Unprocessed western blots.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 3
Unprocessed western blots.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 4
Unprocessed western blots.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 1
Statistical source data.
Source Data Extended Data Fig. 1
Unprocessed western blots.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 2
Unprocessed western blots.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 4
Unprocessed western blots.
Source Data Extended Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 7
Statistical source data.
Source Data Extended Data Fig. 8
Statistical source data.
Source Data Extended Data Fig. 9
Statistical source data.
Source Data Extended Data Fig. 10
Statistical source data.
Source Data Extended Data Fig. 10
Unprocessed western blots.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zheng, X., Zhang, Y., Zhang, L. et al. Taurolithocholic acid protects against viral haemorrhagic fever via inhibition of ferroptosis. Nat Microbiol 9, 2583–2599 (2024). https://doi.org/10.1038/s41564-024-01801-y
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/s41564-024-01801-y
This article is cited by
-
Pilaralisib inhibits the replication of enteroviruses by targeting the PI3K/AKT signaling pathway
Virology Journal (2025)
-
Metabolomic evidence of bovine leukemia virus regulation on ferroptosis in bovine mammary epithelial cells
Veterinary Research Communications (2025)