Abstract
Glucose-6-phosphate dehydrogenase (G6PD) deficiency is one of the most common enzymopathies worldwide. Patients with G6PD deficiency are usually asymptomatic throughout their life but can develop acute hemolysis after exposure to free radicals or certain medications. Several studies have shown that serum miRNAs can be used as prognostic biomarkers in various types of hemolytic anemias. However, the impact of G6PD deficiency on circulating miRNA profiles is largely unknown. The present study aimed to assess the use of serum miRNAs as biomarkers for detecting hemolysis in the nonacute phase of G6PD deficiency. Patients with severe or moderate G6PD Viangchan (871G > A) deficiency and normal G6PD patients were enrolled in the present study. The biochemical hemolysis indices were normal in the three groups, while the levels of serum miR-451a, miR-16, and miR-155 were significantly increased in patients with severe G6PD deficiency. In addition, 3D analysis of a set of three miRNAs (miR-451a, miR-16, and miR-155) was able to differentiate G6PD-deficient individuals from healthy individuals, suggesting that these three miRNAs may serve as potential biomarkers for patients in the nonhemolytic phase of G6PD deficiency. In conclusion, miRNAs can be utilized as additional biomarkers to detect hemolysis in the nonacute phase of G6PD deficiency.
Similar content being viewed by others
Introduction
Glucose-6-phosphate dehydrogenase (G6PD) deficiency is one of the most common enzymopathies, affecting 400 million people worldwide1. The estimated prevalence of G6PD deficiency in Thailand ranges from 3 to 18%2. The G6PD Viangchan form is the most common variant in Thailand, accounting for approximately 54% of the identified mutations3. Individuals with G6PD deficiency are usually asymptomatic4, but hemolysis can be commonly triggered by certain medications and infections4,5. When G6PD-deficient erythrocytes are exposed to severe hypoxia, free radicals, or some medications, such as primaquine, erythrocytes become highly vulnerable to hemolysis4,5. For patients with severe hemolysis, prompt transfusions can substantially and rapidly improve their clinical course4. In Thailand, G6PD deficiency is typically diagnosed only in symptomatic patients, namely, in patients experiencing acute hemolytic attacks. Patients with G6PD deficiency and oxidative stress also experience damage to other organs, called extraerythrocytic manifestations, endothelial cell damage, infection, and coagulation cascade activation6,7,8. However, there are limited useful biomarkers for assessing hemolytic status in asymptomatic patients.
During hemolysis, numerous components of erythrocytes, including microRNAs (miRNAs), are released into the blood or extracellular space9. Previous reports have shown that circulating miRNAs can be used as diagnostic or prognostic biomarkers for various hemolytic anemias10,11,12,13, specifically miR-451a and miR-16, which are abundant in erythrocytes9,14. The serum miR-451a concentration has been shown to be elevated in patients with severe beta-thalassemia/HbE illness11,12. Low levels of miR-510 and miR-629 have been observed in patients with severe sickle cell disease10. However, the impact of G6PD deficiency on miRNA profiles is largely unknown. The present study aimed to characterize the serum miRNAs in patients with G6PD deficiency. Since G6PD deficiency primarily impacts erythrocytes, six blood cell-derived miRNAs were selected based on previous in vitro15,16,17,18,19,20,21,22,23,24 and clinical studies25. Specifically, miR-451a, miR-16, miR-155, and miR-144 are involved in regulating erythropoiesis18,22,24, regulating oxidative stress20,21,23, and maintaining erythrocyte homeostasis and lifespan16,19. Blood cells serve as major sources of circulating miRNAs and potentially contribute to the release of intracellular miRNAs from blood cells into the bloodstream25. Consequently, additional blood cell-derived miRNAs, such as miR-223 from granulocytes17 and miR-126 from platelets15, were also investigated.
Results
Clinical data of G6PD-deficient subjects
The clinical data of the 11 control subjects, 5 subjects with moderate G6PD deficiency, and 8 subjects with severe G6PD deficiency are shown in Table 1. The following mean ages of the patients were not significantly different among the groups: 29.38 ± 11.25 years in the severe G6PD-deficient group, 26.40 ± 12.64 years in the moderate G6PD-deficient group, and 24.73 ± 3.72 years in the normal group (Table 1). Moreover, the erythrocyte indices did not significantly differ among the subjects (P < 0.05), and the erythrocytes in all subjects were normochromic and normocytic. Patients with severe G6PD deficiency had significantly decreased G6PD activity (1.11 ± 0.90) compared to normal controls (15.96 ± 3.03; P < 0.001), while the G6PD activity of those with moderate G6PD deficiency (8.63 ± 0.67) did not significantly differ compared to normal controls.
Limitations of biochemical indices for detecting subclinical hemolysis
No differences in the serum levels of potassium (K +), aspartate transaminase (AST), and lactate dehydrogenase (LDH), which are used as biochemical markers of hemolysis, were found among the three groups. The free serum Hb levels did not differ among the three groups, with levels of 4.82 ± 2.20, 4.63 ± 1.18, and 4.84 ± 2.00 mg/dL for the normal (n = 11), moderate deficiency (n = 5), and severe deficiency (n = 8) groups, respectively. The average concentrations of the following parameters did not differ among the three groups: 4.34 ± 0.66 mEq/L potassium, 16.45 ± 3.24 U/L AST, and 154.27 ± 24.94 U/L LDH in the normal group; 4.00 ± 0.46 mEq/L potassium, 19.00 ± 6.75 U/L AST, and 138.60 ± 35.05 U/L LDH in the moderate deficiency group; and 4.48 ± 0.50 mEq/L potassium, 17.25 ± 2.24 U/L AST, and 145.13 ± 33.49 U/L LDH in the severe deficiency group (Table 2). These results indicated that hemolysis was undetectable with the NanoDrop method and biochemical testing in patients with G6PD deficiency.
Serum miRNA analyses
The level of miR-451a was significantly higher in patients with moderate (5.32 ± 0.95 fmol/µL, P = 0.049) or severe G6PD deficiency (6.11 ± 1.71 fmol/µL, P = 0.020) than in normal controls. Similarly, the levels of miR-16 were significantly higher in moderate cases (0.0005 ± 0.00006, P = 0.049) and severe cases (0.0006 ± 0.00008, P = 0.007) than in normal cases, and the level of miR-155 was significantly higher in the severe group (0.003 ± 0.0004, P = 0.002) than in the normal control group (Fig. 1A). These results showed that the serum levels of erythrocyte-related miRNAs, especially miR-451a, increased in patients with G6PD deficiency even during the nonacute hemolytic phase. The level of miR-223, which is expressed mainly in granulocytes, was not significantly increased in patients with moderate (3.503 ± 0.832) or severe G6PD deficiency (2.451 ± 0.407 fmol/µL). The level of platelet-related miR-126 significantly increased in moderate cases (0.225 ± 0.041 fmol/µL, P = 0.006) but not in severe cases (0.132 ± 0.022 fmol/µL). Similarly, the level of miR-144 was not significantly altered in moderate (0.017 ± 0.008 fmol/µL) or severe cases (0.024 ± 0.007 fmol/µL) (Fig. 1B). Additionally, the elevation of these serum miRNAs was not influenced by confounding factors, such as the age of the recruited subjects (Supplementary Fig. 1).
Increased levels of serum miR-451a, miR-16, and miR-155 in patients with G6PD deficiency. The ∆Ct values of the miRNAs were calculated using the Ct values of the internal control cel-miR-39. (A) Patients with G6PD deficiency had significantly higher serum miR-451a, miR-16, and miR-155 levels compared to the normal control (Ctrl) subjects, whereas (B) the levels of miR-223, miR-126, and miR-144 did not significantly differ. The statistical analyses were performed using the Kruskal‒Wallis test. A P value < 0.05 was considered to indicate statistical significance.
Correlation between miRNA levels and G6PD activity
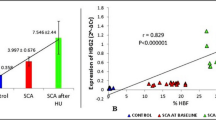
To evaluate the relationship between miRNA expression and G6PD activity, correlation coefficients for miR-451a expression were determined. There were significant associations of G6PD enzyme activity with miR-451a (r2 = 0.3319, P = 0.005) (Fig. 2A), miR-16 (r2 = 0.3988, P = 0.001) (Fig. 2B), and miR-155 (r2 = 0.3916, P = 0.001) levels (Fig. 2C).
MiR-451a, miR-16, and miR-155 as promising biomarkers
To evaluate the prognostic value of serum miR-451, miR-16, or miR-155, the delta Ct values of normal and G6PD-deficient subjects were used to generate receiver operating characteristic (ROC) curves. The miR-16 data separated G6PD-deficient patients from normal individuals with a high sensitivity and an area under the curve (AUC) of 0.902 (P = 0.0009) (Fig. 3A). The miR-451a and miR-155 levels distinguished the normal and G6PD-deficient groups with AUC values of 0.881 (P = 0.0024) and 0.874 (P = 0.0019), respectively. Among the three miRNAs, miR-16 exhibited the highest sensitivity in detecting G6PD deficiency. We determined the optimal cut-off value for these three miRNAs based on high sensitivity and specificity, as well as Youden's index26, as indicated in Supplementary Information 2. The cut-off values for distinguishing patients with G6PD deficiency using miR-451a (2.73 fmol/µL), miR-16 (0.00041 fmol/µL), and miR-155 (0.00155 fmol/µL) are shown in Table 3. Using all three miRNAs as a set separated the G6PD-deficient group from the normal group (Fig. 3B, black squares), but this set of three miRNAs did not distinguish moderate from severe disease. These findings suggested that the panel of three miRNAs (miR-451a, miR-16, and miR-155) has the potential to distinguish G6PD-deficient patients, particularly those with severe G6PD deficiency.
A set of three miRNAs (miR-451a, miR-16, and miR-155) distinguishes G6PD-deficient individuals from normal controls. (A) ROC curve analysis of miR-451a, miR-16, and miR-155 levels. The sensitivity and specificity were calculated to determine the AUC with 95% confidence intervals (CIs). A P value < 0.05 was considered to indicate statistical significance. (B) A 3D scatter plot of the three miRNAs was generated using SPSS version 22 to identify patients with G6PD deficiency.
Discussion
In Thailand, the prevalence of G6PD deficiency ranges from 3 to 18%2 in adults to 10.7–17.0% in newborns3,27,28. The G6PD Viangchan (871G > A) variant is the most prevalent variant in the Thai population and is associated with severe disease3,29. Most G6PD-deficient adults are asymptomatic in their normal state and are unaware of this hereditary disease throughout their life4. Approximately 65% of G6PD-deficient Thai newborns exhibit severe neonatal jaundice, and 21.2–22% develop hyperbilirubinemia3,30. Therefore, screening for G6PD deficiency in newborns with severe hyperbilirubinemia is important. G6PD is the key enzyme for regulating glutathione sulfhydryl (GSH) levels in erythrocytes. Low G6PD activity increases the sensitivity of mature erythrocytes to oxidative stress, resulting in hemolysis in vivo, and an in vitro erythroid culture system has shown that G6PD is dispensable for the production of erythrocytes from erythroid progenitors31. In adulthood, individuals with G6PD deficiency do not have any hematological parameters32,33. The present study demonstrated that all complete blood count (CBC) data, including hemoglobin (Hb) levels and other red blood cell parameters, were within the normal range, consistent with the findings of previous studies29,33. Often with no obvious signs of hemolysis, G6PD-deficient patients are particularly vulnerable to hemolysis when exposed to oxidative agents.
Hemoglobin (Hb) is the major erythrocyte protein that accounts for nearly one-third of the weight of an erythrocyte34. The level of free Hb is a useful clinical biomarker of intravascular hemolysis, whereas the serum unconjugated bilirubin concentration is useful for estimating extravascular hemolysis35. The present study demonstrated that there was no difference in the hemolysis indices or free Hb levels in the nonacute phase of G6PD deficiency (Table 2), which were less than 5 mg/dL, and these results were consistent with the findings of previous studies36. However, the levels of miR-451a, miR-16, and miR-155 were elevated in G6PD-deficient patients (Fig. 1A). Because these miRNAs are abundant in mature erythrocytes14,18, the present findings suggested that the serum miR-451a, miR-16, and miR-155 levels increase due to subclinical hemolysis in G6PD-deficient patients even during the nonacute hemolytic phase. For other blood cell miRNAs, the present data showed that the levels of miR-144 (abundant in immature erythroid cells), miR-223 (derived from granulocytes), and miR-126 (derived from platelets) were not different from those in normal subjects. The altered expression of circulating miRNAs has been linked to the age of subjects37. We aimed to mitigate potential confounding factors, such as age. Our results demonstrated that age was not correlated with the levels of miRNAs in G6PD-deficient patients, as depicted in Supplementary Fig. 1. Additionally, the function of miRNAs released from circulating RBCs remains unclear. One possible function of miR-451a in RBCs is the suppression of oxidization because one of the target genes of miR-451a, namely, FOXO3, is related to the antioxidative pathway23. Several reports have shown that miR-451a binds to the Ago2 protein and is protected from the catalytic activity of RNase38,39. Circulating miR-451a may act as an antioxidant molecule if it is transferred to cells in distant organs as well as to RBCs.
Previous studies have demonstrated that microRNAs are linked to pathophysiology, and several studies have characterized disease-specific abnormalities in plasma miRNAs40,41. In various hemolytic anemias, including thalassemia, sickle cell anemia, and paroxysmal nocturnal hemoglobinuria (PNH), abnormal miRNA profiles have been investigated. For example, elevated levels of circulating miR-451a are associated with severe types of beta-thalassemia11,12. A previous study has shown that low levels of miR-510 and miR-629 are associated with a greater risk of severe sickle cell disease 10. MiR-148b-3p and miR-126-3p are more differentially expressed in PNH patients than in control subjects13. In patients with normal erythropoiesis, miR-155 and miR-451 play crucial roles in erythroid differentiation. miR-451 is upregulated in a lineage-specific manner during erythroid maturation, while miR-155 is downregulated during the early stages of erythropoiesis18,24. Due to ineffective erythropoiesis in thalassemia, significantly higher levels of plasma miR-451 and miR-155 are observed in β0-thalassemia/HbE patients11. Elevated plasma miR-451 levels may originate from the destruction of erythroid cells, while increased serum miR-155 levels can be attributed to the high expression of miR-155 in proliferating nucleated erythroblasts11. Concerning G6PD deficiency, our previous study confirmed no signs of ineffective erythropoiesis. The CD34-positive hematopoietic stem and progenitor cells from patients with G6PD deficiency could differentiate into mature erythrocytes in vitro31. Although severe G6PD-deficient patients did not exhibit increased hemolysis or ineffective erythropoiesis in the nonacute phase, the present findings of severe G6PD deficiency revealed increased levels of miR-451a (approximately 6 fmol/µL). Takada et al. (2021) reported that miR-451a levels are increased in individuals with other hemolytic disorders, such as autoimmune hemolytic anemia (AIHA) (30 fmol/µL), PNH (320 fmol/µL), α-thalassemia (280 fmol/µL), β-thalassemia (150 fmol/µL), and malaria (60 fmol/µL)12. The levels of miR-451a in patients with severe G6PD deficiency are lower than those reported in other hemolytic disorders characterized by intravascular hemolysis, such as AIHA, PNH, malaria infection, and the ineffective erythropoiesis observed in β-thalassemia. Further analysis including acute-phase patients is necessary to confirm the clinical relevance of the analysis of these miRNAs in patients with G6PD deficiency.
Drug-induced hemolysis caused by G6PD deficiency often occurs between 24 and 72 h after exposure to antimalarial drugs42, and G6PD levels may normalize during an acute hemolytic episode due to the increased G6PD activity in reticulocytes compared to that in mature red blood cells. Using serum miRNAs during the crisis phase of the drug-induced hemolytic phase may be helpful in the future to identify which patients have G6PD deficiency. Additionally, the heterozygous genotype is generated by inheritance, whereas the phenotype is defined by the pattern of X chromosome inactivation. Knowing heterozygosity in a female's genotype does not provide a reliable prediction of whether she will experience severe hemolysis with primaquine43. However, if a patient tends to have G6PD deficiency, the circulating miRNA concentration can be used as a predictive factor.
Screening for G6PD deficiency is not necessarily performed for all newborns in Thailand. The WHO has recommended routine screening for G6PD deficiency in infants residing in areas where the prevalence of the condition is as high as 3–5% in males to prevent adverse effects44. The fluorescent spot test and the enzyme activity assay are effective at detecting G6PD deficiency in newborns45,46. Although these technologies are sufficiently accurate and sensitive for screening, a multistep protocol that includes an erythrocyte hemolysis step is required. Circulating miRNAs have distinct advantages as potential clinical biomarkers because they are sensitive and accurate, and they can be measured with minimal blood volume. In Thailand, the diagnosis of G6PD deficiency typically occurs in symptomatic patients, often coinciding with neonatal jaundice. Nationwide neonatal screening for G6PD deficiency has not been implemented2. Consequently, there are avoidable hospital costs associated with phototherapy and hospitalization when infants are subsequently determined to have normal G6PD status. In particular, it would be beneficial to employ this panel of three miRNAs for distinguishing severe G6PD-deficient patients from healthy individuals, as shown in Fig. 3A,B. Therefore, the utilization of miRNA analysis may contribute to the development of an efficient, cost-effective technology that minimizes blood volume for identifying newborns with G6PD deficiency in the future.
However, hemolysis frequently occurs at the preanalytical step of blood sample processing and influences the levels of certain miRNAs detectable in serum and plasma9,25. Takada et al. demonstrated that miR-451a expression increases 1.62-fold before serum separation for 1 h after blood draws12. The present data showed that the fold increase in miR-451a expression was significantly greater than that in normal controls (10.65 and 12.21 for moderate and severe G6PD deficiency, respectively) (data not shown), which indicated that the increase in miR-451a expression was not a result of sample processing. Thus, the elevated levels of serum miR-451a in hemolytic disease patients were sufficiently high to distinguish pathological hemolysis from preanalytical variation. To standardize the use of miRNAs for detecting subclinical hemolysis, studies with larger cohorts and longitudinal studies of G6PD-deficient patients, including those in stable and acute hemolytic phases, are needed. Moreover, the utility of this set of miRNAs for disease specificity and their applicability to other hemolytic conditions should be confirmed through further in vitro and animal model studies.
In conclusion, the present study demonstrated for the first time that a set of three miRNAs (miR-451a, miR-16, and miR-155) is useful for identifying subclinical hemolysis in G6PD-deficient subjects. Overall, miRNA-based biomarkers show promise as tools for identifying individuals in the non-acute phase of G6PD deficiency. These miRNAs could aid in translational medicine for patients experiencing hemolytic crises by integrating miRNA data with family history, hematological indices, and changes in hemoglobin levels. Additionally, determining G6PD status through miRNA analysis would be advantageous by enabling patients to avoid oxidative exposure. In addition, large cohort studies of miRNAs may help discriminate other hemolytic diseases, such as pyruvate kinase deficiency, hereditary spherocytosis, or elliptocytosis.
Methods
Subjects
The present study enrolled 13 patients with the G6PD Viangchan (871G > A) variant (hemizygote = 7, heterozygote = 5, homozygote = 1) and 11 subjects with normal G6PD. Eight of the thirteen subjects with the Viangchan variant had severe G6PD deficiency, and five of these subjects had moderate G6PD deficiency. The genetic data of all the subjects were obtained in a previous study47. Patients with hemoglobinopathies and other noncommunicable chronic diseases were excluded from the present study47. None of the enrolled subjects had been hospitalized for more than a month or had concomitant infections. All procedures involving human subjects were performed in accordance with the ethical standards of the Helsinki Declaration of 1975. The research protocol was approved by the Ethical Review Committees for research involving human subjects at Chulalongkorn University (COA no. 200/65 and COA no. 196/66) and the International University of Health and Welfare (22-Ifh-050). Prior to participation, all the subjects provided informed consent after receiving information on the purpose, potential risks, and benefits of the study.
Measurement of G6PD activity
EDTA blood samples were collected for measurements of G6PD activity, which determined the kinetic change in NADP + to NADPH within 10 min48. NADPH was detected at a wavelength of 340 nm using a Thermo Evolution 600 UV‒Vis Spectrophotometer. G6PD activity is expressed as international units per gram of hemoglobin (IU/g Hb). According to World Health Organization (WHO) guidelines49, G6PD deficiency was defined as G6PD activity less than 1.5 IU/g Hb.
Serum preparation for miRNA analyses
Serum obtained from clotted whole blood was used for miRNA analyses12. Briefly, 12 mL of whole blood was drawn using a vacuum sample tube containing coagulation stimulators. The serum was separated from the primary tube within 1 h by centrifugation at 3,500 × g for 10 min. To remove cell debris, the serum was centrifuged at 10,000 × g for 5 min at 4 °C, and the cleared supernatant was transferred to a new tube prior to miRNA analyses.
Measurement of hemolysis indices
Serum (600 µL) was used for measurements of hemolysis indices, including aspartate aminotransferase (AST), lactate dehydrogenase (LDH), and potassium (K +) levels. The serum free Hb concentration was measured at an optic density of 414 nm by using a NanoDrop apparatus12,50 (ND2000c, Thermo Fisher Scientific Co., Waltham, MA, USA).
MiRNA analyses
miRNA levels were determined using reverse transcriptase-based quantitative polymerase chain reaction (RT‒qPCR)11,12. Briefly, serum miRNAs were extracted with a Nucleospin™ plasma extraction kit (Macherey–Nagel, Takara, Shiga, Japan). One femtomole of cel-miR-39 (CosmoBio, Tokyo, Japan) was added as the spiked-in control during the extraction process. Purified miRNAs were converted into complementary DNAs using a TaqMan reverse transcriptase kit (Thermo Fisher Scientific Co., Waltham, MA, USA). qPCR analysis was performed using TaqMan microRNA assay kits and Universal PCR Master Mix (Thermo Fisher Scientific, Waltham, MA, USA). After the initial denaturation step at 95 °C for 10 min, 40 PCR cycles were performed at 95 °C for 15 s and 60 °C for 60 s, using a real-time PCR machine (ABI7500fast, Thermo Fisher Scientific Co., Waltham, MA, USA). Each miRNA measurement was performed in triplicate. As a negative control, nuclease-free water was used. The expression of each miRNA was compared to the expression of the spiked in cel-miR-39 using the comparative Ct method11. The absolute levels of each miRNA were calculated using a concentration of 0.5 fmol/μL cel-miR-39.
Statistical analyses
The normality of the data was tested using the Kolmogorov‒Smirnov test. Kruskal‒Wallis tests were used to evaluate differences among patients with normal G6PD, moderate G6PD deficiency, and severe G6PD deficiency. Receiver operating characteristic (ROC) curves were constructed to evaluate the diagnostic accuracy of each miRNA. Differences were considered significant at P < 0.05. The data were visualized using Prism software (version 8.0; GraphPad Software, Inc., CA, USA).
Data availability
The data presented in this study are available upon request from the corresponding author.
References
Gomez-Manzo, S. et al. Glucose-6-phosphate dehydrogenase: Update and analysis of new mutations around the world. Int. J. Mol. Sci. https://doi.org/10.3390/ijms17122069 (2016).
Tanphaichitr, V. S. Glucose-6-phosphate dehydrogenase deficiency in Thailand; its significance in the newborn. Southeast Asian J. Trop. Med. Public Health 30(Suppl 2), 75–78 (1999).
Nuchprayoon, I., Sanpavat, S. & Nuchprayoon, S. Glucose-6-phosphate dehydrogenase (G6PD) mutations in Thailand: G6PD Viangchan (871G>A) is the most common deficiency variant in the Thai population. Hum. Mutat. 19, 185. https://doi.org/10.1002/humu.9010 (2002).
Cappellini, M. D. & Fiorelli, G. Glucose-6-phosphate dehydrogenase deficiency. Lancet 371, 64–74. https://doi.org/10.1016/S0140-6736(08)60073-2 (2008).
Luzzatto, L. & Seneca, E. G6PD deficiency: A classic example of pharmacogenetics with on-going clinical implications. Br. J. Haematol. 164, 469–480. https://doi.org/10.1111/bjh.12665 (2014).
Dore, M. P., Parodi, G., Portoghese, M. & Pes, G. M. The controversial role of glucose-6-phosphate dehydrogenase deficiency on cardiovascular disease: a narrative review. Oxid. Med. Cell Longev. 2021, 5529256. https://doi.org/10.1155/2021/5529256 (2021).
Spolarics, Z. et al. Increased incidence of sepsis and altered monocyte functions in severely injured type A- glucose-6-phosphate dehydrogenase-deficient African American trauma patients. Crit. Care Med. 29, 728–736. https://doi.org/10.1097/00003246-200104000-00005 (2001).
Talwar, M. et al. Severe acute kidney injury owing to rhabdomyolysis and intravascular haemolysis in an 11-year-old child with G6PD deficiency. Paediatr. Int. Child Health 39, 150–153. https://doi.org/10.1080/20469047.2018.1439804 (2019).
Kirschner, M. B. et al. The impact of hemolysis on cell-free microRNA biomarkers. Front. Genet. 4, 94. https://doi.org/10.3389/fgene.2013.00094 (2013).
Biassi, T. P. et al. miRNA profile and disease severity in patients with sickle cell anemia. Ann. Hematol. 101, 27–34. https://doi.org/10.1007/s00277-021-04665-y (2022).
Leecharoenkiat, K. et al. Plasma microRNA-451 as a novel hemolytic marker for beta0-thalassemia/HbE disease. Mol. Med. Rep. 15, 2495–2502. https://doi.org/10.3892/mmr.2017.6326 (2017).
Takada, Y. et al. Pre-analytical modification of serum miRNAs: diagnostic reliability of serum miRNAs in hemolytic diseases. J. Clin. Med. https://doi.org/10.3390/jcm10215045 (2021).
Teruel-Montoya, R. et al. Differential miRNA expression profile and proteome in plasma exosomes from patients with paroxysmal nocturnal hemoglobinuria. Sci. Rep. 9, 3611. https://doi.org/10.1038/s41598-019-40453-5 (2019).
Azzouzi, I. et al. Deep sequencing and proteomic analysis of the microRNA-induced silencing complex in human red blood cells. Exp. Hematol. 43, 382–392. https://doi.org/10.1016/j.exphem.2015.01.007 (2015).
Czajka, P. et al. MicroRNA as potential biomarkers of platelet function on antiplatelet therapy: A review. Front. Physiol. 12, 652579. https://doi.org/10.3389/fphys.2021.652579 (2021).
Jafri, I., Alsharif, G., Bland, G.N.-L. & Gambhir, K. K. Erythrocyte miRNA 144 and miRNA 451 as cell aging biomarkers in African American Adults. Open Biochem. J. https://doi.org/10.2174/1874091X01913010081 (2019).
Johnnidis, J. B. et al. Regulation of progenitor cell proliferation and granulocyte function by microRNA-223. Nature 451, 1125–1129. https://doi.org/10.1038/nature06607 (2008).
Masaki, S., Ohtsuka, R., Abe, Y., Muta, K. & Umemura, T. Expression patterns of microRNAs 155 and 451 during normal human erythropoiesis. Biochem. Biophys. Res. Commun. 364, 509–514. https://doi.org/10.1016/j.bbrc.2007.10.077 (2007).
Rasmussen, K. D. et al. The miR-144/451 locus is required for erythroid homeostasis. J. Exp. Med. 207, 1351–1358. https://doi.org/10.1084/jem.20100458 (2010).
Sangokoya, C., Telen, M. J. & Chi, J. T. microRNA miR-144 modulates oxidative stress tolerance and associates with anemia severity in sickle cell disease. Blood 116, 4338–4348. https://doi.org/10.1182/blood-2009-04-214817 (2010).
Srinoun, K. et al. miR-144 regulates oxidative stress tolerance of thalassemic erythroid cell via targeting NRF2. Ann. Hematol. 98, 2045–2052. https://doi.org/10.1007/s00277-019-03737-4 (2019).
Svasti, S. et al. Expression of microRNA-451 in normal and thalassemic erythropoiesis. Ann. Hematol. 89, 953–958. https://doi.org/10.1007/s00277-010-0980-7 (2010).
Yu, D. et al. miR-451 protects against erythroid oxidant stress by repressing 14–3-3zeta. Genes Dev. 24, 1620–1633. https://doi.org/10.1101/gad.1942110 (2010).
Zhan, M., Miller, C. P., Papayannopoulou, T., Stamatoyannopoulos, G. & Song, C. Z. MicroRNA expression dynamics during murine and human erythroid differentiation. Exp. Hematol. 35, 1015–1025. https://doi.org/10.1016/j.exphem.2007.03.014 (2007).
Pritchard, C. C. et al. Blood cell origin of circulating microRNAs: A cautionary note for cancer biomarker studies. Cancer Prev. Res. (Phila) 5, 492–497. https://doi.org/10.1158/1940-6207.CAPR-11-0370 (2012).
Hajian-Tilaki, K. Receiver operating characteristic (ROC) curve analysis for medical diagnostic test evaluation. Caspian J. Intern. Med. 4, 627–635 (2013).
Charoenkwan, P., Tantiprabha, W., Sirichotiyakul, S., Phusua, A. & Sanguansermsri, T. Prevalence and molecular characterization of glucose-6-phosphate dehydrogenase deficiency in northern Thailand. Southeast Asian J. Trop. Med. Public Health 45, 187–193 (2014).
Thielemans, L. et al. Diagnostic performances of the fluorescent spot test for G6PD deficiency in newborns along the Thailand-Myanmar border: A cohort study. Wellcome Open Res. 3, 1. https://doi.org/10.12688/wellcomeopenres.13373.1 (2018).
Nuinoon, M. et al. Prevalence of G6PD deficiency and G6PD variants amongst the southern Thai population. PeerJ 10, e14208. https://doi.org/10.7717/peerj.14208 (2022).
Prachukthum, S. et al. Genetic polymorphisms in Thai neonates with hyperbilirubinemia. Acta Paediatr. 98, 1106–1110. https://doi.org/10.1111/j.1651-2227.2009.01275.x (2009).
Boonpeng, K., Ketprasit, N., Palasuwan, A., Kulkeaw, K. & Palasuwan, D. Glucose-6-phosphate dehydrogenase is dispensable for human erythroid cell differentiation in vitro. Exp. Hematol. 121, 18-29 e12. https://doi.org/10.1016/j.exphem.2023.02.002 (2023).
Kotepui, M., Uthaisar, K., PhunPhuech, B. & Phiwklam, N. Prevalence and hematological indicators of G6PD deficiency in malaria-infected patients. Infect. Dis. Poverty 5, 36. https://doi.org/10.1186/s40249-016-0130-0 (2016).
Mungkalasut, P. et al. Haematological profile of malaria patients with G6PD and PKLR variants (erythrocytic enzymopathies): A cross-sectional study in Thailand. Malar. J. 21, 250. https://doi.org/10.1186/s12936-022-04267-7 (2022).
Pishchany, G. & Skaar, E. P. Taste for blood: Hemoglobin as a nutrient source for pathogens. PLoS Pathog 8, e1002535. https://doi.org/10.1371/journal.ppat.1002535 (2012).
Barcellini, W. & Fattizzo, B. Clinical applications of hemolytic markers in the differential diagnosis and management of hemolytic anemia. Dis. Markers 2015, 635670. https://doi.org/10.1155/2015/635670 (2015).
Tzounakas, V. L. et al. Redox status, procoagulant activity, and metabolome of fresh frozen plasma in glucose 6-phosphate dehydrogenase deficiency. Front Med. (Lausanne) 5, 16. https://doi.org/10.3389/fmed.2018.00016 (2018).
Noren Hooten, N. et al. Age-related changes in microRNA levels in serum. Aging (Albany NY) 5, 725–740. https://doi.org/10.18632/aging.100603 (2013).
Ha, M. & Kim, V. N. Regulation of microRNA biogenesis. Nat. Rev. Mol. Cell Biol. 15, 509–524. https://doi.org/10.1038/nrm3838 (2014).
Jee, D. et al. Dual strategies for argonaute2-mediated biogenesis of erythroid miRNAs underlie conserved requirements for slicing in mammals. Mol. Cell 69, 265-278 e266. https://doi.org/10.1016/j.molcel.2017.12.027 (2018).
Arroyo, A. B. et al. microRNAs in the haemostatic system: More than witnesses of thromboembolic diseases?. Thromb. Res. 166, 1–9. https://doi.org/10.1016/j.thromres.2018.03.021 (2018).
Szabo, D. R. et al. Analysis of circulating microRNAs in adrenocortical tumors. Lab. Invest. 94, 331–339. https://doi.org/10.1038/labinvest.2013.148 (2014).
Poirot, E. et al. Risks of hemolysis in glucose-6-phosphate dehydrogenase deficient infants exposed to chlorproguanil-dapsone, mefloquine and sulfadoxine-pyrimethamine as part of intermittent presumptive treatment of malaria in infants. PLoS One 10, e0142414. https://doi.org/10.1371/journal.pone.0142414 (2015).
Chu, C. S., Bancone, G., Nosten, F., White, N. J. & Luzzatto, L. Primaquine-induced haemolysis in females heterozygous for G6PD deficiency. Malar. J. 17, 101. https://doi.org/10.1186/s12936-018-2248-y (2018).
Glucose-6-phosphate dehydrogenase deficiency. WHO Working Group. Bull World Health Organ 67, 601–611 (1989).
Beutler, E. A series of new screening procedures for pyruvate kinase deficiency, glucose-6-phosphate dehydrogenase deficiency, and glutathione reductase deficiency. Blood 28, 553–562 (1966).
Minucci, A., Giardina, B., Zuppi, C. & Capoluongo, E. Glucose-6-phosphate dehydrogenase laboratory assay: How, when, and why?. IUBMB Life 61, 27–34. https://doi.org/10.1002/iub.137 (2009).
Palasuwan, D. et al. Impairment of invasion and maturation and decreased selectivity of plasmodium falciparum in G6PD Viangchan and Mahidol variants. J. Infect. Dis. 225, 1238–1247. https://doi.org/10.1093/infdis/jiab484 (2022).
Sharma, U., Mishra, S., Gautam, N. & Gupta, B. K. Qualitative and quantitative assay of glucose 6 phosphate dehydrogenase in patients attending tertiary care center. BMC Res. Notes 13, 298. https://doi.org/10.1186/s13104-020-05145-8 (2020).
WHO Scientific Group on the Standardization of Procedures for the Study of Glucose-6-Phosphate Dehydrogenase & World Health Organization. World Health Organization technical report series: no. 366 (World Health Organization, Geneva, 1967).
Shah, J. S., Soon, P. S. & Marsh, D. J. Comparison of methodologies to detect low levels of hemolysis in serum for accurate assessment of serum microRNAs. PLoS One 11, e0153200. https://doi.org/10.1371/journal.pone.0153200 (2016).
Acknowledgements
This research was funded by the Thailand Science Research and Innovation Fund Chulalongkorn University. This research project was supported by the National Research Council of Thailand (NRCT) through the Royal Golden Jubilee (RGJ) PhD Program (Grant No. PHD/0116/2560) and International University of Health and Welfare Research Funding, Kaken (Nos. 17K09020, 20K08431, and 17H07059). We sincerely thank Dr. Sumio Hoka and Dr. Hiroyuki Tsutsui (President of Kouhoukai Takagi Hospital, Okawa, Japan), as well as Prof. Kazuma Fujimoto (Director, International University of Health and Welfare, Okawa, Japan), for administrative support and scientific discussion. We also thank Mr. Kozo Ishida, President of North Kyoto Rotary Club, Japan, for discussion and encouragement. We are grateful to all the members of the Department of Medical Technology and Sciences, International University of Health and Welfare for their support and assistance with this study.
Author information
Authors and Affiliations
Contributions
K.B. performed the experiments, analyzed the data, and wrote the manuscript; T.S. and Y.H. analyzed the data; K.K. reviewed the manuscript; D.P. collected samples, interpreted the data, wrote and reviewed the manuscript; T.U. designed the experiment and wrote the manuscript. All the authors critically reviewed and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that there are no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Boonpeng, K., Shibuta, T., Hirooka, Y. et al. Serum microRNAs as new biomarkers for detecting subclinical hemolysis in the nonacute phase of G6PD deficiency. Sci Rep 14, 16029 (2024). https://doi.org/10.1038/s41598-024-67108-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-67108-4