Abstract
ApoB insertion/deletion (ins/del) genetic variant (rs11279109) is thought to be related to cardio-metabolic markers and obesity. This association has the potential to be modified by dietary patterns. Since the majority of studies concerned the role of dietary acid load (DAL) or ApoB in type 2 diabetes mellitus (T2DM) and its complications independently, and due to the insufficient data regarding the possible interactions between ApoB genetic variants and DAL on anthropometric and metabolic markers, we aimed to study the interaction between this genetic variant and dietary acid load (DAL) on cardio-metabolic markers, along with leptin among Iranian individuals with T2DM. 700 T2DM patients were randomly recruited. A validated semi-quantitative food frequency questionnaire was used for DAL calculation including potential renal acid load (PRAL) and net-endogenous acid production (NEAP). The polymerase chain reaction was used for genotyping the ApoB ins/del (rs11279109). The general linear model was applied to find the interactions in the crude and adjusted models. Patients with del/del genotype (rs11279109) with high PRAL intake have lower low-density lipoprotein cholesterol (LDL-C) (Pinteraction = 0.004), LDL/HDL ratio (Pinteraction = 0.02), total cholesterol (TC) (Pinteraction = 0.04), triglyceride (TG) (Pinteraction = 0.04), leptin (Pinteraction = 0.04) and interleukin-18 (IL-18) (Pinteraction = 0.04). Moreover, the interaction of gene and DAL in the PRAL method on TG concentration (P = 0.04), waist circumference (WC) (P = 0.04), and LDL/HDL ratio (P = 0.04) were significant. Eventually, a positive relationship was observed between the presence of the del/del genotype (rs11279109) and higher levels of TG, TC, LDL-C, IL-18, and LDL/HDL, in individuals with lower adherence to DAL, after adjusting for various covariates. Further studies are needed to investigate and confirm these findings.
Similar content being viewed by others
Introduction
In recent decades, the prevalence and incidence of type 2 diabetes mellitus (T2DM) have steadily risen1 and appear to be associated with a 1.3 to 2 times higher risk of mortality, mostly resulting from cardiovascular diseases (CVDs)2. Since there is a clear link between CVDs and T2DM, treatment goals must focus on CVD risk reduction3,4,5. The lipoprotein abnormalities, which commonly occur in T2DM, may be considered a result of lipoprotein lipase enzyme deficiency and modulation of lipid metabolism-related genes in hyperglycemia6,7,8. Also, the inflammatory state contributes significantly to the progression of atherosclerosis and is involved in various metabolic disorders related to diabetes9. T2DM is the result of both environmental and genetic risk factors as well as gene-environment interactions10. Genome-wide association studies suggested the Apolipoprotein B (ApoB) gene as one of the main candidate genes predisposing individuals to dyslipidemia11,12. ApoB is a structural protein and acts as an integral part of chylomicrons during lipoprotein metabolism. It is required for the synthesis of triglyceride-rich (TG-rich) lipoproteins such as very low-density lipoprotein (VLDL) in the liver13. It has been shown that serum ApoB concentration directly reflects the amount of low-density lipoprotein (LDL) particles in the plasma14. Elevated levels of these atherogenic lipoproteins trigger arterial inflammation by enhancing the migration of monocytes and T lymphocytes to the endothelial surface and the transformation of monocytes/macrophages into foam cells, which secrete several plasma inflammatory markers15. There are multiple genetic variants at the ApoB gene locus13,16. The insertion/deletion of 3 amino acid residues (leucine-alanine-leucine) in the ApoB signal peptide forms the insertion/deletion (ins/del) genetic variant (rs11279109) which is a common genetic variant and located in the first exon of the ApoB gene7,17. The hydrophobicity and translocation of the synthesized ApoB from the endoplasmic network to the cytoplasm were decreased in the del allele through LDL clearance reduction and increasing TG accumulation in the liver, causing a greater uptake of TG by adipose tissues and obesity18,19,20,21,22. It is also proposed that the del allele is related to a decrease and an increase in the secretion and degradation of ApoB, respectively23,24,25,26,27,28. It is additionally reported that ApoB ins/del genetic variant (rs11279109) is likely to be determined as a significant factor causing variety in the TG and insulin levels, especially in response to dietary intake29. A meta-analysis reported an elevated level of LDL, ApoB, and risk of CHD among the ins/del genotype (rs11279109)30, however, a few studies showed no association between the ApoB ins/del genetic variant (rs11279109) and serum lipid levels31,32. These controversial results reveal the necessity of more gene-diet interaction studies. Based on evidence from patients with T2DM, the levels of cardio-metabolic markers may benefit from appropriate diet and exercise due to improvement of glycemic control and insulin sensitivity33. Previous studies showed that the most common barriers in diabetes management are related to the lack of adherence to nutrition recommendations and exercise33,34,35,36,37,38,39. Moreover, several epidemiological studies reported a significant relationship between dietary intake and lipid profile or inflammation36,37,40. The dietary composition also has a major effect on the acid-base balance41,42. Dietary acid load (DAL), as a nutritional indicator is estimated according to the potential renal acid load (PRAL) and net endogenous acid production (NEAP) and reflects the acid-forming potential of a diet. Following a Western dietary pattern, containing a high amount of animal protein, processed meat, excessive intake of artificially sweetened beverages, and low consumption of fruit and vegetables can initiate inflammation by promoting metabolic acidosis condition43. Several studies have suggested a linkage between metabolic risk factors and DAL. They found higher levels of LDL-C and TC among the highest DAL categories44,45. It is also suggested that a pro-inflammatory status can be followed by a high DAL43,46,47. To our knowledge, only a few studies investigated the interaction effect of ApoB ins/del genetic variant (rs11279109) and diet on metabolic markers. According to a study among Iranian patients with diabetes, high levels of TG and LDL-C has been reported in del-allele carriers who had a higher intake of monounsaturated fatty acid (MUFA) and carbohydrate, and there was an elevated level of serum leptin among del-allele carriers with high protein, polyunsaturated fatty acid (PUFA), MUFA, and saturated fatty acid (SFA) intake7,48. Another study on healthy individuals revealed an increased level of TC among del-allele carriers with higher PUFA and SFA intake7,30. Since the majority of studies concerned the role of DAL or ApoB in T2DM and its complications independently, and due to the insufficient data regarding the possible interactions between ApoB genetic variants and DAL on anthropometric and metabolic markers, we intended to investigate the interaction between ApoB ins/del genetic variant (rs11279109) and DAL including PRAL/NEAP on anthropometric measurements, and metabolic markers among patients with T2DM.
Materials and methods
Study design and subjects
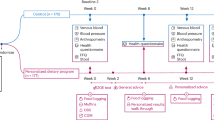
This study is part of a larger cross-sectional investigation on patients with T2DM from the Iranian Diabetes centers conducted in Tehran49. 3100 patients with type 2 diabetes mellitus attended Iranian Diabetes centers, where 1100 individuals were ultimately excluded due to exclusion criteria and 2000 patients remained for the genetic assessment. Accordingly, 700 patients (276 men, 424 women) were included based on both genotyping and inclusion criteria using a simple random sampling method (Fig. 1). More details on the required sample size calculation are provided in the “Statistical analysis” section. Participants aged 35–65 with fasting blood glucose ≥ 126 mg/dl, consuming drugs for diabetes treatment, or both were included after taking written informed consent by explaining the purpose of the study. Patients aged under 35 and over 65 years, having any other chronic conditions, insulin use, pregnancy, lactation, and alcohol consumption 24 h before collecting blood samples were excluded. This study was approved by the Ethics Committee of Tehran University of Medical Sciences with a protocol number of IR.TUMS.VCR.REC.1395.15060 and was conducted based on the Declaration of Helsinki.
Assessment of anthropometric measures and physical activity
The estimation of weight and height was based on minimal clothing by Seca falcon scales, with an accuracy of 100 gr and 0.5 cm, respectively. The measurement of a midpoint between the lower edge of the chest and the upper edge of the iliac crest and the last rib formed waist circumference (WC). The division of weight (kg) by the square of height (m2) determined body mass index (BMI). Subjects were considered underweight, normal, and overweight/obese if their BMI values were < 18⋅5, 18.5–24.9. and 25 ≤ kg/m2, respectively50. Metabolic equivalent task (MET h/day) was used for physical activity calculation8 via a validated physical activity questionnaire51.
Dietary intake and dietary acid load assessment
The regular dietary intake of participants was evaluated throughout the last year for 147 food items by a trained dietitian via interviews and using a semi-quantitative food frequency questionnaire (FFQ) which was validated by Mirmiran et al.52. The amounts listed for each food were converted to grams per day using household measures53. Two common scores including PRAL and NEAP, were used and categorized into tertiles for evaluation of DAL. The NEAP score includes total protein and potassium intake as the major acid-producing foods and the PRAL estimates the intestinal absorption rates for protein, potassium, calcium, magnesium, and phosphate. We used both scores for the DAL assessment.
The NEAP and PRAL scores were assessed by the following previously established algorithms54,55:
Biochemical assessment and genotyping
Whole blood samples were obtained after 12-h fasting. The enzymatic method was utilized for measuring serum TC and TG (using kits from Pars Azmoon, Iran). Turbidimetry on a Roche Hitachi analyzer (Roche, Germany) was carried out for HDL-C and LDL-C levels measurement. Also, the evaluation of leptin level was performed by the ELISA method (Bioassay Technology Co, China, and Mediagnost, Germany, respectively). Interleukin-18 (IL-18) was assessed by means of the ELISA method (Shanghai Crystal Day Biotech Co., Ltd). The sensitivity of the IL-18 ELISA kit was 28 ng/ml. Whole blood was used for isolating genomic DNA through the salting-out extraction method56. Polymerase chain reaction (PCR) was used to detect the ApoB ins/del (rs11279109) in a 25 µL mixture, containing 50 ng of DNA, 0.2 mM of each primer (Forward: 5’-CAGCTGGCGATGGACCCGCCGA-3’ Reverse: 5’-ACCGGCCCTGGCGCCCGCCAGCA-3’), and 75 ng genomic DNA. PCR was done at 94 °C for 50 s and 64 °C for 90 s in 35 cycles. The genotypes were ascertained by 8% polyacrylamide gel electrophoresis7.
Statistical analysis
The sample size was computed through the following formula by considering the type I error of α = 0.05 and type II error of β = 80%:
The frequency of the del allele is unclear in the Iranian population. Provided that the frequency of the minor allele was 1–16% in a different population57, here, 1% was chosen as the frequency of the del allele, hence, the minimum sample size for the present study was 500 subjects (5 / 0.01 = 500). Regarding the necessity of a large sample size for determining the gene-diet interaction, the final sample size was increased to 700 by reason of the indefinite distribution of the genetic variant and further increase of statistical power. The normality was checked by the Kolmogorov-Smirnov test. We analyzed variables among three genotype groups (ins/ins, ins/del, and del/del) and tertiles of indexes using Analysis of variance (ANOVA). The interaction effect of ApoB ins/del (rs11279109) and PRAL/NEAP scores on BMI, WC, TG, TC, LDL-C, HDL-C, LDL/HDL, IL-18, and leptin were tested using the generalized linear models (GLMs) test in three multivariate interaction models: (crude) model, P1 = P value with adjustments for potential confounding factors including age, sex, smoking, alcohol, lipid-lowering medications, and P2 = P value with adjustments for variables in model 1 plus for job, education, family history of diabetes, glucose-lowering medications, physical activity, and energy intake. The data were analyzed by IBM SPSS software version 25 and P-value < 0.05 was considered statistically significant.
Results
Association of DAL with metabolic markers
700 patients with T2DM (Fig. 1) were studied in this cross-sectional study. Genotype distribution of ApoB ins/del (rs11279109) in the type 2 diabetes population was 67.1%, 29.4%, and 3.4% for ins/ins, ins/del, and del/del genotypes (rs11279109), respectively. 67.8%, 29.4%, and 2.7% of participants in ins/ins, ins/del, and del/del genotype groups were men, respectively. The genotype frequency had no deviation from Hardy–Weinberg equilibrium (P = 0.97). Details of the baseline characteristic and biochemical variables between ApoB ins/del (rs11279109) genotypes are presented in Table 1. There were no significant differences according to ApoB ins/del genotypes, however, legume intake for patients with ins/ins and ins/del genotype was 19.33 gr/d. Also, those with del/del genotype consumed 26.12 gr/d which was more than other groups (Supp. 1).
Table 2 shows the general, biochemical, and anthropometric parameters of study participants in the tertiles of DAL. In particular, subjects in the highest tertile of NEAP and PRAL presented higher consumption of energy (PNEAP <0.001, PPRAL <0.001), protein (PNEAP <0.001, PPRAL <0.001), total fat (PNEAP = 0.005, PPRAL = 0.03) and carbohydrate (PNEAP <0.001, PPRAL = 0.01)). For dietary intake differences, a significant result was observed between tertile 1 and 3. In addition, participants with higher PRAL had lower HDL (PPRAL = 0.04) and physical activity (PPRAL = 0.03). Finally, men with the higher DAL (PRAL and NEAP) had higher estimated energy requirements (EER) (PNEAP=0 0.03, PPRAL = 0.03). In terms of HDL, we did not observe a significant difference in a post-hoc analysis, but in EER for men, a significant result was observed between tertiles 2 and 3 of both NEAP and PRAL.
The interaction between ins/del (rs11279109) and DAL (NEAP and PRAL) on metabolic factors
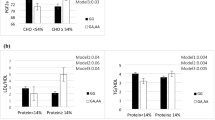
Figures 2 and 3 show the interaction between ins/del (rs11279109) and DAL (PRAL and NEAP) on several biochemical markers. Only in the PRAL method, the interactions of ins/del genetic variant (rs11279109) and DAL on the serum level of TC (Pinteraction = 0.04, P1 = 0,04, P2 = 0.04), LDL-C (Pinteraction = 0.004, P1 = 0,006, P2 = 0.003), LDL/HDL (Pinteraction = 0.02, P1 = 0,02, P2 = 0.03) and IL-18 (Pinteraction = 0.04, P1 = 0,04, P2 = 0.03) were statistically significant in both crude and adjusted models. Del allele carriers who were placed in the higher tertile of PRAL showed lower TC, LDL, LDL/HDL, and IL-18.
Furthermore, in the PRAL method, the interaction of gene and DAL on TG concentration was significant in crude (P = 0.04) and adjusted model 1 (P1 = 0.04), but this interaction was not significant in model 2 (P2 = 0.06). Finally, a significant interaction was found between PRAL and ins/del (rs11279109) on serum levels of leptin just in the crude (P1 = 0.04) model, but in adjusted models (model 1, P1 = 0.23 and model 2, P2 = 0.28), this significant interaction was lost. In particular, patients with the del/del genotype (rs11279109) with high PRAL have lower TG, TC, LDL-C, LDL/HDL ratio, IL-18, and leptin.
Besides, del homozygotes with higher NEAP, had lower WC (P = 0.04), LDL/HDL(P = 0.04), and TG (P = 0.04) in the crude model but in adjusted models, this significant interaction was lost (P > 0.05). No gene-diet interaction was found between DAL (NEAP and PRAL) and ins/del (rs11279109) in associations with other metabolic markers.
Interaction effect between PRAL and ApoB ins/del (rs11279109) on LDL/HDL, LDL, TC, TG, Leptin, and IL-18. P* = P value with the unadjusted (crude) model, P1 = P value with adjustments for potential confounding factors including (age, sex, smoking, alcohol, lipid-lowering medications) P2 = P value with adjustments for variables in model 1 plus for (Job, education, family history of diabetes, glucose-lowering medications, physical activity, and energy intake).
Interaction effect between NEAP and ApoB ins/del (rs11279109) on WC, LDL/HDL, and TG. P* = P value with the unadjusted (crude) model, P1 = P value with adjustments for potential confounding factors including (age, sex, smoking, alcohol, lipid-lowering medications) P2 = P value with adjustments for variables in model 1 plus for (Job, education, family history of diabetes, glucose-lowering medications, physical activity, and energy intake).
Discussion
In the present study, the interaction effect of ApoB ins/del genetic variant (rs11279109) with DAL index on inflammation, leptin, lipid profile, and obesity was evaluated in type 2 patients with diabetes. Here, participants with greater adherence to DAL had significantly higher energy intake as well as consuming more protein, carbohydrate, total fat, and dietary cholesterol along with increased WC and decreased levels of HDL-C. Regarding the ApoB ins/del genetic variant (rs11279109), it may modulate the effect of DAL on cardio-metabolic biomarkers. In particular, the serum levels of TG, TC, LDL-C, IL-18, and LDL/HDL increased unexpectedly in del/del homozygotes (rs11279109) by less adherence to DAL. Considering the role of ApoB ins/del (rs11279109) in the modification of lipid profile and BMI, it is proposed as one of the main potential etiology for dyslipidemia and obesity7,58,59,60. By considering high levels of serum lipid profiles in del/del homozygotes (rs11279109), they are more vulnerable to dyslipidemia and CVDs, however, TG, LDL-C, and TC levels raised by the observation of Vimaleswaran et al. with regard to ins/ins homozygotes after dietary fat intake in healthy adults61. This controversy might be explained by the health status of subjects who participated in different studies.
It is suggested that the production and assemblage of VLDL are impaired in del allele carriers which leads to the storage of fat in hepatocytes and suppressing LDL-receptor secretion, resulting in promoting the synthesis of hepatic TG and ApoB − 100 as well as lowering their clearance62,63. This condition provokes more TG uptake by adipocytes which has a direct relationship with their hypertrophy and hyperplasia64,65. Additionally, leptin production could be affected by the downregulation of ApoB synthesis which causes leptin resistance and obesity at the end66, however, weight and BMI stability in the present study might be the rational explanation for the insignificant interaction between DAL, genotypes, and leptin given that the consumption of a low-calorie diet and the subsequent weight loss was proposed as one of the main mechanisms for decreasing leptin in patients with diabetes67. The significance of the interaction between the genetic variant and DAL on WC was also lost in the second step of confounders adjustment in the present study. The possible direct relationship between energy intake and WC has been reported previously68,69,70 which controlling the effect of energy intake in the aforementioned interaction might be a clarification for such insignificance.
According to previous studies, the Western Dietary Pattern, which consists of high amounts of acid-forming foods like refined grains, animal products, and dietary fats, was associated with DAL and might contribute to the acid-base imbalance71,72 which is in line with findings of the present study. Besides, further odds of obesity were obtained over the median and quartiles of NEAP73,74,75. A marginally significant association was also suggested between DAL and WC by Murakami et al.76.
A number of studies with inconsistent findings have considered the interaction effect of ApoB ins/del genetic variant (rs11279109) with dietary status on cardio-metabolic biomarkers. Del allele carriers had a higher level of TG and LDL-C together with lower BMI and WC by consuming more MUFA, carbohydrate, and n-3 PUFA, respectively7,49. An insignificant interaction was obtained between the ApoB ins/del genetic variant (rs11279109) and dietary inflammatory indexes (DII) on obesity by Mokhtary et al.77, however, a significant change was proposed on LDL-C by considering the interaction of ApoB ins/del genetic variant (rs11279109) and DII in which the LDL level was elevated in del carriers by increasing the DII score78.
In the current study, del/del homozygotes (rs11279109) consumed more protein foods significantly than other genotypes with an emphasis on legumes as a major part of protein consumption. Various studies proposed a protective role of legumes as opposed to the high levels of pro-inflammatory markers and serum lipid profile in accordance with ours. As indicated by Becerra et al. addition to others, a legume-rich diet tended to improve fasting serum TC, LDL-C, and TG79,80,81,82,83,84,85,86,87,88, whereas a non-significant association was observed with serum lipid profile through inconsistent evidence89. Furthermore, an inverse linkage of legumes consumption and inflammatory markers e.g. hs-CRP, IL-6, and IL-18 were observed via multiple studies80,82,90,91,92,93.
The precise underlying mechanisms of the aforementioned relationships are not clarified so far, yet, some attainable justifications might be available for the interaction between DAL and ApoB ins/del genetic variant (rs11279109) on the above-mentioned markers. On the one hand, a legume-rich diet might ameliorate serum lipid profile by means of high fiber content through fat absorption reduction and diminishes hepatic synthesis of cholesterol while binding to bile acid which assists in modifying lipid circulation94,95. On the other hand, decreased glycemic index and glycemic load of legumes, boost insulin sensitivity which hinders the mobilization of free fatty acids from adipose tissue in order to preserve low levels of LDL-C and TC84. Moreover, the level of blood cholesterol could be dropped by some specific phytochemicals like phytosterols which hydrolyzation of saponin to diosgenin might affect cholesterol absorption96. It was proposed that dietary fiber might reduce inflammation not only by downregulating lipid oxidation but also by normal bowel flora which could play a positive role in a healthy intestinal environment82,97,98. The Magnesium content of legumes may also contribute to inflammation suppression. Hypomagnesemia leads to inflammatory responses by several pathways which could contribute to the pathogenesis of various conditions such as diabetes, cardiovascular diseases, osteoporosis, and neurodegenerative diseases99,100. It could exacerbate inflammation by the activation of cellular oxidative stress, renin-angiotensin system, nuclear factor-κB signaling, phagocytic cells, and the transcription of cytokines and pro-inflammatory genes101,102,103,104,105,106,107,108,109,110,111,112,113,114. Accordingly, it seems that healthy eating habits and lifestyles might ameliorate the vulnerability of del/del homozygotes (rs11279109) for dyslipidemia. Even so, more investigations are required to shed light on the interaction of ApoB ins/del genetic variant (rs11279109) with DAL indexes on cardio-metabolic markers.
This investigation was the first attempt to study the interaction effect of ApoB ins/del genetic variant (rs11279109) with DAL indexes on cardio-metabolic markers among type 2 patients with diabetes, still, it had some limitations. First and foremost, this cross-sectional study cannot verify any causality about the observed interactions, and the serum level of ApoB was not measured here. Furthermore, the bias in the study such as recall bias and over-or under-reporting of participants couldn’t be neglected since we used the FFQ for dietary intake evaluation.
Conclusion
In conclusion, carriers of the del/del genotype of the rs11279109 variant in the ApoB may have higher serum concentrations of LDL/HDL, TG, TC, LDL-C, and IL-18 due to lower adherence to DAL, possibly associated with reduced legume consumption. However, high-quality intervention studies are needed to confirm this relationship. This perspective opens new avenues for understanding gene-diet interactions and underscores the importance of considering genetic variability when addressing metabolic health in patients with type 2 diabetes.
Data availability
Data described in the manuscript, code book, and analytic code will be made available upon request pending to Masoumeh Rafiee (masomeh.rafiei@gmail.com.)
Abbreviations
- Ins/del :
-
Insertion/deletion
- DAL:
-
Dietary acid load
- T2DM:
-
Type 2 diabetes mellitus
- PRAL:
-
Potential renal acid load
- NEAP:
-
Net-endogenous acid production
- LDL-C:
-
Low density lipoprotein cholesterol
- TC:
-
Total cholesterol
- TG:
-
Triglyceride
- IL-18:
-
Interleukin-18
- WC:
-
Waist circumference
- CVDs:
-
Cardiovascular diseases
- GWAS:
-
Genomewide association studies
- VLDL:
-
Very low-density lipoprotein
- CHD:
-
Coronary heart disease
- MUFA:
-
Monounsaturated fatty acid
- PUFA:
-
Polyunsaturated fatty acid
- SFA:
-
Saturated fatty acid
- FFQ:
-
Food frequency questionnaire
- SOD:
-
Superoxide dismutase
- TAC:
-
Total antioxidant capacity
- PTX3:
-
Pentraxin-3
- PGF2α:
-
8-Isoprostane F2α
- hs-CRP:
-
High-sensitivity C-reactive protein
- PCR:
-
Polymerase chain reaction
- ANCOVA:
-
Analysis of covariance
- DII:
-
Dietary inflammatory indexes
- TLC:
-
Therapeutic lifestyle change
References
Hameed, I. et al. Type 2 diabetes mellitus: From a metabolic disorder to an inflammatory condition. World J. Diabetes 6(4), 598 (2015).
Wright, A. K. et al. Life expectancy and cause-specific mortality in type 2 diabetes: A population-based cohort study quantifying relationships in ethnic subgroups. Diabetes Care 40(3), 338–345 (2017).
Naeini, Z. et al. Effects of DHA-enriched fish oil on gene expression levels of p53 and NF-κB and PPAR-γ activity in PBMCs of patients with T2DM: A randomized, double-blind, clinical trial. Nutr. Metab. Cardiovasc. Dis. 30(3), 441–447 (2020).
Leon, B. M. & Maddox, T. M. Diabetes and cardiovascular disease: Epidemiology, biological mechanisms, treatment recommendations and future research. World J. Diabetes 6(13), 1246 (2015).
Narindrarangkura, P., Bosl, W., Rangsin, R. & Hatthachote, P. Prevalence of dyslipidemia associated with complications in diabetic patients: A nationwide study in Thailand. Lipids Health Dis. 18(1), 1–8 (2019).
Howard, B. V. Insulin resistance and lipid metabolism. Am. J. Cardiol. 84(1), 28–32 (1999).
Rafiee, M. et al. The interaction between apolipoprotein b insertion/deletion polymorphism and macronutrient intake on lipid profile and serum leptin and ghrelin levels in type 2 diabetes mellitus patients. Eur. J. Nutr. 58(3), 1055–1065 (2019).
Ainsworth, B. E. et al. Compendium of physical activities: classification of energy costs of human physical activities. Med. Sci. Sports Exerc. 25(1), 71–80 (1993).
Devaraj, S., Dasu, M. R. & Jialal, I. Diabetes is a proinflammatory state: A translational perspective. Expert. Rev. Endocrinol. Metab. 5(1), 19–28 (2010).
Olokoba, A. B., Obateru, O. A. & Olokoba, L. B. Type 2 diabetes mellitus: A review of current trends. Oman Med. J. 27(4), 269 (2012).
Chasman, D. I. et al. Genetic loci associated with plasma concentration of low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, triglycerides, apolipoprotein A1, and Apolipoprotein B among 6382 white women in genome-wide analysis with replication. Circulation: Cardiovasc. Genet. 1(1), 21–30 (2008).
Johansen, C. T., Kathiresan, S. & Hegele, R. A. Genetic determinants of plasma triglycerides. J. Lipid Res. 52(2), 189–206 (2011).
Al-Bustan, S. A., Alnaqeeb, M. A., Annice, B. G., Ebrahim, G. A. & Refai, T. M. Genetic association of APOB polymorphisms with variation in serum lipid profile among the Kuwait population. Lipids Health Dis 13, 157. https://doi.org/10.1186/1476-511x-13-157 (2014).
Onat, A. et al. Serum apolipoprotein B predicts dyslipidemia, metabolic syndrome and in women, hypertension and diabetes, independent of markers of central obesity and inflammation. Int. J. Obes. (Lond.) 31(7), 1119–1125. https://doi.org/10.1038/sj.ijo.0803552 (2007).
Faraj, M. et al. Apolipoprotein B: A predictor of inflammatory status in postmenopausal overweight and obese women. Diabetologia 49(7), 1637–1646. https://doi.org/10.1007/s00125-006-0259-7 (2006).
Huang, L. S., de Graaf, J. & Breslow, J. L. ApoB gene MspI RFLP in exon 26 changes amino acid 3611 from Arg to Gln. J. Lipid. Res. 29(1), 63–67 (1988).
Niu, C. et al. Associations of the APOB rs693 and rs17240441 polymorphisms with plasma APOB and lipid levels: A meta-analysis. Lip. Health Dis. 16(1), 1–20 (2017).
Siri, P. et al. Post-transcriptional Stimulation of the Assembly and Secretion of Triglyceride-rich Apolipoprotein B Lipoproteins in a Mouse with selective deficiency of brown adipose tissue, obesity, and insulin resistance*. J. Biol. Chem. 276(49), 46064–46072. https://doi.org/10.1074/jbc.M108909200 (2001).
Visvikis, S., Chan, L., Siest, G., Drouin, P. & Boerwinkle, E. An insertion deletion polymorphism in the signal peptide of the human apolipoprotein B gene. Hum. Genet. 84(4), 373–375. https://doi.org/10.1007/BF00196239 (1990).
Jenkins, D. J. et al. Effect of legumes as part of a low glycemic index diet on glycemic control and cardiovascular risk factors in type 2 diabetes mellitus: A randomized controlled trial. Arch. Intern. Med. 172(21), 1653–1660. https://doi.org/10.1001/2013.jamainternmed.70 (2012).
Pajukanta, P. E. et al. The effects of the apolipoprotein B signal peptide (ins/del) and XbaI polymorphisms on plasma lipid responses to dietary change. Atherosclerosis 122(1), 1–10 (1996).
Rafiee, M. et al. Association between Insertion/Deletion Polymorphism of ApoB gene with dyslipidemia and obesity risk in patients with type 2 diabetes deletion polymorphism of ApoB gene with dyslipidemia and obesity risk in patients with type 2 diabetes. J. Obes. Overweig. 2(2), 204. https://doi.org/10.15744/2455-7633.2.301 (2016).
Sturley, S. L. et al. Human apolipoprotein B signal sequence variants confer a secretion-defective phenotype when expressed in yeast. J. Biol. Chem. 269(34), 21670–21675 (1994).
Heng, C., Saha, N. & Low, P. Evolution of the apolipoprotein B gene and coronary artery disease: A study in low and high risk Asians. Ann. Hum. Genet. 63(1), 45–62 (1999).
Horvath, A., Chorbov, V., Zaharova, B. & Ganev, V. Five polymorphisms of the apolipoprotein B gene in healthy Bulgarians. Hum. Biol. 75, 69–80 (2003).
Ruixing, Y. et al. Effect of the 3’APOB-VNTR polymorphism on the lipid profiles in the Guangxi Hei Yi Zhuang and Han populations. BMC Med. Genet. 8(1), 1–13 (2007).
Kallel, A., Jemaa, R., Feki, M., El Asmi, M., Souissi, M., Sanhaji, H., Haj-Taieb, S., Omar, S., Kaabachi, N. XbaI polymorphism of apolipoprotein B gene in a Tunisian population: alleles frequencies and relationship with plasma lipid parameters. In Annales de biologie clinique 265–70 (2007).
Tsunoda, K., Harihara, S., Tanabe, Y. & Dashnyam, B. Polymorphism of the apolipoprotein B gene and association with plasma lipid and lipoprotein levels in the Mongolian Buryat. Biochem. Genet. 50(3), 249–268 (2012).
Vimaleswaran, K. S. et al. The APOB insertion/deletion polymorphism (rs17240441) influences postprandial lipaemia in healthy adults. Nutr. Metab. (Lond.) 12, 7. https://doi.org/10.1186/s12986-015-0002-9 (2015).
Zhang, J.-Z. et al. Association between apolipoprotein B gene polymorphisms and the risk of coronary heart disease (CHD): An update meta-analysis. J. Renin-Angiotensin-Aldosterone Syst. 16(4), 827–837 (2015).
Cavalli, S. A. et al. Apolipoprotein B gene polymorphisms: Prevalence and impact on serum lipid concentrations in hypercholesterolemic individuals from Brazil. Clin. Chim. Acta 302(1–2), 189–203 (2000).
Glisic, S., Prljic, J., Radovanovic, N. & Alavantic, D. Study of apoB gene signal peptide insertion/deletion polymorphism in a healthy Serbian population: No association with serum lipid levels. Clin. Chim. Acta 263(1), 57–66 (1997).
Nelson, K. M., Reiber, G. & Boyko, E. J. Diet and exercise among adults with type 2 diabetes: Findings from the third national health and nutrition examination survey (NHANES III). Diabet. Care 25(10), 1722–1728 (2002).
Glasgow, R. E., Hampson, S. E., Strycker, L. A. & Ruggiero, L. Personal-model beliefs and social-environmental barriers related to diabetes self-management. Diabet. Care 20(4), 556–561 (1997).
Harris, M. I. Diabetes in America: Epidemiology and scope of the problem. Diabet. Care 21(Supplement 3), C11–C14 (1998).
Toeller, M. et al. Nutritional intake of 2868 IDDM patients from 30 centres in Europe. Diabetologia 39(8), 929–939 (1996).
Eeley, E. et al. UKPDS 18: Estimated dietary intake in type 2 diabetic patients randomly allocated to diet, sulphonylurea or insulin therapy. Diabet. Med. 13(7), 656–662 (1996).
Campbell, L. V., Barth, R. & Cosper, J. Unsatisfactory nutritional parameters in non-insulin-dependent diabetes mellitus. Med. J. Aust. 151(3), 146–150 (1989).
Virtanen, S. M. et al. Comparison of diets of diabetic and non-diabetic elderly men in Finland, The Netherlands and Italy. Eur. J. Clin. Nutr. 54(3), 181–186 (2000).
Tan, S.-Y., Batterham, M. & Tapsell, L. Increased intake of dietary polyunsaturated fat does not promote whole body or preferential abdominal fat mass loss in overweight adults. Obes. Facts 4(5), 352–357 (2011).
Hietavala, E. et al. Effect of diet composition on acid–base balance in adolescents, young adults and elderly at rest and during exercise. Eur. J. Clin. Nutr. 69(3), 399–404 (2015).
Siener, R. Dietary treatment of metabolic acidosis in chronic kidney disease. Nutrients 10(4), 512 (2018).
Wu, T. et al. Associations between dietary acid load and biomarkers of inflammation and hyperglycemia in breast cancer survivors. Nutrients 11(8), 1913 (2019).
Ostrowska, J., Janiszewska, J. & Szostak-Węgierek, D. Dietary acid load and cardiometabolic risk factors—A narrative review. Nutrients 12(11), 3419 (2020).
Han, E. et al. Association between dietary acid load and the risk of cardiovascular disease: Nationwide surveys (KNHANES 2008–2011). Cardiovasc. Diabetol. 15(1), 1–14 (2016).
Fjell, A. M. et al. One-year brain atrophy evident in healthy aging. J. Neurosci. 29(48), 15223–15231 (2009).
Abbasalizad Farhangi, M., Nikniaz, L. & Nikniaz, Z. Higher dietary acid load potentially increases serum triglyceride and obesity prevalence in adults: An updated systematic review and meta-analysis. PLoS ONE 14(5), e0216547. https://doi.org/10.1371/journal.pone.0216547 (2019).
Taskinen, M.-R. & Borén, J. New insights into the pathophysiology of dyslipidemia in type 2 diabetes. Atherosclerosis 239(2), 483–495 (2015).
Rafiee, M. et al. Dietary ω-3 polyunsaturated fatty acid intake modulates impact of Insertion/Deletion polymorphism of ApoB gene on obesity risk in type 2 diabetic patients. Nutrition 32(10), 1110–1115 (2016).
Identification EPot, Overweight To, Adults Oi, Heart N, Lung, Institute B, Diabetes NIo, Digestive, Diseases K. Clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults: the evidence report: National Institutes of Health, National Heart, Lung, and Blood Institute, 1998.
Moghaddam, M. H. B. et al. The Iranian version of international physical activity questionnaire (IPAQ) in Iran: Content and construct validity, factor structure, internal consistency and stability. World Appl. Sci. J. 18, 1073–1080. https://doi.org/10.5829/idosi.wasj.2012.18.08.754 (2012).
Mirmiran, P., Esfahani, F. & Azizi, F. Relative validity and reliability of the food frequency questionnaire used to assess nutrient intakes: Tehran Lipid and Glucose Study. Iran J. Diabetes Lipid. 9(2), 185–197 (2009).
Ghaffarpour, M., Houshiar-Rad, A. & Kianfar, H. The manual for household measures, cooking yields factors and edible portion of foods. Tehran: Nashre Olume Keshavarzy 7(213), 42–58 (1999).
Frassetto, L. A., Todd, K. M., Morris, R. C. Jr. & Sebastian, A. Estimation of net endogenous noncarbonic acid production in humans from diet potassium and protein contents. Am. J. Clin. Nutr. 68(3), 576–583 (1998).
Remer, T., Dimitriou, T. & Manz, F. Dietary potential renal acid load and renal net acid excretion in healthy, free-living children and adolescents. Am. J. Clin. Nutr. 77(5), 1255–1260 (2003).
Miller, S., Dykes, D. & Polesky, H. A simple salting out procedure for extracting DNA from human nucleated cells. Nucl. Acids Res. 16(3), 1215 (1988).
Süsleyici, B., Öztürk, M., Yilmazer, S., Cağatay, P. & Hatemi, H. Apolipoprotein B gene variants are involved in the determination of blood glucose and lipid levels in patients with non-insulin dependent diabetes mellitus. Cell Biochem. Funct. 24, 261–267. https://doi.org/10.1002/cbf.1218 (2006).
Marshall, H. W. et al. Apolipoprotein polymorphisms fail to define risk of coronary artery disease. Results of a prospective, angiographically controlled study. Circulation 89(2), 567–577. https://doi.org/10.1161/01.cir.89.2.567 (1994).
Gardemann, A. et al. Association of the insertion/deletion gene polymorphism of the apolipoprotein B signal peptide with myocardial infarction. Atherosclerosis 141(1), 167–175. https://doi.org/10.1016/s0021-9150(98)00161-0 (1998).
Lamia, R. et al. Association of four apolipoprotein B polymorphisms with lipid profile and stenosis in Tunisian coronary patients. J. Genet. 91, 75–79. https://doi.org/10.1007/s12041-012-0127-9 (2012).
Vimaleswaran, K. S. et al. The APOB insertion/deletion polymorphism (rs17240441) influences postprandial Lipaemia in healthy adults. Nutr. Metab. 12(1), 7. https://doi.org/10.1186/s12986-015-0002-9 (2015).
Siri, P. et al. Post-transcriptional stimulation of the assembly and secretion of triglyceride-rich apolipoprotein B lipoproteins in a mouse with selective deficiency of brown adipose tissue, obesity, and insulin resistance. J. Boil. Chem. 276(49), 46064–46072. https://doi.org/10.1074/jbc.M108909200 (2001).
Twisk, J. et al. The role of the LDL receptor in apolipoprotein B secretion. J. Clin. Invest. 105(4), 521–532. https://doi.org/10.1172/jci8623 (2000).
Li, A. C. et al. Peroxisome proliferator-activated receptor gamma ligands inhibit development of atherosclerosis in LDL receptor-deficient mice. J. Clin. Invest. 106(4), 523–531. https://doi.org/10.1172/jci10370 (2000).
Mattevi, V. S., Coimbra, C. E. Jr., Santos, R. V., Salzano, F. M. & Hutz, M. H. Association of the low-density lipoprotein receptor gene with obesity in Native American populations. Hum. Genet. 106(5), 546–552. https://doi.org/10.1007/s004390000299 (2000).
Maffei, M. et al. Leptin levels in human and rodent: Measurement of plasma leptin and ob RNA in obese and weight-reduced subjects. Nat. Med. 1(11), 1155–1161. https://doi.org/10.1038/nm1195-1155 (1995).
Halle, M. et al. Concurrent reductions of serum leptin and lipids during weight loss in obese men with type II diabetes. Am. J. Physiol. Endocrinol. Metab. 277(2), E277–E282. https://doi.org/10.1152/ajpendo.1999.277.2.E277 (1999).
Du, H. et al. Dietary energy density in relation to subsequent changes of weight and waist circumference in European men and women. PLoS ONE 4(4), e5339. https://doi.org/10.1371/journal.pone.0005339 (2009).
Mendoza, J. A., Drewnowski, A. & Christakis, D. A. Dietary energy density is associated with obesity and the metabolic syndrome in U.S. adults. Diabetes Care 30(4), 974–979. https://doi.org/10.2337/dc06-2188 (2007).
Murakami, K., Sasaki, S., Takahashi, Y. & Uenishi, K. Dietary energy density is associated with body mass index and waist circumference, but not with other metabolic risk factors, in free-living young Japanese women. Nutrition 23(11), 798–806. https://doi.org/10.1016/j.nut.2007.08.014 (2007).
Jayedi, A. & Shab-Bidar, S. Dietary acid load and risk of type 2 diabetes: A systematic review and dose–response meta-analysis of prospective observational studies. Clin. Nutr. ESPEN 23, 10–18 (2018).
Remer, T. Influence of nutrition on acid-base balance—Metabolic aspects. Eur. J. Nutr. 40(5), 214–220. https://doi.org/10.1007/s394-001-8348-1 (2001).
Arisawa, K. et al. Association of dietary acid load with the prevalence of metabolic syndrome among participants in baseline survey of the Japan multi-institutional collaborative cohort study. Nutrients 12(6), 1605 (2020).
Fatahi, S., Qorbani, M. & Azadbakht, L. Association between dietary acid load with weight status, dietray quality index (DQI), mean adequacy ratio and energy density among women. J. Gorgan Univ. Med. Sci. 21(1), 76–86 (2019).
Mozaffari, H., Namazi, N., Larijani, B., Bellissimo, N. & Azadbakht, L. Association of dietary acid load with cardiovascular risk factors and the prevalence of metabolic syndrome in Iranian women: A cross-sectional study. Nutrition 67–68, 110570. https://doi.org/10.1016/j.nut.2019.110570 (2019).
Murakami, K., Sasaki, S., Takahashi, Y. & Uenishi, K. Association between dietary acid–base load and cardiometabolic risk factors in young Japanese women. Br. J. Nutr. 100(3), 642–651 (2008).
Mokhtary, N. et al. Association between dietary inflammatory indices (DII, EDII) and obesity with consideration of Insertion/Deletion Apo B polymorphism in type 2 diabetic patients. Obes. Med. 19, 100241. https://doi.org/10.1016/j.obmed.2020.100241 (2020).
Mohamadinarab, M., Yekaninejad, M. S., Siassi, F. & Koohdani, F. Association between dietary inflammatory index and lipid profiles with consideration of Apo B Ins/ Del SNP in type 2 diabetic patients. Meta Gene 26, 100811. https://doi.org/10.1016/j.mgene.2020.100811 (2020).
Becerra-Tomás, N., Papandreou, C. & Salas-Salvadó, J. Legume consumption and cardiometabolic health. Adv. Nutr. 10(Supplement_4), S437–S450. https://doi.org/10.1093/advances/nmz003 (2019).
Hosseinpour-Niazi, S., Mirmiran, P., Hedayati, M. & Azizi, F. Substitution of red meat with legumes in the therapeutic lifestyle change diet based on dietary advice improves cardiometabolic risk factors in overweight type 2 diabetes patients: A cross-over randomized clinical trial. Eur J. Clin. Nutr. 69(5), 592–597. https://doi.org/10.1038/ejcn.2014.228 (2015).
Ha, V. et al. Effect of dietary pulse intake on established therapeutic lipid targets for cardiovascular risk reduction: A systematic review and meta-analysis of randomized controlled trials. Can. Med. Assoc. J. 186(8), E252. https://doi.org/10.1503/cmaj.131727 (2014).
Hermsdorff, H. H. M., Zulet, M. Á., Abete, I. & Martínez, J. A. A legume-based hypocaloric diet reduces proinflammatory status and improves metabolic features in overweight/obese subjects. Eur. J. Nutr. 50(1), 61–69. https://doi.org/10.1007/s00394-010-0115-x (2011).
Bazzano, L. A., Thompson, A. M., Tees, M. T., Nguyen, C. H. & Winham, D. M. Non-soy legume consumption lowers cholesterol levels: A meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 21(2), 94–103. https://doi.org/10.1016/j.numecd.2009.08.012 (2011).
Zhang, Z. et al. A high legume low glycemic index diet improves serum lipid profiles in men. Lipids 45(9), 765–775. https://doi.org/10.1007/s11745-010-3463-7 (2010).
Crujeiras, A. B., Parra, D., Abete, I. & Martínez, J. A. A hypocaloric diet enriched in legumes specifically mitigates lipid peroxidation in obese subjects. Free Radic. Res. 41(4), 498–506. https://doi.org/10.1080/10715760601131935 (2007).
Anderson, J. W. & Major, A. W. Pulses and lipaemia, short- and long-term effect: Potential in the prevention of cardiovascular disease. Br. J. Nutr. 88(S3), 263–271. https://doi.org/10.1079/BJN2002716 (2002).
Tovar, J., Nilsson, A., Johansson, M. & Björck, I. Combining functional features of whole-grain barley and legumes for dietary reduction of cardiometabolic risk: A randomised cross-over intervention in mature women. Br. J. Nutr. 111(4), 706–714. https://doi.org/10.1017/S000711451300305X (2014).
Mohammadifard, N., Sarrafzadegan, N., Paknahad, Z. & Nouri, F. Inverse association of legume consumption and dyslipidemia: Isfahan Healthy Heart Program. J. Clin. Lipidol. 8(6), 584–593. https://doi.org/10.1016/j.jacl.2014.08.006 (2014).
Ruiz-Esparza-Cisneros, J., Vasconcelos-Ulloa, J. J., González-Mendoza, D., Beltrán-González, G. & Díaz-Molina, R. Effect of dietary intervention with a legume-based food product on malondialdehyde levels, HOMA index, and lipid profile. Endocrinología, Diabetes y Nutrición (English ed) 67(4), 235–244. https://doi.org/10.1016/j.endien.2019.08.007 (2020).
Esmaillzadeh, A. & Azadbakht, L. Legume consumption is inversely associated with serum concentrations of adhesion molecules and inflammatory biomarkers among Iranian women. J. Nutr. 142(2), 334–339. https://doi.org/10.3945/jn.111.146167 (2011).
Hartman, T. J. et al. Consumption of a legume-enriched, low-glycemic index diet is associated with biomarkers of insulin resistance and inflammation among men at risk for colorectal cancer. J. Nutr. 140(1), 60–67. https://doi.org/10.3945/jn.109.114249 (2009).
Saraf-Bank, S., Esmaillzadeh, A., Faghihimani, E. & Azadbakht, L. Effect of non-soy legume consumption on inflammation and serum adiponectin levels among first-degree relatives of patients with diabetes: A randomized, crossover study. Nutrition 31(3), 459–465. https://doi.org/10.1016/j.nut.2014.09.015 (2015).
Nilsson, A., Johansson, E., Ekström, L. & Björck, I. Effects of a brown beans evening meal on metabolic risk markers and appetite regulating hormones at a subsequent standardized breakfast: A randomized cross-over study. PLoS ONE 8(4), e59985. https://doi.org/10.1371/journal.pone.0059985 (2013).
Trinidad, T. P., Mallillin, A. C., Loyola, A. S., Sagum, R. S. & Encabo, R. R. The potential health benefits of legumes as a good source of dietary fibre. Br. J. Nutr. 103(4), 569–574. https://doi.org/10.1017/S0007114509992157 (2010).
Mallillin, A. C., Trinidad, T. P., Raterta, R., Dagbay, K. & Loyola, A. S. Dietary fibre and fermentability characteristics of root crops and legumes. Br. J. Nutr. 100(3), 485–488. https://doi.org/10.1017/S000711450891151X (2008).
Rochfort, S. & Panozzo, J. Phytochemicals for health, the role of pulses. J. Agric. Food Chem. 55(20), 7981–7994. https://doi.org/10.1021/jf071704w (2007).
King, D. E. et al. Effect of a high-fiber diet vs a fiber-supplemented diet on C-reactive protein level. Arch. Intern. Med. 167(5), 502–506. https://doi.org/10.1001/archinte.167.5.502 (2007).
Ma, Y. et al. Association between dietary fiber and serum C-reactive protein. Am. J. Clin. Nutr. 83(4), 760–766. https://doi.org/10.1093/ajcn/83.4.760 (2006).
Liu, M. & Dudley, S. C. Magnesium, oxidative stress inflammation, and cardiovascular disease. Antioxidants 9(10), 907 (2020).
Maier, J. A., Castiglioni, S., Locatelli, L., Zocchi, M. & Mazur, A. Magnesium and inflammation: Advances and perspectives. Semin. Cell Dev. Biol. 115, 37–44. https://doi.org/10.1016/j.semcdb.2020.11.002 (2021).
Rayssiguier, Y., Libako, P., Nowacki, W. & Rock, E. Magnesium deficiency and metabolic syndrome: Stress and inflammation may reflect calcium activation. Magn. Res. 23(2), 73–80. https://doi.org/10.1684/mrh.2010.0208 (2010).
Sontia, B., Montezano, A. C., Paravicini, T., Tabet, F. & Touyz, R. M. Downregulation of renal TRPM7 and increased inflammation and fibrosis in aldosterone-infused mice: Effects of magnesium. Hypertension 51(4), 915–921. https://doi.org/10.1161/hypertensionaha.107.100339 (2008).
Mak, I. T., Kramer, J. H. & Weglicki, W. B. Suppression of neutrophil and endothelial activation by substance P receptor blockade in the Mg-deficient rat. Magn. Res. 16(2), 91–97 (2003).
Guerrero-Romero, F. & Rodríguez-Morán, M. Hypomagnesemia, oxidative stress, inflammation, and metabolic syndrome. Diabetes/Metab. Res Rev. 22(6), 471–476. https://doi.org/10.1002/dmrr.644 (2006).
Ahokas, R. A., Sun, Y., Bhattacharya, S. K., Gerling, I. C. & Weber, K. T. Aldosteronism and a proinflammatory vascular phenotype: Role of Mg2+, Ca2+, and H2O2 in peripheral blood mononuclear cells. Circulation 111(1), 51–57. https://doi.org/10.1161/01.Cir.0000151516.84238.37 (2005).
Maier, J. A. M., Malpuech-Brugère, C., Zimowska, W., Rayssiguier, Y. & Mazur, A. Low magnesium promotes endothelial cell dysfunction: Implications for atherosclerosis, inflammation and thrombosis. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 1689(1), 13–21. https://doi.org/10.1016/j.bbadis.2004.01.002 (2004).
Ferrè, S. et al. Magnesium deficiency promotes a pro-atherogenic phenotype in cultured human endothelial cells via activation of NFkB. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 1802(11), 952–958. https://doi.org/10.1016/j.bbadis.2010.06.016 (2010).
Bernardini, D., Nasulewic, A., Mazur, A. & Maier, J. A. Magnesium and microvascular endothelial cells: A role in inflammation and angiogenesis. Front. Biosci. J. Virtual Libr. 10, 1177–1182. https://doi.org/10.2741/1610 (2005).
Rochelson, B., Dowling, O., Schwartz, N. & Metz, C. N. Magnesium sulfate suppresses inflammatory responses by human umbilical vein endothelial cells (HuVECs) through the NFkappaB pathway. J. Reprod. Immunol. 73(2), 101–107. https://doi.org/10.1016/j.jri.2006.06.004 (2007).
Weglicki, W. B., Phillips, T. M., Freedman, A. M., Cassidy, M. M. & Dickens, B. F. Magnesium-deficiency elevates circulating levels of inflammatory cytokines and endothelin. Mol. Cell. Biochem. 110(2), 169–173. https://doi.org/10.1007/BF02454195 (1992).
Malpuech-Brugère, C. et al. Exacerbated immune stress response during experimental magnesium deficiency results from abnormal cell calcium homeostasis. Life Sci. 63(20), 1815–1822. https://doi.org/10.1016/S0024-3205(98)00455-X (1998).
Mazur, A. et al. Magnesium and the inflammatory response: Potential physiopathological implications. Arch. Biochem. Biophys. 458(1), 48–56. https://doi.org/10.1016/j.abb.2006.03.031 (2007).
Zierler, S. et al. TRPM7 kinase activity regulates murine mast cell degranulation. J. Physiol. 594(11), 2957–2970. https://doi.org/10.1113/JP271564 (2016).
Libako, P., Nowacki, W., Rock, E., Rayssiguier, Y. & Mazur, A. Phagocyte priming by low magnesium status: Input to the enhanced inflammatory and oxidative stress responses. Magn. Res. 23(1), 1–4. https://doi.org/10.1684/mrh.2009.0201 (2010).
Acknowledgements
We would like to express our gratitude to the research deputy of the school of nutritional sciences and dietetics, and especially, the subjects who participate in this study.
Funding
This work was supported by the Tehran University of Medical Sciences [grant number 15060].
Author information
Authors and Affiliations
Contributions
Z. Esmaeily: Conceptualization, Methodology, Formal analysis, Investigation, Writing - Original Draft; F. Abaj: Conceptualization, Methodology, Formal analysis, Investigation, Writing - Original Draft; Z. Naeini: Conceptualization, Investigation, Writing - Original Draft; E. Alvandi: Performing the molecular experiments; M. Rafiee: Formal analysis, Writing – Editing, Interpretation of Data; F. Koohdnai: Conceptualization, Methodology, Supervision, Project administration.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Tehran University of Medical Sciences with a protocol number of IR.TUMS.VCR.REC.1395.15060. Written consent was taken from participants by explaining the purpose of the study.
Consent for publication
The consent of publication has been obtained from all participants.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Esmaeily, Z., Abaj, F., Naeini, Z. et al. Dietary acid load adopts the effect of ApoB ins/del genetic variant (rs11279109) on obesity trait, cardiovascular markers, lipid profile, and serum leptin level among patients with diabetes: a cross-sectional study. Sci Rep 14, 25650 (2024). https://doi.org/10.1038/s41598-024-75352-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-75352-x