Abstract
To determine the correlation between the severity of chronic kidney disease (CKD) and treatment of diabetic macular edema (DME). The retrospective 2-year cohort study included eyes with DME confirmed using spectral-domain optical coherence tomography in Taipei Veterans General Hospital, Taiwan, between 2010 and 2020. All the eyes were treated with an intravitreal injection of anti-vascular endothelial growth factor (anti-VEGF) during regular follow-up around 2 years. They were categorized into 3 different groups: an estimated glomerular filtration rate ≥ 60 (mL/min per 1.73 m2) (group A), < 60 (group B), and patients undergoing hemodialysis (group C). The main outcome measures were (1) visual improvement, (2) structural improvement, and (3) the injection frequency of anti-VEGF in the different groups. In this study, 167 eyes from 120 patients were enrolled. Compared with groups B and C, the eyes in group A experienced the only significant visual improvement at month 3, month 6, and month 12 (P = 0.0001, 0.0002, 0.0013, respectively). The presence of subretinal fluid and intraretinal cysts was significantly decreased in groups A and B. In the treatment frequency analysis, the number of injections was the highest in group A and lowest in group C during the study period (P = 0.04). The severity of CKD had an impact on the DME treatment. The less severe CKD was, the greater the visual improvement that could be achieved. In addition, relatively poor renal function required a lower anti-VEGF injection frequency. The active prevention of the progression of CKD may play a key role in DME treatment.
Similar content being viewed by others
Introduction
Diabetic macular edema (DME) is one of the most common causes of visual disturbance in diabetic mellitus1. In previous studies, the presumed pathogenesis of DME has been linked to the breakdown of the blood-retinal barrier2,3, which is induced by oxidative stress from a prolonged hyperglycemic status. Vascular endothelial growth factors (VEGF) and inflammatory cytokines are upregulated by tissue hypoxia and overexpressed through maintained hyperglycemia. In recent years, based on this pathogenesis, the mainstream treatment of DME has been an intra-vitreal injection (IVI) of anti-VEGF, steroids, or laser photocoagulation4. However, these treatment modalities are sometimes unable to provide effective functional and anatomical improvements. Therefore, the development of DME may stem from complex interplay of multiple factors.
Abnormal renal profiles and advanced DR usually exist simultaneously, suggesting that they may share similar pathophysiology in a microvasculcar environment5,6,7,8,9,10,11,12. Besides, the severity of chronic kidney disease (CKD) has also increased the attention on the treatment of DME in recent years13,14,15. In theory, the accumulation of extracellular fluid in retinal parenchyma may be eliminated through hemodialysis to some extent16,17,18,19,20. However, the impact of CKD on DME treatment remains undetermined because most studies have either involved small case numbers, a cross-sectional study design, or a follow-up period shorter than 6 months13,21,22. Therefore, the present study contributes to existing literature in 3 ways. First, we conducted a 2-year retrospective cohort study involving the analysis of 167 eyes. Second, we correlated renal function and DME treatment, including the frequency of treatment and visual improvement. Third, we evaluated the change in macular edema by using optical coherence tomography (OCT) images.
Materials and methods
Study design and population
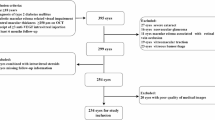
This retrospective cohort study strictly adhered to the tenets of the Declaration of Helsinki and obtained ethics approval from the Institutional Review Board of Taipei Veterans General Hospital (No. 2018-11-004CC). All the eyes enrolled in the study had been diagnosed with DME according to the medical records between 2010 and 2020 from a single medical center, TVGH. DME was defined as central macular thickness of ≥ 300 μm (in the central 1 mm diameter area) and the presence of subretinal fluid (SRF) or intraretinal cysts (IRCs) in central 3 mm diameter area on spectral-domain OCT images (OptoVue, Fremont, CA, USA).
The inclusion criteria were the following: (1) age ≥ 20 years old; (2) DME with the presence of SRF or IRCs and retinal thickening recorded using OCT images; (3) a DR stage evaluated using fundus photography or fluorescein angiography; (4) diabetes mellitus under regular medical treatment; (5) eligible laboratory data related to serum creatinine (Cr), blood urea nitrogen (BUN), and glycated hemoglobin (HbA1C) at baseline and 3 months after initial treatment; (6) a regular follow-up of visual acuity and structural change through an OCT examination every 1–2 months for the cases under treatment, or every 3–6 months for stable cases, during the follow-up period.
The exclusion criteria included the following: (1) eyes with other causes of macular edema such as vitreomacular interface disease or retinal vascular disease; (2) eyes that were not receiving regular follow-up in TVGH or did not receive scheduled anti-VEGF IVI during the study period; (3) eyes that received IVIs of any medication within 6 months before enrollment; (4) eyes that had previously undergone vitrectomy; and (5) pregnancy.
Three consecutive monthly injections of anti-VEGF were applied as a loading dose for treatment-naïve patients with DME. Subsequently, they received pro re nata treatment as required based on the change in visual acuity, level of edema in the OCT images, and patients’ decision. The follow-up interval was decided by retinal specialists in TVGH based on clinical presentation and treatment response.
Data collection
Age, sex, diabetic medication, status of hypertension, DR stage at baseline and month 12, status of panretinal photocoagulation, treatment-naïve status, and IVI medications, including 2 mg/0.05 mL of aflibercept (Eylea; Regeneron Pharmaceuticals, Tarrytown, NY, USA), 0.25 mg/0.05 mL of ranibizumab (Lucentis; Genentech, San Francisco, CA, USA), and 1.25 mg/0.05 mL of bevacizumab (Avastin; Genentech), were recorded.
Serum Cr, BUN, albumin (Alb), urine albumin and creatinine ratio (UACR), and hemoglobin at baseline were recorded. HbA1C at baseline and month 3 were also recorded. CKD was defined according to the Kidney Disease Outcomes Quality Initiative (KDOQI) guidelines. All the eyes were categorized into 3 groups according to the baseline estimated glomerular filtration rate (eGFR): eGFR ≥ 60 mL/min per 1.73 m2 (group A), < 60 mL/min per 1.73 m2 (group B), and patients undergoing hemodialysis (group C)23,24.
Best-corrected visual acuity (BCVA) and OCT images were measured and collected at baseline and at months 3, 6, and 12. BCVA was measured using Snellen eye charts and was converted to the logarithm of the minimum angle of resolution (logMAR) for statistical analysis. Data on the central macular thickness (CMT) and presence of SRF or IRCs were obtained using spectral-domain OCT, RTVue XR Avanti (Optovue, Inc., Fremont, CA).
Statistical analysis
The categorical variables between the different groups were analyzed using a Chi-square test or Fisher’s exact test, with adjustments made for multiple testing using the Bonferroni correction. For continuous variables, analysis of variance was performed for age. A nonparametric Kruskal–Wallis test was used to examine the difference in HbA1C, renal parameters, BCVA, OCT characteristics, and the number of injections at different CKD stages. We evaluated the correlation between the frequency of IVI treatment and renal parameters by using Spearman correlation coefficients. A Wilcoxon signed-rank test was used to compare the BCVA and CMT before and 3, 6, and 12 months after treatment. A McNemar test was performed to compare the SRF and IRCs before and 3, 6, and 12 months after treatment. The SRF and IRC status before and 3, 6, and 12 months after treatment at different stages of CKD and DR were evaluated using a Chi-square test. A P-value < 0.05 was considered statistically significant in all the analyses. We used MedCalc statistical software version 19.6.1 (MedCalc Software, Ostend, Belgium; https://www.medcalc.org; 2020) for all statistical analyses.
The Medical Ethics Committee of Taipei Veterans General Hospital has waived the need of informed consent in this study.
Results
Baseline characteristics
This study enrolled 164 eyes from 120 patients. The mean follow-up time was 21.4 ± 4.4 months. Each eye, even within the same individuals, was treated as an independent subject, with distinct consideration for its unique retinal condition and response to treatment. Of these, 147 eyes (87.4%) were treatment naïve. The baseline characteristics were stratified according to different stages of CKD. Of the 164 eyes, 75 with eGFR ≥ 60 (mL/min/1.73 m2) were stratified as group A (average eGFR = 82.2 mL/min per 1.73 m2), 78 with eGFR < 60 were stratified as group B (average eGFR = 39.4 mL/min per 1.73 m2), and 11 from patients undergoing dialysis were stratified as group C(average eGFR = 16.3 mL/min per 1.73 m2)(Table 1). Of the 120 patients, 63 were male and 57 were female. Of the 164 eyes, 143 eyes were pseudophakic, 21 eyes were phakic without progression of cataracts during the follow-up period. Age, hypertension, serum hemoglobin, serum BUN, HbA1c, and UACR exhibited statistically significant differences (p < 0.05) in different groups, which were compatible with different renal and systemic condition.
The mean BCVA logMAR was 0.53 ± 0.34, and the mean CMT was 391.1 ± 118.0 μm at baseline. SRF was present in 28% of eyes and IRCs in 97.4% of eyes before treatment. The baseline BCVA (logMAR), DR stage, and percentage of treatment-naïve eyes in different groups revealed no significant difference (p = 0.43, 0.462, and 0.22, respectively). In our OCT analysis, the initial presence of SRF and IRCs showed no significant difference between groups.
Correlation between injection frequency regarding renal dysfunction
Table 2 presents the correlations between injection frequency and the renal profiles. Injection frequency was significantly positively correlated with eGFR (r = 0.23, p = 0.015) and baseline HbA1C (r = 0.22, p = 0.0328) within 2 years and negatively correlated with Cr (r = − 0.230, P = 0.0134) in the second year.
In Table 3, the average injection frequency of all eyes was 4.4 ± 2.1 in the first year, 1.70 ± 1.90 in the second year, and 6.17 ± 3.5 during the study period. The injection frequency was the greatest in group A and the lowest in group C in the first and second year and over the 2 years (p = 0.422, 0.014, and 0.039, respectively).
Visual and anatomical improvements after anti-vegf ivis in different groups
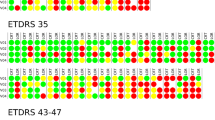
Table 4 and Fig. 1 present the longitudinal changes in the BCVA(logMAR), CMT, and presence of SRF or IRCs after anti-VEGF IVIs. Compared with groups B and C, group A experienced the only significant improvements in visual outcomes at 3, 6, and 12 months. Throughout the OCT analysis, each group showed a significant decrease in CMT at every time point (Table 4). Groups A and B had a significant reduction in SRF and IRCs at each time point. In group C, the SRF exhibited an apparent resolution after 3 consecutive anti-VEGF IVIs (Fig. 1) even without statistical significance (Table 4). These results revealed that the structural improvements were not coordinated with the visual improvements experienced by diabetic patients with DME and severe CKD.
Longitudinal change in best corrected visual acuity (BCVA), central macular thickness (CMT), and macular edema in OCT images. This figure showed the longitudinal changes of BCVA in logMAR (top left), and structural changes in CMT (top right), presence of SRF (lower left), and IRC (lower right) at baseline, 3th month, 6th month, and 12th month.
Visual and anatomical improvements at different DR stages
Table 5 presents the stratified analysis of the change in the BCVA (logMAR) and macular edema by different DR stages. Patients with severe NPDR and PDR experienced statistically significant improvements in the BCVA and a significant reduction in CMT and macular edema at months 3, 6, and 12 after treatment. Nevertheless, no notable difference in BCVA was observed among patients with mild and moderate NPDR at any given time point. With the exception of mild NPDR at the 6th month, a noteworthy reduction in CMT was observed across various stages of DR at each time point.
Discussion
The study reveals three key findings. Firstly, patients with DME and mild CKD demonstrated significant visual and structural improvements during follow-up, comparing to those with severe CKD or undergoing hemodialysis. Second, in Group B, the presence of SRF and IRCs was significantly decreased after treatment but did not coordinate with visual improvements; in Group C, despite fewer injection, vision and the presence of SRF/IRCs did not statistically improve during the follow-up period. This information may suggest that the severity of CKD could be correlated with the pathological cascade of atrophy to some extent, leading to irreversible visual and structural change and ultimately treatment futility. Third, prevention of the progression of CKD may play important role in the treatment of DME.
Microvascular dysfunction secondary to chronic hyperglycemia contributes to the development of DME and CKD. The association between the presence of DR, DME, and the severity of CKD has been widely explored in other studies5,7,8,9,10,25,26,27. However, only few studies have investigated the association between renal function and DME treatment13,15,18,21. To our knowledge, our study is the first long-term cohort study providing a comprehensive analysis of the relationship between CKD and DME treatment.
Intraocular VEGF, which is overexpressed by a chronic hyperglycemic environment and tissue hypoxia, may also result in the breakdown of the blood-retinal barrier. Based on Starling’s rules, the active transcellular transport of plasma proteins by the blood–retinal barrier causing elevated interstitial osmotic pressure is probably the main factor in the formation of DME28. We postulated that the progression of CKD would cause not only an elevation in serum VEGF levels but also collateral damage to the blood–retinal barrier, further interfering in DME treatment through anti-VEGF IVIs29,30,31,32. Studies have reported that increased serum VEGF plays a role in the disruption of the external limiting membrane and ellipsoid zone and macular atrophy33,34. This might explain why the resolution of macular edema was not coordinated with the visual improvements of patients with DME with severe CKD.
An understanding of the complex process involved in leakage from the breakdown of the blood–retinal barrier is growing rapidly, but appropriate models for human DME are lacking. The functional and structural changes in pericytes and astrocytes may either alter the transcellular transport that drives the fluid into the extracellular space or change the response to anti-VEGF and recurrence after treatments17,28. Studies have also noticed that the induction of hemodialysis for patients with DME with severe CKD provides anatomical improvements even without anti-VEGF IVIs. This can explain why severe CKD, especially in patients undergoing hemodialysis, requires fewer anti-VEGF IVIs and has a lower recurrence rate.
Anti-VEGF IVIs have evolved into the standard treatment for DME35,36. In the Diabetic Retinopathy Clinical Research Network Protocol T, the 3 main anti-VEGFs, aflibercept, ranibizumab, and bevacizumab, led to significant visual improvements in center-involved DME37. In the protocol T study, these 3 agents were considered to provide similar visual improvements and the frequency of injections was similar within the 2-year study period. Among eyes with a poorer baseline BCVA, aflibercept was superior in terms of 2-year visual outcomes. Therefore, we did not further analyze the treatment response to different anti-VEGFs because no significant difference in baseline BCVA was detected among the groups.
HbA1c is considered to be not only a predictor of vascular complications in patients with diabetes38 but also a prognostic factor in DME treatment13,21. However, HbA1c can be easily affected by anemia, uremic environment, or erythropoietin stimulating agents39. Lower HbA1c levels may indicate effective glycemic control, poor nutritional status, anemia, or even poor glycemic control with severe uremic status. Therefore, HbA1c should not be solely relied upon as a predictive factor for DME treatment outcome in patients with CKD. In our study, both hemoglobin and HbA1c levels decreased as the severity of CKD increased, as illustrated in Table 1. This elucidates the discrepancies found in other studies, where low HbA1c were purported to predict the presence of residual SRF and a more reduction in CMT following anti-VEGF IVIs13,21.
Proteinuria has been suggested to be a key marker of kidney damage and progressing factor of CKD40,41. In our univariate regression model to evaluate the correlation between the UACR and DME treatment, the additional results revealed that the logMAR changes and frequency of IVIs were statistically nonsignificant (P = 0.62 and 0.55, respectively), indicating that the UACR plays less of a role in predicting DME treatment outcomes.
The stages of CKD were defined by the KDOQI guidelines according to the eGFR and evidence of kidney damage42. CKD stage 3 (eGFR < 60 mL/min per 1.73 m2) is a key clinical boundary because the rate of hospitalization, mortality, and cardiovascular events drastically increase at this stage23,24. Therefore, we used an eGFR of 60 as demarcation point to group study participants. In our stratified analysis, patients with DME with eGFR\({ \geqq }\)60 were prone to visual improvements; for patients with DME with eGFR < 60, even if anatomical outcomes could be achieved with fewer IVIs, obtaining significant visual improvements was difficult. This suggests that prevention of the progression of CKD may have a role in the treatment of DME.
Strengths and limitations
The strength of this study is that it was a 2-year cohort study that included the analysis of 167 eyes, compared with other studies with fewer than 100 eyes and a shorter follow-up period13,21. Second, many studies have used fundus photography as a diagnostic tool for DME5,27. The diagnosis of DME in the present study was verified through OCT, which is more accurate and sensitive than fundus photography. Third, we analyzed not only the visual outcomes but also the anatomical response to anti-VEGF IVIs for DME in the follow-up period. To our knowledge, this is the first long-term study to address the difference in visual and structural outcomes between different groups of patients with CKD.
Our study also has limitations. The distribution of factors and missing data in each group was not balanced, which is why we did not use a multiple regression model for adjusting confounding factors. In the present study, we used a stratification analysis that allowed us to obtain a simple comparison by dividing the study population into different groups to offset the limitations. Furthermore, the selection of both eyes from 44 patients might cause statistical bias. Fortunately, these participants were distributed equally across the groups, which reduced the potential for confounding. We also performed a GEE analysis on different groups of CKD and the treatment responses, which are consistent with our original results (see supplement). In clinical practice, the treatment response in different eyes within the same patient may present inconsistently. Therefore, it is reasonable to regard the two eyes as individual subjects in this study. Third, the duration of diabetes, medications for diabetes, other comorbidities, and the severity of DR may confound the results in this study. However, we analyzed the distribution of DR stages across the groups, and the Chi-square test revealed no statistical differences (P = 0.46, Table 1). Fourth, we only analyzed the clinical parameters at baseline (eGFR, UACR, and HbA1c), which might fluctuate during the follow-up period. We did obtain long-term data during the follow-up, revealing that the biochemical data were stable over time. Thus, the application of baseline data in this study was acceptable for clinical convenience. Fifth, although most of our participants had diabetic nephropathy, different etiologies of CKD might differ in the relationship between CKD and DME treatment. Sixth, during the follow-up period, the undertreatment may occur, potentially affecting visual and structural outcomes. Seventh, for the treatment of DME, laser photocoagulation and injection of steroid were not discussed in this study. In the future, we plan to analyze the complexity of the treatment and evaluate the correlations among different regimens. Finally, the generalizability of this study should be carefully considered because all the participants were Chinese and based in Taiwan.
Conclusion
The severity of CKD affects the treatment of DME. The less severe CKD is, the greater the visual improvements are. In addition, the poorer renal function is, the fewer anti-VEGF injections are required over 2 years. These results indicate that the active prevention of the progression of CKD may play a key role in the treatment of DME.
Data availability
The datasets generated and analysed during the current study are not publicly available due the restriction of institutional review board of Taipei Veterans General Hospital, but are available from the corresponding author on reasonable request.
References
Flaxman, S. R. et al. Global causes of blindness and distance vision impairment 1990–2020: A systematic review and meta-analysis. Lancet Glob. Health. 5(12), e1221–e1234 (2017).
Capitão, M. & Soares, R. Angiogenesis and inflammation crosstalk in diabetic retinopathy. J. Cell Biochem. 117(11), 2443–2453 (2016).
Yamagishi, S. et al. Role of advanced glycation end products (AGEs) and oxidative stress in diabetic retinopathy. Curr. Pharm. Des. 14(10), 962–968 (2008).
Das, A., McGuire, P. G. & Rangasamy, S. Diabetic macular edema: Pathophysiology and novel therapeutic targets. Ophthalmology. 122(7), 1375–1394 (2015).
Hsieh, Y. T. et al. Association of abnormal renal profiles and proliferative diabetic retinopathy and diabetic macular edema in an asian population with type 2 diabetes. JAMA Ophthalmol. 136(1), 68–74 (2018).
Chen, H. et al. A microalbuminuria threshold to predict the risk for the development of diabetic retinopathy in type 2 diabetes mellitus patients. PLoS One. 7(5), e36718 (2012).
He, B. B. et al. Factors associated with diabetic retinopathy in chinese patients with type 2 diabetes mellitus. Int. J. Endocrinol. 2012, 157940 (2012).
Stratton, I. M. et al. UKPDS 50: Risk factors for incidence and progression of retinopathy in Type II diabetes over 6 years from diagnosis. Diabetologia. 44(2), 156–163 (2001).
Wu, J. et al. The relationship between estimated glomerular filtration rate and diabetic retinopathy. J. Ophthalmol. 2015, 326209 (2015).
Rodríguez-Poncelas, A. et al. Chronic kidney disease and diabetic retinopathy in patients with type 2 diabetes. PLoS One. 11(2), e0149448 (2016).
Yamamoto, M. et al. Overt proteinuria, moderately reduced eGFR and their combination are predictive of severe diabetic retinopathy or diabetic macular edema in diabetes. Invest. Ophthalmol. Vis. Sci. 60(7), 2685–2689 (2019).
Romero-Aroca, P., et al., Glomerular filtration rate and/or ratio of urine albumin to creatinine as markers for diabetic retinopathy: a ten-year follow-up study. 2018, 5637130 (2018).
Tsai, M. J. et al. Systemic associations with residual subretinal fluid after Ranibizumab in diabetic macular edema. J. Ophthalmol. 2017, 4834201 (2017).
Wykoff, C. C. et al. Predictors of diabetic macular edema treatment frequency with ranibizumab during the open-label extension of the RIDE and RISE trials. Ophthalmology. 123(8), 1716–1721 (2016).
Lai, I. P. et al. Renal biomarkers for treatment effect of ranibizumab for diabetic macular edema. J. Diabetes Res. 2020, 7239570 (2020).
Tokuyama, T., Ikeda, T. & Sato, K. Effects of haemodialysis on diabetic macular leakage. Br. J. Ophthalmol. 84(12), 1397–1400 (2000).
Tsai, M.-J., Cheng, C.-K. & Wang, Y.-C. Association of body fluid expansion with optical coherence tomography measurements in diabetic retinopathy and diabetic macular edema. Investig. Ophthalmol. Visual Sci. 60(10), 3606–3612 (2019).
Takamura, Y. et al. Functional and anatomical changes in diabetic macular edema after hemodialysis initiation: One-year follow-up multicenter study. Sci. Rep. 10(1), 7788 (2020).
Theodossiadis, P. G. et al. Hemodialysis-induced alterations in macular thickness measured by optical coherence tomography in diabetic patients with end-stage renal disease. Ophthalmologica. 227(2), 90–94 (2012).
Ong, S. S., Thomas, A. S. & Fekrat, S. Improvement of recalcitrant diabetic macular edema after peritoneal dialysis. Ophthalmic Surg. Lasers Imaging Retina. 48(10), 834–837 (2017).
Wong, W. M. et al. Systemic factors associated with treatment response in diabetic macular edema. J. Ophthalmol. 2020, 1875860–1875860 (2020).
Hwang, H. et al. Systemic factors and early treatment response to intravitreal injection for diabetic macular edema: The role of renal function. Retina. 41(6), 1275–1282 (2021).
Go, A. S. et al. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N. Engl. J. Med. 351(13), 1296–1305 (2004).
Jankowski, J. et al. Cardiovascular disease in chronic kidney disease: Pathophysiological insights and therapeutic options. Circulation. 143(11), 1157–1172 (2021).
Park, Y. H. et al. The association between chronic kidney disease and diabetic retinopathy: The Korea National Health and Nutrition Examination Survey 2008–2010. PLoS One. 10(4), e0125338 (2015).
Wong, T. Y. et al. Prevalence and risk factors for diabetic retinopathy: The Singapore Malay eye study. Ophthalmology. 115(11), 1869–1875 (2008).
Man, R. E. et al. The association of estimated glomerular filtration rate with diabetic retinopathy and macular edema. Invest. Ophthalmol. Vis. Sci. 56(8), 4810–4816 (2015).
Klaassen, I., Van Noorden, C. J. & Schlingemann, R. O. Molecular basis of the inner blood-retinal barrier and its breakdown in diabetic macular edema and other pathological conditions. Prog. Retin. Eye Res. 34, 19–48 (2013).
Schrijvers, B. F., Flyvbjerg, A. & De Vriese, A. S. The role of vascular endothelial growth factor (VEGF) in renal pathophysiology. Kidney Int. 65(6), 2003–2017 (2004).
Maharaj, A. S. et al. Vascular endothelial growth factor localization in the adult. Am. J. Pathol. 168(2), 639–648 (2006).
Cha, D. R. et al. Vascular endothelial growth factor is increased during early stage of diabetic nephropathy in type II diabetic rats. J. Endocrinol. 183(1), 183–194 (2004).
Pawlak, K., Mysliwiec, M. & Pawlak, D. Oxidative stress, phosphate and creatinine levels are independently associated with vascular endothelial growth factor levels in patients with chronic renal failure. Cytokine. 43(1), 98–101 (2008).
Karst, S. G. et al. Atrophy of the central neuroretina in patients treated for diabetic macular edema. Acta ophthalmol. 97(8), e1054–e1061 (2019).
Jain, A. et al. Status of serum VEGF and ICAM-1 and its association with external limiting membrane and inner segment-outer segment junction disruption in type 2 diabetes mellitus. Mol. Vis. 19, 1760–1768 (2013).
Nguyen, Q. D. et al. Ranibizumab for diabetic macular edema: Results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology. 119(4), 789–801 (2012).
Elman, M. J. et al. Randomized trial evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 117(6), 1064-1077.e35 (2010).
Wells, J. A. et al. Aflibercept, Bevacizumab, or Ranibizumab for diabetic macular edema: Two-year results from a comparative effectiveness randomized clinical trial. Ophthalmology. 123(6), 1351–1359 (2016).
The relationship of glycemic exposure (HbA1c) to the risk of development and progression of retinopathy in the diabetes control and complications trial. Diabetes. 44(8), 968–83 (1995).
Speeckaert, M. et al. Are there better alternatives than haemoglobin A1c to estimate glycaemic control in the chronic kidney disease population?. Nephrol. Dial. Transplant. 29(12), 2167–2177 (2014).
Tsai, W. C. et al. Risk factors for development and progression of chronic kidney disease: A systematic review and exploratory meta-analysis. Medicine (Baltimore). 95(11), e3013 (2016).
Hunsicker, L. G. et al. Predictors of the progression of renal disease in the modification of diet in renal disease study. Kidney Int. 51(6), 1908–1919 (1997).
K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am. J. Kidney Dis. 39(2 Suppl 1), S1-266 (2002).
Acknowledgements
This retrospective cohort study strictly adhered to the tenets of the Declaration of Helsinki and obtained ethics approval from the Institutional Review Board of Taipei Veterans General Hospital (2018-11-004CC). The Medical Ethics Committee of Taipei Veterans General Hospital has waived the need of informed consent in this study.
Author information
Authors and Affiliations
Contributions
YBC wrote the main manuscript and analyzed the data. JYC prepared the figure and tables. YJC and CP reviewed the manuscript. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chou, YB., Chang, JY., Chou, YJ. et al. Association between renal function and the Treatment of Diabetic Macular Edema in Long-Term Cohort Study. Sci Rep 14, 26098 (2024). https://doi.org/10.1038/s41598-024-77530-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-77530-3