Abstract
Prenatal exposure to heavy metals such as Lead (Pb), arsenic (As), cadmium (Cd), and selenium (Se) is associated with various adverse pregnancy outcomes. This study examines the association between early pregnancy maternal blood levels of these metals and adverse pregnancy outcomes, while also addressing the differences between low-risk and high-risk groups based on having a history of preterm birth (PTB). This prospective cohort study recruited parous women during first trimester, categorized into low-risk and high-risk groups. Participants completed a questionnaire, and heavy metal levels were measured in blood samples. Pregnancy outcomes including PTB, low birth weight (LBW), gestational age at delivery, birth weight and head circumference were recorded following delivery. Multivariable analyses were conducted to evaluate the independent associations between heavy metal levels and pregnancy outcomes, while adjusting for maternal age, BMI, employment, smoking, fertility treatments and education. Among 404 participants, the mean (± SD) levels were Pb: 3.12 ± 1.82 µg/L, As: 0.41 ± 0.4 µg/L, Cd: 0.26 ± 0.34 µg/L, and Se: 119.84 ± 21.05 µg/L. Significant differences in Pb, Se, Cd and As levels were observed between the low-risk and high-risk groups, with higher levels in the low-risk group. However, no significant associations were found between heavy metal levels and any of the study outcomes in either univariable comparison or multivariable models. These findings highlight the need for further research to understand the potential impact of these metals on pregnancy, considering population-specific factors and exposure timing.
Similar content being viewed by others
Introduction
Human exposure to heavy metals, including Arsenic (As), Cadmium (Cd), Selenium (Se) and Lead (Pb) is widespread, despite adverse effects of heavy metals on health that have been conclusively proved and reviewed1. These metals can enter the human body in various ways such as food, contaminated water, air and soil2.
While exposure to certain levels of heavy metals may offer some benefits, excessive exposure can harm the human body in multiple ways3. Heavy metals tend to accumulate in organs such as liver, heart, kidneys, bones and brain4,5. Once accumulated, they are used as substitutes for essential elements, causing disruption to metabolic mechanisms and antioxidant imbalance, affecting hormones and causing change in carbohydrate, proteins, and lipid metabolism, leaving the body susceptible to health complications4. Additionally, as free ions, these metals can trigger the formation of reactive oxygen species, damage to DNA and impair DNA repair mechanisms6. Consequently, metals can be carcinogenic, and have been associated with increased risk for breast, prostate, skin, lung, and stomach cancers4.
The reproductive system is also affected by heavy metals exposure. Studies have shown that cadmium (Cd) can mimic estrogen’s effects in the uterus and mammary glands7. By competing with estradiol, cadmium can activate estrogen receptors, altering regulatory pathways and gene expression. This can lead to an increase in uterine wet weight and early onset of puberty in females7.
Prenatal exposure to heavy metals has been associated with adverse pregnancy course and outcomes. Since fetuses differ from adults in various biological aspects, they may be affected by even small amounts of heavy metals, potentially at levels lower than those established by international guidelines8.
Both low and high levels of Pb in maternal blood measured in the third trimester have been linked to increased risk of pregnancy hypertension9, and high level of Pb in cord blood has been associated with increased risk for spontaneous abortion, miscarriage, intrauterine fetal death and preterm delivery (PTB)10,11. Exposure to As during the entire pregnancy has been associated with PTB, stillbirths and spontaneous abortions12. High Cd and Pb in maternal blood measured close to delivery have been associated with low birthweight (LBW), birth length and head circumference8.
In contrast, some heavy metals have a positive impact on pregnancy course and outcomes. Through its incorporation into numerous selenoproteins, Se is involved in anti-oxidation, anti-inflammatory and anti-apoptotic functions13. Lower serum levels of Se at 12 gestational weeks were found among women who experienced PTB14. In one study women were categorized as being at high or low risk for PTB, based on whether or not they had a history of PTB. Among the High-risk population, Se levels below the median (< 83.3 ppm) were 1.5 times more likely to be found among women with recurrent PTB, although this difference was not statistically significant (RR 1.5, 95% CI 0.9–2.7)15. Additionally, Se level below median at 20 gestational weeks was 2.7 times more likely to be found among women with recurrent PTB (RR 2.7, 95% CI 1.5–4.8; p = 0.01(15. A study comparing Se concentrations in women with and without severe pre-eclampsia found that Se levels were significantly lower in maternal blood and cord blood of the pre-eclampsia group16. Despite its role in antioxidant defense and cellular protection, its specific impact on pregnancy outcomes, such as preterm birth (PTB) and low birth weight (LBW), remains uncertain14,16,17.
Early pregnancy, particularly the first trimester, is a critical window of fetal development, involving organogenesis and rapid cell differentiation18. Exposure to toxic metals during this phase can disrupt these developmental processes and lead to adverse outcomes such as PTB, LBW which affect offspring long-term health8. Recommended levels for these metals in blood are typically based on guidelines form the Centers for Disease Control and Prevention (CDC) and the World Health Organization (WHO), which suggests that blood Pb levels should be below 50 µg/L for pregnant women to minimize health risks19,20. According to The American College of Obstetricians and Gynaecologists (ACOG), Pb levels below 50 µg/L in maternal blood are considered low risk, while levels exceeding 100 µg/L may require intervention21. While there are no universally established blood levels for As or Cd, specifically for pregnancy, it is generally advised to minimize exposure due to their known toxicities. According to the National Institutes of Health (NIH), the normal range for Se serum level is typically between 70 and 150 µg/L22. During pregnancy, maintaining Se levels within the normal range is critical, with a recommended dietary allowance of 60–70 µg/day22.
Numerous studies have shown an association between various pregnancy outcomes and heavy metal levels11,23. While some studies have specifically examined heavy metal exposure during the first trimester of pregnancy, the majority of research has focused on metal levels measured in the third trimester or with sample collection at different gestational weeks without a consistent approach or hypothesis regarding a certain exposure window11,18,24. This emphasis on later stages of pregnancy leaves a gap in understanding how exposure during the most critical period of fetal development impacts pregnancy outcomes18. In Israel, limited data exist on the blood levels of heavy metals among pregnant women, particularly during the early stages of pregnancy, despite ongoing monitoring of water and environmental quality25. By focusing in the first trimester, we seek to determine whether early exposure to these metals is associated with increased risks of adverse outcomes.
Furthermore, a history of pregnancy complications is a key risk factor for recurrence in subsequent pregnancies. Persistent environmental factors are believed to contribute to this recurrence11,26. This assumption is one of the reasons for examining the impact of early pregnancy metal exposure in our study. While the association between metal exposure and PTB has been extensively studied, little is known regarding the association between metal exposure and recurrent PTB. The aim of the current study is to measure the association between early pregnancy levels of Pb, Cd, As, Se and pregnancy outcomes, including PTB, recurrent PTB, newborn head circumference, gestational week of delivery, LBW and birth weight, in a cohort of parous pregnant Jewish women residing in the Negev region of Israel.
Materials and methods
Study design and setting
This study is a prospective cohort investigation involving parous pregnant Jewish women residing in the Negev region of Israel, designed to evaluate the potential interaction between pre-existing risk factors, specifically a history of PTB, and maternal blood levels of heavy metals. Participants were divided into two distinct groups: women lacking a history of PTB, denoted as the low-risk group, and those with a history of PTB, denoted as the high-risk group. By comparing metal levels between low-risk and high-risk groups, we aim to determine if women with a history of PTB are more susceptible to adverse effects from metal exposure.
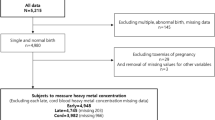
Based on primary assessment of metal levels, the required sample size was a total of 378 participants. The inclusion criteria were: pregnant Jewish women who were at up to 13.5 gestational weeks, with a singleton pregnancy, with or without a history of a spontaneous singleton PTB. Due to the significant genetic variation across ethnicities, the study included only Jewish women. Solely singleton pregnancies were included in the study (current, and the previous pregnancy if ended with PTB), as twin pregnancies are associated with a significantly higher risk of PTB, which could confound the results. Exclusion criteria included known fetal genetic or congenital anomalies in either the current or previous pregnancy.
Recruitment for the low-risk group took place at the nuchal translucency clinic at Soroka University Medical Center (SUMC), where pregnant women typically undergo nuchal translucency ultrasound screening between 11 and 13.5 gestational weeks. These screenings are standard practice for all pregnant women and are covered by national health insurance legislation through health maintenance organizations. High-risk participants were recruited from both this clinic as well as other clinics across the Negev region. A comparison between participants recruited from SUMC and other clinics showed no significant differences in demographic characteristics, pregnancy outcomes, or heavy metal levels, except for a minor difference in mean Pb levels (pv = 0.04). Each participant completed a comprehensive questionnaire and provided a blood sample for the measurement of the following heavy metal levels: Pb, Cd, As and Se.
Metal concentration measurement
Blood metal concentrations (Pb, Cd, As and Se) were measured using Inductively Coupled Plasma Mass Spectrometry (ICP-MS). After each clinic visit, serum samples were frozen at – 80 °C and stored. At the end of each month, these samples were transferred to the National Public Health Laboratory in Abu Kabir for analysis. Metal analysis was done using an Agilent 7800x ICP-MS instrument equipped with an Integrated Sample Introducing System (ISIS) and High Matrix Introducing mode (HMI). The procedure involved matrix-match dilution and direct injection to the ICP-MS instrument, followed by helium dilution in the HMI27. The Method followed standard quality assurance and quality control procedures. Blood metals were quantified using internal standard calibration procedure and certified analytical standards. Quality control was performed by analysing aliquots of control materials (G-EQUAS) in each series (each ten samples) and accuracy was validated by the annual successful participation in international proficiency test (G-EQUAS) for all parameters. Limits of Quantification (LOQ) derived from methods validation were 0.1 µg/L for all metals.
Variables definitions
The definitions of several variables used in the analysis are as follows: PTB was defined as a birth occurring before 37 weeks of gestation, while LBW was defined as a birth weight below 2500 g. Parity, defined as the number of previous births, was considered for all participants. All women included in this study had at least one prior birth. The presence of private insurance served as an indicator of high socioeconomic status. Body mass index (BMI) was categorized as follows: underweight: <18.5, normal: 18.5–24.99, overweight: 25–29.99, and obese: >29.99.
Statistical analysis
Initially, the frequencies of all variables were assessed and described using mean ± SD for continuous variables with a normal distribution, median and range for continuous variables not normally distributed, or frequencies (n, %) for categorical variables. Continuous variables, such as maternal age and metal levels, were examined for normal distribution. The normal distribution of heavy metal levels was tested using measures such as mean, mode, standard deviation, skewness, and kurtosis. The levels of Cd and As displayed a positively skewed distribution, and therefore a square root function was applied to achieve their normal distribution.
Univariable analyses, including T-tests, ANOVA, Mann-Whitney tests, and Spearman or Pearson correlation coefficients was conducted to evaluate the association between background characteristics, dependent variables (pregnancy outcomes), and independent variables (heavy metal levels). Heavy metal levels were compared between women with and without a history of PTB using t-tests.
Findings with p-values < 0.1 were considered in multivariable analyses, to assess the independent association between heavy metal levels and the studied outcomes. This included linear models for gestational age and birth weight, and logistic models for PTB, recurrent PTB and LBW. The models adjusted for variables associated with the metals levels based on the univariable analyses.
The levels of metals were also categorized into high vs. low, with high levels defined as those above the 75th percentile or median, and the incidence of the studied outcomes compared between women with high and low metals levels.
Results
A total of 404 women were initially enrolled in the study, comprising 194 classified as low-risk and 210 as high-risk. Among these, 14 were excluded due to abortions or pregnancy terminations (7 from each group). An additional 12 women from the low- risk group were excluded since they delivered outside of SUMC, resulting in a final sample size of 378 women (203 high-risk and 175 low-risk). Furthermore, technical issues with metal assays resulted in exclusion of: 42 samples for Pb, 14 samples for Se, 28 samples for Cd and 17 samples for As.
Table 1 presents the mean ± SD levels of the four metals by background and pregnancy characteristics. Significant differences in Pb, Se, Cd and As levels were observed between the low-risk and high-risk groups (PTB history), with higher levels among the low-risk group (Pb: 3.58 ± 2.02 µg/L vs. 2.7 ± 1.49 µg/L, pv < 0.001, Se: 122.7 ± 22.25 µg/L vs. 117 ± 19.43 µg/L, pv = 0.006, Cd: 0.5 ± 0.26 µg/L vs. 0.36 ± 0.26 µg/L, pv < 0.001, As: 0.62 ± 0.31 µg/L vs. 0.51 ± 0.32, pv = 0.001). Notably, variations in Cd levels were observed across different BMI groups (pv < 0.001), although not in a linear fashion. Elevated levels of Pb and Cd were evident among smoking mothers compared to non-smoking counterparts (Pb: 3.74 ± 1.85 µg/L vs. 2.08 ± 1.82 µg/L, pv = 0.038; Cd: 0.78 ± 0.33 µg/L vs. 0.39 ± 0.22 µg/L, pv < 0.001). Additionally, possessing private insurance, indicative of higher socioeconomic status, was linked to lower Pb levels (3.1 ± 1.8 µg/L vs. 4.06 ± 2.35 µg/L, pv = 0.035). Moreover, elevated levels of As were noted among employed mothers (0.57 ± 0.33 µg/L vs. 0.48 ± 0.26 µg/L, pv = 0.042). Variations in Se levels were noted by parity (pv = 0.007); however, these differences did not follow a linear trend. No significant associations were observed between other maternal characteristics and metal levels.
Characteristics of the study population by low and high risk for PTB are presented in Table 2. Participants were 21–49 years old, with a mean age of 32.7 ± 4.56 years. Among them, 361 (89.6%) were non-smokers while 42 (10.4%) reported smoking. Additionally, 69 women (17.1%) were categorized as overweight and 135 (33.5%) were classified as obese. Most pregnancies, (377, 93.3%), occurred spontaneously, while 27 (6.7%) were the result of fertility treatments.
Serum levels of Pb and Se exhibited a normal distribution, with mean ± SD values of 3.12 ± 1.82 µg/L (minimum: 0; maximum: 16.257) and 119.84 ± 21.05 µg/L (minimum: 54.67; maximum: 241.39), respectively. The mean ± SD serum levels were 0.26 ± 0.34 µg/L (minimum: 0; maximum: 3.03) for Cd and 0.41 ± 0.4 µg/L (minimum: 0; maximum: 2.4) for As. Notably, As, Cd and Pb levels were below the limit of detection in 64 (15.3%), 67 (16%) and 5 (1.2%) women, respectively.
A history of PTB was associated with decreased levels of Pb, Cd, and As (pv<0.001 for all) as well as Se (pv=0.006). Graphs 1-4 depict the mean metal levels by gestational week at recruitment, stratified into low-risk and high-risk groups. Notably, the levels of Pb were consistently higher in the low-risk group across all gestational weeks, with mean±SD of 3.6±2.04 μg/L for the low-risk group and 2.88±1.88 μg/L for the high-risk group (pv=0.008). Conversely, for Se, As and Cd, no specific pattern or consistent difference between the groups was observed.

Significant differences in heavy metal levels were observed between the low-risk and high-risk groups. For example, 24.1% of women with a history of PTB had Pb levels in the highest tertile compared to 43.9% of women without a history of PTB (pv < 0.001). Similarly, 28.6% of women with a history of PTB had As levels in the highest tertile compared to 39.4% without such a history (pv = 0.001), and 27.1% of women with a history of PTB had Cd levels in the highest tertile compared to 40.4% without a history (pv < 0.001). However, no significant associations were found between the metal level tertiles and PTB. For instance, 17.4% of women in the lowest Pb tertile delivered preterm, compared to 13.2% in the middle tertile and 10.2% in the highest tertile (pv = 0.36, linear by linear association pv = 0.16). Due to the small sample size, this was not tested separately among the high and low risk groups.
Table 3 presents the mean ± SD levels of the four metals by pregnancy outcomes, comparing the levels of the studied metals between women with and without selected pregnancy complications. It is evident that maternal levels of the metals were not associated with PTB or other pregnancy complications.
Based on the Pearson correlation analysis (presented in Table 4), the levels of the studied metals were not correlated with either head circumference, gestational week, or birthweight. No strong correlation was observed between the levels of the studied metals, with the strongest correlation noted between As and Se (r = 0.357, pv < 0.001, results not presented).
Table 5 presents the results of multivariable analyses for the association between selected metals levels and pregnancy outcomes. There was no significant association between sqrt_Cd, Pb levels and gestational week at delivery (Beta = 0.38, and 0.1; pv = 0.73, 0.48, respectively). Similar results were found when analyses were performed separately for the low-risk and high-risk groups. No significant association was found between sqrt_As levels and birth weight (B = 164.55, pv = 0.13), and this remained consistent when stratified by risk group.
Discussion
In this cohort study, no association was observed between early pregnancy maternal blood levels of Pb, Cd, As and Se, and PTB, LBW, gestational week at delivery, birth weight and head circumference. However, statistically significant differences were observed between the high risk and low risk groups regarding Pb, Se and Cd levels, with decreased levels noted in the high-risk group.
In contrast with our findings, previous studies have found associations between Pb, Cd and As and increased incidence of PTB and other adverse birth outcomes11.
According to the ACOG, Pb levels below 50 µg/L are considered low and do not require special follow-up21. Moreover, previous studies that have shown a relationship between Pb levels and adverse outcomes typically reported higher Pb levels, often exceeding 17 µg/L, compared to the levels observed in our study, which were notably lower, with a mean of 3.12 µg/L and a maximum of 16.257 µg/L. Interestingly, a study specifically examining the association between low Pb levels and PTB in a population with low Pb exposure did not find a statistically significant relationship28. That study measured RBC Pb levels, which are approximately three times higher than whole blood levels (which were measured in the current study), with a mean 12 µg/L and a range of range 0–50 µg/L.
Regarding As, the majority of studies have examined the association between birth outcomes and As levels in drinking and ground water rather than in maternal blood. These studies have predominantly been conducted in developing countries11. Two studies conducted in Bangladesh revealed a significantly increased rate of PTB among women residing in regions with elevated As exposure12,29. Since As undergoes metabolism and excretion through urine, the concentration of As in water does not accurately reflect its concentration in the blood, therefore results of previous studies cannot be compared to our findings11. A study that measured As levels in maternal blood at multiple time points during pregnancy found no correlation between levels of As in maternal blood in the 1st trimester and birth outcomes, and later pregnancy levels only were associated with increased SGA risk. This suggests that As levels during early gestation, as tested in the current study, may not be relevant for SGA and possibly other pregnancy outcomes. It should be noted that the mean As level was 0.85 µg/L, with levels above 6.68 µg/L defined as high levels (75th percentile)26, while in our study As levels were lower, with a mean of 0.41 µg/L and a maximum of 2.4 µg/L.
Several studies have identified associations between maternal blood Cd levels and birth outcomes11. One study conducted in a Chinese population revealed an association between Cd exposure and moderate and late PTB (32 to < 34 weeks and 34 to < 37 weeks, respectively). No significant association was found with early PTBs among subjects who exhibited similar Cd concentrations to those in our study30. Moreover, a large cohort study comprising data from over 14,000 women assessed maternal blood Cd levels during pregnancy and stratified the results into quartiles. Cd levels in the fourth quartile (levels ≥ 0.95 µg/L ) demonstrated an association with increased risk for early PTB (22–34 weeks)17. In our study, Cd mean levels were 0.26 µg/L, with a maximum of 3.03 µg/L, indicating relatively low levels. The relatively low Cd levels observed in our study, as well as their findings regarding early PTB only, may explain the differences between results from previous and the current study.
Several studies have indicated an association between low maternal blood Se levels and adverse birth outcomes. According to the NIH, the normal range for Se serum level is typically between 70 and 150 µg/L22. In our study, the mean Se levels were 119.84 µg/L, with a minimum of 54.67 µg/L. A case-control study conducted in Turkey demonstrated that Se levels among women who experienced PTB were significantly lower compared to women who delivered at term. For women who delivered at term, the mean Se levels were 78.98 µg/L, ranging from 72.36 to 84.14 µg/L, whereas for those who delivered preterm, the mean levels were 65.36 µg/L, ranging from 54.68 to 75.65 µg/L31. These levels were measured at birth. Another study assessing levels at 12 gestational weeks showed a similar association: among the group who delivered preterm, the mean Se levels were 19.87 µg/L, compared to 21.12 µg/L among the group who delivered at term14. Therefore, despite the mean Se levels in our study being slightly below the normal range, they are still relatively high compared to the levels reported in studies that have shown an association between low levels and PTB.
We identified only one study that conducted a stratification similar to ours, dividing women into low and high-risk groups based on having a history of PTB15. This study revealed that women with recurrent PTB were more likely to have Se levels below the median at 20 gestational weeks (RR 2.7, 95% CI 1.5–4.8, pv = 0.001). This highlights the potential importance of stratifying risk groups when assessing the impact of heavy metals on pregnancy outcomes.
The discrepancy between our findings and most previous studies regarding the association between maternal blood Pb, As and Se levels and PTB may be attributed to several factors. First, the range of Pb, As and Cd concentrations observed in our study differs from those in previous research, with notably lower (or higher, regarding Se) levels detected. These differences could be attributed to population-specific factors and occupational exposure, which influence heavy metal exposure and its effects on pregnancy outcomes.
Higher metal levels in women without a history of PTB were found in the current study, which is in contrast with most previous studies. However, a cross-sectional study from Indonesia also reports lower Pb levels in maternal serum among women who had PTB, which is consistent with our study32. Intriguingly, the same study found higher Pb levels in the placenta of women with PTB, aligning with the more commonly reported association of elevated Pb levels and adverse pregnancy outcomes. The discrepancy between maternal blood and placental Pb levels highlights the complexity of Pb’s transfer from maternal circulation to the placenta and its potential impact on fetal development. These findings suggest that Pb’s effects on pregnancy outcomes may be influenced by multiple factors, including timing of exposure and mechanism regulating placental transfer. This discrepancy indicates that Pb’s impact on fetal development and pregnancy outcomes might differ depending on its distribution between maternal blood and the placenta.
The underlying reasons for the observed differences in metal levels between women with and without a history of PTB, and the absence of a significant correlation with PTB in the index pregnancy, remain uncertain. Potential factors could include variations in nutritional intake or heightened awareness and preventive behaviors among women in the high-risk group due to their previous pregnancy complications. Further research is warranted to explore these differences and their potential implications.
This study has several limitations that should be acknowledged. First, the study exclusively focuses on Jewish women, which may restrict the generalizability of the findings to the broader population of the Negev region, particularly the significant Bedouin population. The selection of Jewish women only was to minimize heterogenicity, which will enable addressing genetic factors in future studies in this population. Additionally, the enrollment of participants during the 11-13.5 gestational week precludes the investigation of the association between heavy metal levels and early pregnancy loss. Still, this study was able to address many pregnancy outcomes among pregnancies which survived until at least the end of first trimester. Furthermore, the reliance on a single measurement of heavy metal levels during pregnancy raises concerns about its representativeness of chronic exposure levels for both the mother and the fetus. Nonetheless, this is the common approach in most studies11.
Notwithstanding its limitations, this study boasts several strengths that enhance its scientific value. First, the standardized timing of measurements during first trimester ensures consistency across participants, facilitating accurate comparisons of heavy metal levels. Furthermore, the inclusion of both a low-risk group and a high-risk group in our study design is instrumental in providing a comprehensive understanding of the associations between heavy metal exposure and pregnancy outcomes. Stratifying participants into these risk categories based on their obstetric history enabled exploring potential differences in metal levels and their impacts on pregnancy among women at varied risk profiles.
This study contributes to our understanding of the potential consequences of heavy metal exposure during early pregnancy on the risk for adverse pregnancy outcomes, and specifically exposure to low levels. Future studies are recommended to also include diverse populations and additional pregnancy outcomes, besides the identification of high-risk populations, and implementing targeted interventions. Considering the well-documented adverse effects of elevated heavy metal levels, further exploration of the possible effect on pregnancy outcomes is warranted, although our study did not reveal such associations. Still, based on previous findings, caution should be taken regarding exposure to high levels of certain metals, particularly in the later stages of pregnancy.
Data availability
Data will be made available by contacting the corresponding author and following IRB approval.
References
World Health Organization. Health risks of heavy metals from long-range transboundary air pollution. WHO Regional Office for Europe, Copenhagen, 2007; pages 2–144.
Fu, Z. & Xi, S. The effects of heavy metals on human metabolism. Toxicol. Mech. Methods. 30, 167–176 (2020).
Lane, T. W. & Morel, F. M. M. A biological function for cadmium in marine diatoms. (2000).
Rehman, K., Fatima, F., Waheed, I. & Akash, M. S. H. Prevalence of exposure of heavy metals and their impact on health consequences. J. Cell. Biochem. 119, 157–184 (2018).
Engwa, G. A., Ferdinand, P. U., Nwalo, F. N. & Unachukwu, M. N. Mechanism and Health effects of Heavy Metal toxicity in humans. Poisoning Mod. World - New. Tricks Old Dog? https://doi.org/10.5772/INTECHOPEN.82511 (2019).
Beyersmann, D. & Hartwig, A. Carcinogenic metal compounds: recent insight into molecular and cellular mechanisms. Arch. Toxicol. 82, 493–512 (2008).
Johnson, M. D. et al. Cadmium mimics the in vivo effects of estrogen in the uterus and mammary gland. Nat. Med. 9, 1081–1084 (2003). (2003).
Bank-Nielsen, P. I., Long, M., & Bonefeld-Jørgensen, E. C. Pregnant Inuit women’s exposure to metals and association with fetal growth outcomes: ACCEPT 2010–2015. Int. J. Environ. Res. Public Health 16, 1171 (2019).
Magri, J., Sammut, M. & Savona-Ventura, C. Lead and other metals in gestational hypertension. Int. J. Gynecol. Obstet. 83, 29–36 (2003).
Torres-Sánchez, L. E. et al. Intrauterine lead exposure and preterm birth. Environ. Res. 81, 297–301 (1999).
Khanam, R. et al. Prenatal environmental metal exposure and preterm birth: a scoping review. Int. J. Environ. Res. Public. Heal 2021. 18, 573 (2021).
Ahmad, S. A. et al. Arsenic in drinking water and pregnancy outcomes. Environ. Health Perspect. 109, 629 (2001).
Rayman, M. P. Selenium and human health. Lancet. 379, 1256–1268 (2012).
Rayman, M. P., Wijnen, H., Vader, H., Kooistra, L. & Pop, V. Maternal selenium status during early gestation and risk for preterm birth. C Can. Med. Assoc. J. 183, 549 (2011).
Care, A. G. et al. Maternal selenium levels and whole genome screen in recurrent spontaneous preterm birth population: a nested case control study. Eur. J. Obstet. Gynecol. Reprod. Biol. 265, 203–211 (2021).
Katz, O. et al. Severe pre-eclampsia is associated with abnormal trace elements concentrations in maternal and fetal blood. (2012). https://doi.org/10.3109/14767058.2011.624221 25, 1127–1130.
Tsuji, M. et al. The association between whole blood concentrations of heavy metals in pregnant women and premature births: the Japan Environment and Children’s study (JECS). Environ. Res. 166, 562–569 (2018).
Fisher, M. et al. Association between toxic metals, vitamin D and preterm birth in the maternal–infant research on environmental chemicals study. Paediatr. Perinat. Epidemiol. 37, 447–457 (2023).
WHO. Guidelines for drinking-water quality, 4th edition, incorporating the 1st addendum. (2017).
Vision, D. Guidelines for the identification and management of lead exposure in pregnant and lactating women. (2021).
Lead Screening During Pregnancy and Lactation | ACOG. https://www.acog.org/clinical/clinical-guidance/committee-opinion/articles/2012/08/lead-screening-during-pregnancy-and-lactation
Selenium - Health Professional Fact Sheet. https://ods.od.nih.gov/factsheets/Selenium/healthProfessional/#h6
Milton, A. H. et al. A review of the effects of Chronic Arsenic exposure on adverse pregnancy outcomes. Int. J. Environ. Res. Public. Health 14, (2017).
Wu, T. et al. Effects of exposure to multiple metallic elements in the first trimester of pregnancy on the risk of preterm birth. Matern Child. Nutr. e13682 https://doi.org/10.1111/MCN.13682 (2024).
Contaminants in food and pesticides Ministry of Health. https://www.gov.il/he/pages/contaminants?chapterIndex=5
Wang, H. et al. Accepted manuscript maternal serum arsenic level during pregnancy is positively associated with adverse pregnant outcomes in a Chinese population. Toxicol. Appl. Pharmacol. https://doi.org/10.1016/j.taap.2018.07.030 (2018).
Gajek, R., Barley, F. & She, J. Determination of essential and toxic metals in blood by ICP-MS with calibration in synthetic matrix. Anal. Methods. 5, 2193–2202 (2013).
Perkins, M. et al. Very low maternal lead level in pregnancy and birth outcomes in an eastern Massachusetts population. Ann. Epidemiol. 24, 915–919 (2014).
Rahman, M. L. et al. Prenatal arsenic exposure, child marriage, and pregnancy weight gain: associations with preterm birth in Bangladesh. Environ. Int. 112, 23–32 (2018).
Wang, H. et al. Association of maternal serum cadmium level during pregnancy with risk of preterm birth in a Chinese population. Environ. Pollut. 216, 851–857 (2016).
Yıldırım, E. et al. Is the concentration of cadmium, lead, mercury, and selenium related to preterm birth? Biol. Trace Elem. Res. 191, 306–312 (2019).
Irwinda, R., Wibowo, N., & Putri, A. S. The concentration of micronutrients and heavy metals in maternal serum, placenta, and cord blood: a cross-sectional study in preterm birth. J. Pregnancy 2019, 5062365 (2019).
Acknowledgements
None to declare.
Funding
This study was funded by the Israeli science foundation (grant #1435/20, PI: Tamar Wainstock).
Author information
Authors and Affiliations
Contributions
I.B.G. -Investigation, Writing the main manuscript text, Visualization, Formal analysis.E.S.- Data Curation, Supervision, Review and Editing .M.H.B.- Data curation, Conceptualization.D.B., N.T.D., R.R. and A.H.- : Data curation, Investigation.T.W.- Funding Acquisition, Resources, Writing- Review and Editing, Supervision. All authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics declaration
The study protocol was approved by the institutional review board (Soroka University Medical Center IRB committee, #0390-20-SOR). The study has been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments (Helsinki Declaration 1975, revision 2013). All participants gave signed informed consent at recruitment.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Goldberg, I.B., Sheiner, E., Bal, M.H. et al. Early pregnancy metal levels in maternal blood and pregnancy outcomes. Sci Rep 14, 27866 (2024). https://doi.org/10.1038/s41598-024-79107-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-79107-6