Abstract
Evidence regarding the effect of systemic immune-inflammation index on delirium occurrence is limited. This study aimed to investigate the association between SII and delirium in intensive care unit (ICU) patients. Methods: Information was extracted from Medical Information Mart for Intensive Care-IV. Four logistic regression model was established and incorporated with subgroup analysis and restricted cubic spline (RCS). The cutoff value of SII was acquired from receiver operator characteristic curve (ROC), and propensity score matching (PSM) was utilized to attenuate the confounding effect. Survival analysis was utilized to evaluate the relationship between SII and 30-day or 90-day all-cause mortality. Results: Among the 7,518 participants, 1,685 cases of delirium occurred. Individuals in the highest quartile of SII exhibited a heightened delirium risk, with a significant multivariable-adjusted odds ratio (OR) of 3.12(2.24,4.33). Tendency analysis, subgroup analysis and PSM together confirmed the positive relationship. Results of Cox regression displayed the risk of both 30-day and 90-day mortality increased about 50% in the higher-SII group. Conclusion: Higher levels of SII is positively associated with the occurrence of delirium and increased all-cause mortality risk.
Similar content being viewed by others
Introduction
Delirium is an acute neuropsychiatric disturbance syndrome characterized by fluctuating consciousness symptoms, which is commonly seen among patients admitted in ICU1. Many adverse influences of delirium in ICU have been proved. For instance, it can delay the weaning of mechanical ventilation, prolong the stay in ICU, make patients loss of independence and cause difficulties to perform treatment2,3,4,5. Moreover, delirium has been confirmed to related to the morbidity and mortality6.
Thus, delirium in ICU has been paid more attention nowdays7. According to recent research, the incidence was up to 25% in the general ICU patients1,8. Particularly, in the patients with severe illness, its occurrence could be even up to 60% to 80%9. The primary therapies for delirium mainly limited to symptomatic treatment10. Actually, clinical experts’ consensus claims that the best way to deal with delirium is to prevent its pathological development, rather than to treat it after symptoms have emerged11,12,13. Hence, researching the risk factors on delirium is of great significance. Till now, quite a number of alarming factors and indicators for delirium have been verified. Studies have reported that both demographic factors (such as ageing) and underlying chronic diseases (such as chronic obstructive lung diseases ,COPD) can stimulate the development of delirium14,15 besides, specific status like trauma and malnutrition are related to the occurrence of delirium16,17. Even daily lifestyle like smoking also exhibits a positive correlation with post operative delirium18. Except that, infection and inflammation were identified to play critical roles in the pathological process of delirium19,20. Researchers have found that elevated preoperative neutrophil‐to‐lymphocyte ratio (NLR) levels mediate the development of postoperative delirium21. Inflammatory mediator including interleukin-1, interleukin-2, interleukin-8 and C-reactive protein etc. were also demonstrated as predictors to delirium22.
However, as a novel inflammation index derived from the counts of circulating platelets, neutrophils and lymphocytes, systemic immune-inflammation index (SII) has rarely been researched in the delirium of ICU patients, whose inflammation response are usually intensively activated23. In view of this, this study investigated the relationship between SII and the delirium risk in ICU patients. This paper discovered that SII was positively associated with the incidence of delirium in critical patients, and enriched the predicting method to delirium in these patients.
Method
Data source and extraction
Data was extracted from Medical Information Mart for Intensive Care—IV (MIMIC-IV), a free accessible database documenting more than 50,000 ICU admissions from Beth Israel Deaconess Medical Center during 2008 and 201924. Navicat Premium (version 16) was used to extract dataset. Demographic information, vital signs, laboratory findings, and diagnoses defined by International Classification of Diseases and Ninth Revision (ICD-9) and International Classification of Diseases and Tenth Revision (ICD-10) codes were acquired from MIMIC-IV database. The researchers in this study have completed the courses and received the necessary certification to access the dataset (certification number: 10854849). Individual permission was not necessary and waived in this paper since the project has no intervention in clinical treatment, and the patients’ information are anonymized. All procedures in this study were performed in accordance with the Declaration of Helsinki.
In this paper, the demographic information mainly includes age, sex and the dead time; vital signs contain systolic blood pressure (SBP), diastolic blood pressure (DBP), heart rate (HR), respiratory rate (RR), body temperature and oxygenated hemoglobin saturation (SpO2). Scoring systems refers to sequential organ failure assessment (SOFA) and Charlson comorbidity index (CCI). Chronic comorbidities involve diabetes mellitus (DM), chronic kidney disease (CKD) and chronic obstructive pulmonary disease (COPD). Extracted laboratory findings consist of serum glucose, hemoglobin, creatinine, albumin, bicarbonate, calcium, chloride, potassium, prothrombin time (PT), partial thromboplastin time (PTT), alanine aminotransferase (ALT), aspartate aminotransferase (AST) and bilirubin.
Study population
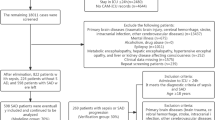
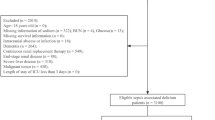
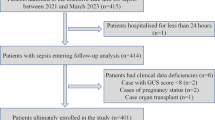
Patients firstly admitted to ICU were included, and the exclusion criteria are listed as follows: (i) patients younger than 18 years; (ii) with mental disorders; (iii) without records of delirium or were diagnosed with delirium within 24 h since ICU admission; (iv) without SII. The details were shown in Fig. 1.
Definition of delirium, SII and outcomes
SII was calculated as the formular: SII = platelet × neutrophil/lymphocyte (× 109/L)23. Quartiles (Qs) of SII was calculated, and the study population was divided according the quartiles: Q1: SII ≤ 596.04; Q2: 596.04 < SII ≤ 1095.87; Q3: 1095.87 < SII ≤ 2269.85; Q4: SII > 2269.85.
In MIMIC-IV database, delirium was assessed using the confusion assessment method for the intensive care unit (CAM-ICU) and diagnosed with the Diagnostic and Statistical Manuals of Mental Disorders (DSM-5)25,26. In this study, the primary outcome was the occurrence of delirium in ICU. The secondary outcome was the 30-day and 90-day all-cause mortality from ICU admission.
Statistical analysis
Continuous variables are expressed as median (interquartile range); categorical variables are exhibited as counts (percentage). The baseline differences among the four groups (Q1-Q4) were assessed by Kruskal–Wallis test, Chi-square test for the comparison of continuous and categorical variables, respectively. The quartiles of SII (Q1, Q2, Q3, Q4) were treated as nominal variable, and was recode as 1, 2, 3, 4. The other variates were listed as follows: delirium (no = 0, yes = 1); age grade (age < 45 years old = 1; 45 ≤ age < 60 years old = 2, ≥ 60 years old = 3); gender (male = 1, female = 2); DM (no = 0, yes = 1); COPD (no = 0, yes = 1); CKD (no = 0, yes = 1). Vital signs and laboratory indexes were taken for continuous variables.
Next, four logistic regression models were established. Model 1: crude model, univariable logistic regression analysis; Model 2: adjusted for basic vital signs including heart rate, respiratory rate, SBP, DBP, temperature, SpO2, SOFA and CCI; Model 3: further adjusted for laboratory findings including serum glucose, hemoglobin, serum creatinine, albumin, bicarbonate, calcium, chloride, potassium, PT, PTT, ALT, AST and bilirubin; Model 4: further adjusted for chronic disease DM, COPD, CKD. Followed, the tendency analysis was applied. In addition, to further verify the association, multiplicative interaction and subgroup analysis was performed in the stratified covariates including SOFA, CCI, COPD, DM and gender. Moreover, as the effect of NLR to delirium in critically ill patients has not been reported, the association between NLR and delirium was also evaluated based on the above models.
To explore non-linear relationship between SII and delirium, RCS (4 knots) based on the full adjusted model 4 was conducted, with analysis of variance testing for nonlinearity. In cases of identified nonlinearity, RCS was further validated in stratified SOFA, CCI, gender, and age covariates. ROC analysis was performed to calculate the most appropriate cutoff values of SII. Then the area under the curve (AUC) based on SII and NLR was compared, and the net reclassification index (NRI) was calculated to evaluate the prediction performance of two different indicators. PSM and Cox proportional hazard regression were applied in the patients grouped by the cutoff value of SII. 1:1 PSM analysis was utilized by the nearest-neighbor method, and the caliper was set as 0.03. The propensity score was calculated using logistic regression analysis. The standardized mean difference less than 0.01 was considered sufficient balance between the two groups27. Univariable and multivariable regressions were adopted again for sensitive analysis. Cox proportional hazard regression analyses (variables presenting p < 0.05 were included in multivariable regression) were used to identify the association between SII and 30-day and 90-day mortality since ICU admission.
Statistical analyses were carried out using Stata 16.1 (Stata Corporation, College Station, TX, United States) and the R 4.3.1 software (R Foundation for Statistical Computing, Vienna, Austria), with a significance level set at p < 0.05 (two-sided).
Results
Baseline characteristics of the study population
7518 patients were encompassed in this study, and 61.9% were male. Patients were divided into four groups according to SII quartiles (Q1 ≤ 596.04; 596.04 < Q2 ≤ 1095.87; 1095.87 < Q3 ≤ 2269.85; Q4 > 2269.85). The baseline characteristics of the study cohort was displayed in Table 1. The delirium incidence was 22.4%, and elevated apparently across group Q1 to Q4. The median age was 67.17 (56.63, 76.23). As for basic vital signs, heart rate, respiratory rate, diastolic blood pressure (DBP), body temperature generally increased in participants in the Q4. Besides, in Q4 group, apparent elevation was observed in laboratory findings including serum glucose, hemoglobin, alanine aminotransferase (ALT) and aspartate aminotransferase (AST); while albumin, prothrombin time (PT) and bicarbonate showed decreasing trend. Additionally, climbing incidence of chronic obstructive pulmonary disease (COPD) and chronic kidney disease (CKD) were found across group Q1 to Q4.
The association between SII and delirium
In total study population, 1685 (22.4%) individuals experienced the development of delirium. The results of 4 logistic regression models were shown in Table 2. Generally, the risk of delirium elevated with rising SII levels in all models. In the crude model (model 1), both p values at Q3 and Q4 showed statistically different, except for Q2. In the full adjusted model (model 4), the ORs of delirium were 1.00(reference), 1.87(1.29,2.72), 2.36(1.67,3.33) and 3.12(2.24,4.33) across Q1 to Q4. To confirm the positive association, sensitive analysis was carried out and all p for trend in 4 models show statistical significance (Table 2). In addition, NLR also positively associated with delirium. The ORs in the full adjusted model were 1.00(reference), 1.30(1.05,1.61), 1.61(1.32,1.97) and 1.87(1.54,2.28) across the quartiles of NLR, and the details were shown in supplementary table.
Results of Subgroup analysis and interaction effect analysis
To explore whether interaction effect affecting the association between delirium and SII, subgroup analyses were conducted in the stratified factors including SOFA, CII, DM, COPD and gender. As shown in Table 3, the risk of delirium still exhibited an increasing trend generally in the stratified groups, while all p values for interaction were bigger than 0.05, indicating that the positive relationship still hold in the stratified patients.
The exploration of the nonlinear association between SII and delirium
RCS regression model was employed to further explore the positive association. Result revealed that a reversed J – shaped association exist between SII and the risk of delirium, both p for overall and p for nonlinear were less than 0.001(Fig. 2 A). In general, the risk of delirium was low if SII less than about 1,100 (× 109/L); the risk kept climbing with SII elevation until reaching around 4,890 (× 109/L); then the ORs started to descend but still maintained higher than 1.00. To further investigate the nonlinear relationship, RCS was performed in the stratified covariates including SOFA, CCI, gender and age (Fig. 2 B-E). Though the curves seemed different in subgroups of diverse factors, the p for interaction were all bigger than 0.05, indicating the non-linear relationship had no apparent difference in these subgroups.
Results of ROC and logistic regression after PSM
In this part, ROC analysis was based on the full adjusted model, and the AUC was 0.886 (Fig. 3). The cutoff point was 1,406.07(× 109/L). Then, the study population was segmented into two groups: SII > = 1406.07 group and SII < 1406.07 group. Based on this cutoff point, PSM was conducted. Figure 4A,B displayed Kernel density plots before and after PSM, showing that the distribution of patients’ propensity scores more concentrated after PSM, and the two groups were well matched. 1,004 pairs of patients were reserved after PSM for further logistic regression. As described in Fig. 4C, the ORs were 1.50 (1.25, 1.81) in the univariable logistic regression and 1.95 (1.24, 3.07) in the multivariable logistic regression after PSM. Moreover, the discriminative performance of different models based on SII or NLR was evaluated by comparing the AUC. Results exhibited that the model based on SII has a slightly better discrimination (AUC = 0.888) than the model based on NLR (AUC = 0.883), and the p value was less than 0.001 (Fig. 5). Besides, the NRI was 0.03 (p = 0.62), which means that the predictive performance of SII and NLR has no significant difference (supplementary figure).
Survival analysis between SII and 30 or 90-day mortality
Table 4 and Table 5 displayed the outcomes of the Cox proportional regression for 30-day and 90-day all-cause mortality. In the unadjusted model, the HRs of 30-day and 90-day mortality were 1.44 (1.23–1.69) and 1.39 (1.22–1.58) in the group with higher SII. The similar outcomes were found after adjusted by multiple factors, with the HRs of 1.35(1.10,1.65) and 1.29(1.08,1.54) respectively, indicating that the mortality apparently elevated in the patients with higher SII. Kaplan–Meier (K-M) survival curve analysis also revealed the statistical difference in the mortalities of the two groups (Fig. 6). Compared to the lower SII group the survival probability of 90 days and 30 days apparently decreased in the high SII group ( p < 0.001).
Discussion
This study reveals that SII is positively associated with delirium in the critical patients. In addition, an inverted J-shaped relationship between SII and delirium was found. Besides, the high level of SII is linked to low 30-day and 90-day survival probability. These findings indicates that SII may be an indicator to delirium and guiding the early intervention.
In critical patients, pathological dysregulation of proinflammatory and anti-inflammatory homeostasis is commonly seen. The exaggerated inflammatory response to noxious stressors (such as infection, trauma, surgery, acute inflammation, etc.) could impair the body28. Besides, the abnormal inflammatory status can facilitate the occurrence of delirium in ICU29. Given this, SII was studied in this research. Results displayed that the risk of delirium increased significantly across Q1 to Q4. In full adjusted model after PSM, the OR of delirium even rise up to 1.95. It should be noted that the 95% CI seems wider before PSM. This may result from the reduction in sample size, as only 1,004 pairs of patients were reserved from the sample (7,518 participants) after PSM. Tendency analysis further confirmed this positive association. This result was consistent with the Yan’s study30, in which SII was suggested as a biomarker for POD in intertrochanteric fracture patients; differently, the best cutoff point of SII in his study was 752.6 × 109/L, much less than the value 1406.07 × 109/L in our paper. This discrepancy could be attributed to the different study population: the inflammatory response in critical patients tend to be more intensive and dysregulated31. In another research in accordance with our findings, SII was suggested as a reliable predictor for postoperative cognitive decline32. In addition, the ROC corroborated the discriminative ability of SII to delirium; this result is parallel to Xiang’s research, in which SII act as an important component in a predictive nomogram model for postoperative delirium33. Logistic regression after PSM reinforced the positive association between SII and delirium. Meanwhile, in this paper, supplementary outcomes demonstrated that NLR was also positively correlated with delirium among ICU patients. While before this, NLR was merely proved to related with delirium under specific circumstances such as perioperative period, stroke and sepsis etc.21,34,35. Though Seo suggested NLR may be a potential biomarker for delirium in ICU by comparing the NLR value between the patient with and without delirium36, but no models were established and the OR of NLR to delirium was not investigated. Despite the predictable performance of SII and NLR was proved no statistical significance in our paper, but the discrimination ability of SII was proved slightly higher than NLR, and this could be important in the large numbers of critically ill patients.
Till now, researches on SII in delirium mainly limited to POD. To our knowledge, this is the first time to report SII affecting the prevalence of delirium in general critical patients. Several possible mechanisms may account for the association.
First, previous work has confirmed inflammation interact with cognitive dysfunction29,37,38,39; As previously described, in the patients with critical illness, inflammation can be induced by various noxious stressors. In this circumstance, production of neutrophils from the bone marrow can be accelerated, while lymphopenia was induced40. This results in an elevated NLR and can partially explain the positive association between NLR and delirium. Similarly, both stressors and inflammatory stimuli can result in rising platelet counts41. All these changes together lead to an elevated SII, and higher SII indicates stimulated inflammatory activity. In view of this, SII is a more comprehensive indicator, which is reasonable in predicting delirium among ICU patients.
Second, from the pathophysiological perspective, neutrophil involves in the microglia activation42; and activated microglia exactly plays a critical role in delirium pathogenesis43. High levels of SII means strong inflammatory reaction and rising count of neutrophil, which could initiate the microglia activation and provoke delirium. This could also explain why SII and NLR both are positively associated with the occurrence of delirium.
Third, 5-hydroxytryptamine 3 (5-HT3) may be related. 5-HT3 is an essential excitatory neurotransmitter, and 5-HT3 receptor participates in emotional regulation44. It has been proved that 5-HT3 receptor antagonists can decrease the prevalence of POD in older patients undergoing orthopedic lower limb surgery45. Another fundamental experiment also proved 5-HT receptor antagonist can improve behavior performance of delirium through induced NLRP3 activity46. It’s worth noting that the 5-HT3 was stored in the dense granules of platelet, accounting for the most part in the peripheral circulation. When platelet activated, 5-HT3 can be secreted into serum47,48. All these studies seem to indicate the increased and activated platelet can lead to the cognition impairment via the metabolic disturbance of 5-HT3, and it is likely for 5-HT3 permeating through the blood–brain barrier impaired by inflammatory and changing neurotransmitter balance49. Another study demonstrated platelet transfusion increased the incidence of delirium with a dose–response effect50, which is an evidence accounting for the conclusion.
Finally, as previously described, symptomatic treatment by sedative and analgesics was the main therapy for delirium. The high levels of SII indicating severe status of ICU patients, who usually need large amount of sedation and analgesics and prolonged treatment of these drugs. This may partially explain the descending branch of RCS curves and the increased mortality in Cox regression.
In conclusion, this article revealed SII is positively associated with the delirium prevalence in ICU patients; high level of SII is an independent risk factor for mortality in critical patients. Besides, SII is a marker easy to acquire with low cost from routine blood examination in ICU. By assessing the possibility of delirium, SII can provide the basis for early intervention to delirium. This could decrease the difficulty to treat patients for medical workers, reduce the length and cost of hospital stay, and lower the mortality in ICU. Some limitations still need to be noted in the present study. First, this is a study from MIMIC database, which might affect the generalizability of the findings. The conclusion needs to be validated in different populations around the world; Second, this paper is only a retrospective study, so prospective clinical study should be conducted to cover the deficiency of retrospective study. Third, the underlying biological mechanism remains unclear. To further explore the mechanisms, relevant foundational experiments should be designed, which may be a new promising research point.
Data availability
“The data sets used for the analysis in the current study can be acquired at the website: https://mimic.mit.edu/.”
References
Stollings, J. L. et al. Delirium in critical illness: clinical manifestations, outcomes, and management. Intensive Care Med. 47, 1089–1103. https://doi.org/10.1007/s00134-021-06503-1 (2021).
Kennedy, M. et al. Delirium in older patients with COVID-19 presenting to the emergency department. JAMA Netw. Open 3, e2029540. https://doi.org/10.1001/jamanetworkopen.2020.29540 (2020).
Sanchez, D. et al. Frailty, delirium and hospital mortality of older adults admitted to intensive care: the Delirium (Deli) in ICU study. Crit. Care 24, 609. https://doi.org/10.1186/s13054-020-03318-2 (2020).
Alexandrou, D. N. C. et al. Delirium in the intensive care unit and its importance in the post-operative context: A review. Front. Med. (Lausanne) 10, 1071854. https://doi.org/10.3389/fmed.2023.1071854 (2023).
Bento, A. F. G. & Sousa, P. P. Delirium in adult patients in intensive care: nursing interventions. Br. J. Nurs. 30, 534–538. https://doi.org/10.12968/bjon.2021.30.9.534 (2021).
Wu, P., Yang, Y., Yuan, A., Wang, Y. & Zhang, Y. Postoperative delirium increases follow-up mortality following hip arthroplasty in older patients with femoral neck fracture. Australas. J. Ageing https://doi.org/10.1111/ajag.13366 (2024).
Wilson, J. E. et al. Delirium. Nat. Rev. Dis. Primers 6, 90. https://doi.org/10.1038/s41572-020-00223-4 (2020).
Tenser, R. B. Haloperidol and ziprasidone for treatment of delirium in critical illness. N. Engl. J. Med. 380, 1778. https://doi.org/10.1056/NEJMc1901272 (2019).
Faisal, H., Farhat, S., Grewal, N. K. & Masud, F. N. ICU delirium in cardiac patients. Methodist. Debakey Cardiovasc. J. 19, 74–84. https://doi.org/10.14797/mdcvj.1246 (2023).
Prendergast, N. T., Tiberio, P. J. & Girard, T. D. Treatment of delirium during critical illness. Annu. Rev. Med. 73, 407–421. https://doi.org/10.1146/annurev-med-042220-013015 (2022).
Devlin, J. W. et al. Clinical practice guidelines for the prevention and management of pain, agitation/sedation, delirium, immobility, and sleep disruption in adult patients in the ICU. Crit. Care Med. 46, e825–e873. https://doi.org/10.1097/ccm.0000000000003299 (2018).
Mart, M. F., Williams Roberson, S., Salas, B., Pandharipande, P. P. & Ely, E. W. Prevention and management of delirium in the intensive care unit. Semin. Respir. Crit. Care Med. 42, 112–126. https://doi.org/10.1055/s-0040-1710572 (2021).
Seo, Y., Lee, H. J., Ha, E. J. & Ha, T. S. 2021 KSCCM clinical practice guidelines for pain, agitation, delirium, immobility, and sleep disturbance in the intensive care unit. Acute Crit. Care 37, 1–25. https://doi.org/10.4266/acc.2022.00094 (2022).
Marcantonio, E. R. et al. A clinical prediction rule for delirium after elective noncardiac surgery. JAMA 271, 134–139 (1994).
Bilge, E., Kaya, M., Şenel, G. & Ünver, S. The incidence of delirium at the postoperative intensive care unit in adult patients. Turk. J. Anaesthesiol. Reanim. 43, 232–239. https://doi.org/10.5152/tjar.2015.93798 (2015).
Mazzola, P. et al. Association between preoperative malnutrition and postoperative delirium after hip fracture surgery in older adults. J. Am. Geriatr. Soc. 65, 1222–1228. https://doi.org/10.1111/jgs.14764 (2017).
Shoulders, B. R. et al. Risk factors for delirium and association of antipsychotic use with delirium progression in critically ill trauma patients. Am. Surg. 89, 1610–1615. https://doi.org/10.1177/00031348211069792 (2023).
Zhou, S., Shi, S., Xie, C. & Chen, G. Association between smoking and postoperative delirium in surgical patients with pulmonary hypertension: a secondary analysis of a cohort study. BMC Psychiatry 22, 371. https://doi.org/10.1186/s12888-022-03981-5 (2022).
Krinitski, D., Kasina, R., Klöppel, S. & Lenouvel, E. Associations of delirium with urinary tract infections and asymptomatic bacteriuria in adults aged 65 and older: A systematic review and meta-analysis. J. Am. Geriatr. Soc. 69, 3312–3323. https://doi.org/10.1111/jgs.17418 (2021).
Romero-Ortuno, R. Infection in dementia and delirium in frailty. Nat. Aging 1, 157–158. https://doi.org/10.1038/s43587-021-00031-9 (2021).
Wu, X. et al. Relationship between preoperative neutrophil-to-lymphocyte ratio and postoperative delirium: The PNDABLE and the PNDRFAP cohort studies. Brain Behav. 13, e3281. https://doi.org/10.1002/brb3.3281 (2023).
Dunne, S. S. et al. Biomarkers in delirium: A systematic review. J. Psychosom. Res. 147, 110530. https://doi.org/10.1016/j.jpsychores.2021.110530 (2021).
Song, Y. et al. Systemic immune-inflammation index predicts postoperative delirium in elderly patients after surgery: a retrospective cohort study. BMC Geriatr. 22, 730. https://doi.org/10.1186/s12877-022-03418-4 (2022).
Johnson, A. E. W. et al. MIMIC-IV, a freely accessible electronic health record dataset. Sci. Data 10, 1. https://doi.org/10.1038/s41597-022-01899-x (2023).
Chen, Z. et al. Predictive value of the geriatric nutrition risk index for postoperative delirium in elderly patients undergoing cardiac surgery. CNS Neurosci. Ther. https://doi.org/10.1111/cns.14343 (2023).
Liu, H. et al. Incidence and interaction factors of delirium as an independent risk of mortality in elderly patients in the intensive units: a retrospective analysis from MIMIC-IV database. Aging Clin. Exp. Res. 34, 2865–2872. https://doi.org/10.1007/s40520-022-02215-8 (2022).
Xiong, Y., Zhang, Y. C., Jin, T., Qin, F. & Yuan, J. H. Depressive males have higher odds of lower urinary tract symptoms suggestive of benign prostatic hyperplasia: a retrospective cohort study based on propensity score matching. Asian J. Androl. 23, 633–639. https://doi.org/10.4103/aja.aja_12_21 (2021).
Brun-Buisson, C. The epidemiology of the systemic inflammatory response. Intensive Care Med. 26(Suppl 1), S64-74. https://doi.org/10.1007/s001340051121 (2000).
Pang, Y. et al. Effects of inflammation and oxidative stress on postoperative delirium in cardiac surgery. Front. Cardiovasc. Med. 9, 1049600. https://doi.org/10.3389/fcvm.2022.1049600 (2022).
Yan, X., Huang, J., Chen, X. & Lin, M. Association between increased systemic immune-inflammation index and postoperative delirium in older intertrochanteric fracture patients. J. Orthop. Surg. Res 19, 219. https://doi.org/10.1186/s13018-024-04699-8 (2024).
Chakraborty, R. K. & Burns, B. in StatPearls (StatPearls Publishing Copyright © 2024, StatPearls Publishing LLC., 2024).
Lu, W., Zhang, K., Chang, X., Yu, X. & Bian, J. The association between systemic immune-inflammation index and postoperative cognitive decline in elderly patients. Clin. Interv. Aging 17, 699–705. https://doi.org/10.2147/cia.S357319 (2022).
Xiang, D., Xing, H. & Zhu, Y. A predictive nomogram model for postoperative delirium in elderly patients following laparoscopic surgery for gynecologic cancers. Support Care Cancer 31, 24. https://doi.org/10.1007/s00520-022-07517-1 (2022).
Guldolf, K. et al. Neutrophil-to-lymphocyte ratio predicts delirium after stroke. Age Ageing 50, 1626–1632. https://doi.org/10.1093/ageing/afab133 (2021).
Dragoescu, A. N. et al. The role of neutrophil-to-lymphocyte ratio (NLR) in urosepsis-associated delirium. Cureus 16, e62110. https://doi.org/10.7759/cureus.62110 (2024).
Seo, C. L. et al. Neutrophil-lymphocyte ratio as a potential biomarker for delirium in the intensive care unit. Front. Psychiatry 12, 729421. https://doi.org/10.3389/fpsyt.2021.729421 (2021).
Forget, M. F. et al. Delirium and inflammation in older adults hospitalized for COVID-19: A cohort study. Clin. Interv. Aging 16, 1223–1230. https://doi.org/10.2147/cia.S315405 (2021).
Girard, T. D. et al. Associations of markers of inflammation and coagulation with delirium during critical illness. Intensive Care Med. 38, 1965–1973. https://doi.org/10.1007/s00134-012-2678-x (2012).
Schreuder, L. et al. Pathophysiological and behavioral effects of systemic inflammation in aged and diseased rodents with relevance to delirium: A systematic review. Brain Behav. Immun. 62, 362–381. https://doi.org/10.1016/j.bbi.2017.01.010 (2017).
Guo, Z., Zhang, Z., Prajapati, M. & Li, Y. Lymphopenia caused by virus infections and the mechanisms beyond. Viruses https://doi.org/10.3390/v13091876 (2021).
Araneda, M. et al. Reactive and clonal thrombocytosis: proinflammatory and hematopoietic cytokines and acute phase proteins. South Med. J. 94, 417–420 (2001).
Bozoyan, L., Dumas, A., Patenaude, A. & Vallières, L. Interleukin-36γ is expressed by neutrophils and can activate microglia, but has no role in experimental autoimmune encephalomyelitis. J. Neuroinflammation 12, 173. https://doi.org/10.1186/s12974-015-0392-7 (2015).
Hoogland, I. C., Houbolt, C., van Westerloo, D. J., van Gool, W. A. & van de Beek, D. Systemic inflammation and microglial activation: systematic review of animal experiments. J. Neuroinflammation 12, 114. https://doi.org/10.1186/s12974-015-0332-6 (2015).
Franco, R. et al. 5-hydroxytryptamine, glutamate, and ATP: much more than neurotransmitters. Front. Cell Dev. Biol. 9, 667815. https://doi.org/10.3389/fcell.2021.667815 (2021).
Shin, H. J., Yoon, J. & Na, H. S. 5-HT(3) receptor antagonists decrease the prevalence of postoperative delirium in older patients undergoing orthopedic lower limb surgery. Perioper Med (London) 10, 51. https://doi.org/10.1186/s13741-021-00222-3 (2021).
Qiu, Y. et al. 5-HT(1A) receptor antagonist improves behavior performance of delirium rats through inhibiting PI3K/Akt/mTOR activation-induced NLRP3 activity. IUBMB Life 68, 311–319. https://doi.org/10.1002/iub.1491 (2016).
Holinstat, M. Normal platelet function. Cancer Metastasis Rev. 36, 195–198. https://doi.org/10.1007/s10555-017-9677-x (2017).
Mendelson, S. D. The current status of the platelet 5-HT(2A) receptor in depression. J. Affect Disord. 57, 13–24. https://doi.org/10.1016/s0165-0327(99)00177-9 (2000).
Ritter, C. et al. Inflammation biomarkers and delirium in critically ill patients. Crit. Care 18, R106. https://doi.org/10.1186/cc13887 (2014).
Nelligan, M., Nellis, M. E., Mauer, E. A., Gerber, L. M. & Traube, C. Association between Platelet Transfusion and Delirium in Critically Ill Children. Children (Basel) https://doi.org/10.3390/children10050825 (2023).
Acknowledgements
We thank all staff and patients involved in the construction of the MIMIC-IV database.
Author information
Authors and Affiliations
Contributions
Yanyuan Zhang analyzed the data and wrote the main manuscript text; Feifei Xu prepared Figs. 1,2,3,4,5 and tables. Shurong Zhang revised the paper and performed additional data analyses; Yanyuan Zhang, Feifei Xu and Shurong Zhang participated in the study design. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Ethics approval and consent to use MIMIC-IV database were approved by the Institutional Review Boards of the Massachusetts Institute of Technology, and consent was obtained for the original data collection. Thus, the informed consent was waived by an Institutional Review Board of West China Hospital, Sichuan University, or by ethics committee.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xu, F., Zhang, S. & Zhang, Y. High level of systemic immune inflammation index elevates delirium risk among patients in intensive care unit. Sci Rep 14, 30265 (2024). https://doi.org/10.1038/s41598-024-81559-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-81559-9