Abstract
Whether lamotrigine (LTG) is associated with ventricular tachycardia (VT) in bipolar disorder (BPD), partial seizures (PSZ) and generalized tonic-clonic seizures (GTSZ) with and without structural heart disease (SHD) remains controversial leading to a real-world comparative cohort observational study. A retrospective observational comparative safety study was performed using a large healthcare claims database of adult participants, analyzing the risk of VT incidence. Patients diagnosed with BPD, PSZ or GTSZ where free from supraventricular (SVT) or VT during the 6-month baseline period. Exposure to LTG versus commonly prescribed alternative agents was the CTR. One-year cumulative VT incidence was calculated for GTSZ, PSZ and BPD using Kaplan-Meier estimator adjusted for baseline characteristics. The analytic cohort included 153,852 LTG and 213,593 CTR for BPD, 10,275 LTG and 24,971 CTR for PSZ, and 5,860 LTG and 17,506 CTR for GTSZ. The 1-year VT cumulative incidence from LTG or CTR free from was 0.79% vs. 0.68% in BPD, 0.76% vs. 0.58% in PSZ, and 0.93% vs. 0.40% in GTSZ cohorts, The adjusted HR [95% CI] estimates were 1.326 [1.122–1.568, p < 0.01], 1.403 [0.920–2.138, p = 0.11], and 1.180 [0.607–2.295, p = 0.63]. In adult participants with BPD, LTG has a statistically significant association to increase VT risk compared to commonly prescribed alternatives.
Similar content being viewed by others
Introduction
Lamotrigine (LTG) is widely prescribed in the U.S., with thirteen million annual prescriptions for two million children, adolescents and adults1 The drug has been established as first-line therapy for primary generalized tonic-clonic and partial onset seizures, and bipolar I disorder2,3,4 The mechanism of action of LTG is not completely elucidated, but is believed to selectively bind and inhibit voltage-gated sodium channels (INa), stabilizing presynaptic neuronal membranes and inhibiting presynaptic glutamate release5–6 Case reports of lamotrigine-associated electrocardiographic events in epilepsy and bipolar disorder patients, with and without structural heart disease, have been published7,8,9,10 Paired with recent publications that the inhibitory effect of LTG on INa blockade can occur at effective plasma concentrations that lead to pro-arrhythmia events,11–12 prompted the United States Food and Drug Association (FDA) to issue a warning that “Lamictal exhibits class IB antiarrhythmic activity at therapeutically relevant concenttrations”13 This warning was issued due to evidence of LTG slowing conduction velocity, widening the QRS interval, leading to higher risk of pro-arrhythmic events in patients with structural heart disease and/or myocardial ischemia, but not in healthy subjects13 New findings suggest that LTG may have enhanced affinity for the INa which may, in part, explain the potential for a clinical arrhythmogenic phenotype noted in epilepsy and possibly in bipolar disorders11,14 However, not all data has shown a significance difference in arrhythmogenesis with lamotrigine14–15 The current practice remains to be vigilant to the effects of LTG and consult a cardiologist before starting the agent, especially in patients with structural heart disease13.
To this end, a large population-based real-world study was designed to determine if LTG monotherapy was associated with ventricular tachycardia incidence in participants with and without structural heart disease treated for partial-, complex-seizures and bipolar disorders. Addressing this knowledge gap appears to be crucial to confirm the safety for millions of people prescribed LTG.
Methods
Study design and participants
This study is a retrospective comparative safety cohort study using Merative Health MarketScan® Commercial Claims and Medicare Supplemental databases (Merative L.P., Ann Arbor, MI). The data source covered medical and outpatient pharmacy claims from January 2009 to December 2021. Direct patient identifiers were removed before investigators had access to the data. Therefore, informed consent was not obtained from subjects in this study. All research was performed in accordance with relevant guidelines/regulations for human subjects’ research and the STROBE Guidelines for observational research and in accordance with the Declaration of Helsinki. The University of Illinois at Chicago Institutional Review Board deemed the use of the database for this study exempt.
Participants were defined based on the major LTG indications, namely bipolar I disorder (BPD), partial seizure (PSZ), or generalized tonic-clonic seizure (GTSZ), defined by the International classification of Diseases Ninth revision (ICD-9: 345.4, 345.5, 345.1, 296 and subcodes) or Tenth revision (ICD-10: G40.0, G40.1, G40.2, G40.6, F31 and subcodes) as a diagnosis of respective condition at either inpatient or outpatient setting. Eligible subjects must have had a record of dispensing LTG to be assigned to the LTG exposure cohort (LTG) or dispensing alternative medications indicated for respective condition to become a control subject (CTR): lithium, quetiapine, valproate or risperidone for BPD; carbamazepine, levetiracetam, oxcarbazepine or eslicarbazepine for PSZ; valproate, levetiracetam, carbamazepine or zonisamide for GTSZ. Prescription dispensing for each drug was defined by the National Drug Codes and generic name available from the outpatient pharmacy service records. The index date was defined by the first dispensing of the respective medication with a presence of BPD, PSZ, or GTSZ diagnosis within 180-day baseline period, without any record of exposure or comparator during the baseline period. The index medication had to be given at least seven days or longer without crossover between LTG and CTR.
To minimize confounding by pre-existing condition on the measure of association, we excluded participants who had ventricular tachycardia (VT), during the baseline period as defined by diagnosis codes (ICD-9 427 and 780.2 including all subcodes; ICD-10 I47, I48, I49, and R55 including all subcodes). Children or adolescents (< 18 years) at index date were excluded from the analytic cohort.
Baseline characteristics, outcome and follow-up
Participant characteristics were collected at index date and during the 180-day baseline period. Demographics include age, sex, geographic location, and type of insurance. Clinical characteristics included chronic conditions and past/concurrent medications related to an arrhythmia or conditions to which the FDA LTG warning applied. We used diagnosis codes for the Charlson Comorbidity Index score and other relevant conditions (i.e., hypertension, hypercholesterolemia, depression, anxiety, substance abuse, eating disorders), and generic medication names or medication categories (angiotensin-converting enzyme inhibitors [ACEi], angiotensin II receptor blockers [ARB], non-dehydropyridine calcium channel blockers [NDHP-CCB], ßeta-adrenergic blockers [BB], spironolactone [SPL], and statins)16,17,18 The impact of structural heart disease (SHD) on the association between LTG and VT13 were applied (Supplement material, Table S1).
The outcome of interest was the onset of VT following the exposure to LTG or CTR for 7 days or longer period. The outcome was defined by ICD-9 (427.4x, 427.8x, 427.9x and 780.2x) or ICD-10 CM (I49.x and R55.x) diagnosis code for VT in any position from an outpatient service encounter, or in the first four positions from inpatient admission encounter. We followed the healthcare records for up to 365 days from the index date to assess the outcome. All participants met at least 7-day enrollment from the index date, during which subjects were free from the outcome of interest or the end of enrollment. Participants were required to have ≥ 7 days of medication supply. To minimize potential contamination between lamotrigine and comparator medications, participants were censored from the study population if those in the treatment group switched to comparator medications or vice versa.
Statistical analysis
Descriptive statistics and bivariate analyses were conducted to compare baseline characteristics between LTG and CTR cohorts. Summary statistics of age in year were mean and standard deviation (SD), which were compared using student t-test. Age was grouped into four categories of 18–25, 25–44, 45–64, and ≥ 65 years. All the other categorical variables were summarized using frequency (n) and percentage (%) out of the total number of participants in each cohort. We used Chi-square test for the statistical comparison of the categorical variables.
Time to VT and cumulative incidence was analyzed using a Kaplan-Meier estimator, for which participants were censored with treatment switching between LTG and CTR medications, the last date with available medication as defined by the days of supply from the most recent dispensing, or end of one-year follow up, whichever incurred first. CTR patient follow-up continued irrespective of drug switching among the control medications.
The measure of association was hazard ratio (HR). We calculated the HR and 95% confidence interval [95CI] of VT for LTG versus CTR using Cox-proportional hazard regression model. To adjust for the potential confounders, the multivariable regression included baseline characteristics that were significantly different (p < 0.05) as covariates.
We assessed the impact of SHD on the risk of VT. First, we included SHD as part of covariate of the multivariable regression analysis with and without all the other covariates. The regression was followed by the inclusion of interaction between SHD and LTG, which allowed us to assess if the impact of LTG on the onset of VT differs by baseline SHD.
Sensitivity analysis – propensity score matching
Sensitivity estimates and statistical conclusion to the analytic cohort definition for each of the three indications (BPD, PSZ, and GTSZ) were assessed using propensity score (PS) matching (Supplement Table S2). The PS to receive LTG versus comparator was calculated using logistic regression where all baseline characteristics were assessed as regressor on the stepwise selection process with p < 0.15 as a selection criterion. LTG and CTR participants were exactly matched based on age group, sex, and propensity score caliper of 0.2 times of the standard deviation range. Cumulative incidence estimation using Kaplan-Meier approach and HR [95CI] calculation was undertaken using the PS-matched group where residual confounder after matching (standardized mean difference > 0.1) were adjusted.
Results
Patient characteristics
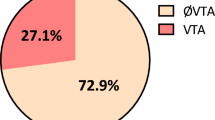
The three analytic cohorts for BPD, PSZ and GTSZ were made up of 367,445 (153,852 LTG and 213,593 CTR), 35,246 (10,275 LTG and 24,971 CTR), and 23,366 (5,860 LTG and 17,506 CTR) participants satisfying all inclusion and exclusion criteria, respectively (Fig. 1). Across the BPD, PSZ and GTSZ cohorts, LTG participants were younger than CTR participants with the respective mean year age (SD) of 37.5 (13.6) vs. 40.9 (15.7), 41.3 (16.1) vs. 46.3 (17.5) and 36.8 (15.6) vs. 42.5 (17.6), and slightly more female dominated (69.7% vs. 59.9% BPD, 60.6% vs. 56.2% PSZ, and 60.0% vs. 53.2% GTSZ).
While comorbidity profiles show that participants were generally healthy, the LTG cohort carried a lower risk of developing cardiovascular complications compared to the CTR cohort. The proportion of participants with history of acute myocardial infarction (AMI), congestive heart failure (CHF), peripheral vascular disease, hypertension and hypercholesterolemia were lower in the LTG cohort versus CTR cohort with a strong statistical significance (p < 0.01) across all the three diagnostic groups. The prevalence of SHD21 was significantly lower among LTG cohorts than CTR cohorts. (Table 1) Correspondingly, LTG participants were less likely to be prescribed ACEi, ARB, BB, NDHP-CCB and statin than CTR participants.
Outcome
Cumulative incidence of VT at 12 months among LTG was 0.79%. 0.76% and 0.93% in BPD, PSZ, and GTSZ cohorts, respectively, higher than the estimates from the CTR cohort (p-value from log-rank test) of 0.68% (0.0096), 0.58% (0.1245), and 0.40% (0.0092).(Fig. 1) The difference in the cumulative incidence resulted in the crude HR [95CI] of VT for LTG versus CTR of 1.270 for BPD [1.079–1.495], 1.381 [0.913–2.087] for PSZ, and 1.245 [0.647–2.396], for GTSZ, respectively. After statistical adjustment using a multivariable regression model, the risk of VT among LTG in BPD, but not in PSZ or GTSZ, the HR [95CI] was 1.326 [1.122–1.568] for BPD, 1.403 [0.920–2.138] for PSZ, and 1.180 [0.607–2.295] for GTSZ. (Fig. 2)
Our analysis also demonstrated an increase in the risk of VT with SHD. With SHD as the single covariate of the LTG vs. CTR, the HR [95CI] was 1.878 [1.339–2.635] for BPD, 1.393 [0.802–2.417] for PSZ, 1.931 [0.937–3.981] for GTSZ. SHD was positively associated with the onset of VT, but the 95CI started to cross the null when SHD was a part of the full multivariable regression model with the respective HR [95CI] of 1.538 [1.011–2.341], 1.539 [0.752–3.149], 2.056 [0.775–5.45]. Analysis including the interaction term resulted in a strong positive but statistically insignificant difference of SHD impact on LTG versus CTR (HR [95CI] of 1.615 [0.819–3.183] for BPD, 1.35 [0.396–4.606] for PSZ, and 1.789 [0.319–10.031] for GTSZ, respectively. (Table 2)
Sensitivity analysis
A matched cohort with 148,645, 10,275 and 5,855 pairs for BPD, PSZ and GTSZ was created. The rate of the onset of VT was higher among those exposed to LTG compared to comparators, with 365-day cumulative incidence estimates of 0.60 vs. 0.51% (log-rank test, p = 0.0173) in the BPD, 0.76 vs. 0.67% (log-rank test, p = 0.1323) in the PSZ, and 0.54 vs. 0.21% (log-rank test, p = 0.3987) in the GTSZ cohort. The respective HR [95CI] for LTG versus CTR at 12-month follow-up were 1.243 [1.039–1.487] for BPD, 1.477 [0.886–2.462] for PSZ, and 1.427 [0.622–3.271] for GTSZ. (Fig. 3)
Discussion
In a large observational cohort study from a United States-based commercial insurance and Medicare supplement claims database, LTG exposure was associated with an increase in the cumulative incidence of ventricular tachycardia across the diagnostic categories of partial and primary generalized onset seizures, and bipolar disorders when compared to commonly used agents. After adjustment for multivariate regression model, LTG use in BPD remained significant, but not for the epilepsy cohorts. The association was significant when controlled for SHD as a single covariate but was insignificant when the multivariate model included SHD on top of the other covariates. The increase in ventricular tachycardia incidence was approximately 1% of the population. Most of the encounters with the diagnosis of VT occurred in the first 30 days. The measure of association was robust across the different cohort set-up and analytic approaches.
This study is the first to report a higher risk of LTG-associated ventricular tachycardia in BPD participants, with and importantly without structural heart disease. These findings project the number needed to harm of 980, placing 3% of the 7 million bipolar disorder I persons ≥ 45 years, approximately 210,000 participants, at risk of LTG-associated ventricular tachycardia yearly19 LTG-associated ventricular tachycardia may be more prevalent in males between 45 and 64 years compared to younger participants, findings previously noted9 VT onset of approximately 20–30 + days (Fig. 1), may be associated with up-titration of LTG dosage and administration guidelines in pre-disposed at-risk participants20 LTG has distinct offset kinetics that with elevated heart rate may result in excessive INa block21 Thereby, higher LTG concentrations with greater affinity for the INa, may contribute to enhanced arrhythmogenic phenotype in BPD patients11,22–23 Our finding of an increase in LTG-associated VT in BPD adult participants without structural heart disease maybe have been masked over time by high number of suicides recorded with BPD. BPD has the highest rate of suicide of all psychiatric conditions and is approximately 20–30 times that of the general population24.
LTG in participants without structural heart disease is a new finding given that healthy people undergoing QT testing have not shown changes in QRS duration,25 but QRS prolongation should be considered a risk for LTG ventricular tachycardia26 Toxic concentrations of LTG (i.e., 7–10 fold higher than therapeutic concentrations) have been associated with QRS prolongation and/or development of sustained ventricular tachycardia12 LTG exhibits voltage-gated sodium channel use-dependent block properities27 supporting that rapid heart rates (i.e., exercise) may be associated with ventricular tachycardia risk at human physiologic concentrations12 Outside of sodium channel availability,28 ventricular conduction slowing is partly determined by resistance across intracellular connections, or gap junctions, between cardiac myocytes29 Ventricular gap junctions are constituted by connexin proteins with connexin isoform-43 being the predominant protein in humans30 In studies of human cardiac gap junctions using a dye transfer “parachute assay” to determine IC50 values for compounds, LTG was found to uncouple connexin isoform-43 at clinically relevant concentrations31 This activity is associated with proclivity for ventricular conduction slowing and maybe dependent on direct gap junction uncoupling or by non-gap-junction-mediated ephaptic mechanisms32 Thereby, LTG, a potent Nav1.5 blocker, may also uncouple connexin 43, thereby heterogeneously slowing ventricular conduction velocity, causing ventricular tachycardia.
The potential association between LMG and VT remains unclear. A meta-analysis of 26 studies with 24,962 patients, from which 2326 were LMG users, found no positive correlation between lamotrigine and ECG abnormalities or sudden death33 A Danish population-based cohort study of 90,000 + patients, did not report an association between LMG and cardiac conduction disorders in people without pre-existing cardiac morbidity, or all-cause mortality with pre-existing cardiac conditions34 Additionally, a retrospective, nested case–control study of 101 sudden unexpected death in epilepsy (SUDEP) cases and 199 living epilepsy controls concluded that LMG is unlikely to be associated with an increased long-term risk of SUDEP35 In contrast, a retrospective study with 96 elder Veteran Affairs (VA) hospital patients detected abnormalities in the ECGs in 7 patients, potentially related to LMG36 Notably, Aboukaoud et al. performed a retrospective pharmacovigilance study using the FDA Adverse Event Reporting System (FAERS) database to explore the potential link between LMG use and arrhythmias14 In the 2917 cases, LMG use was associated with a non-significant higher risk of cardiac arrest, particularly in patients prescribed the medication for psychiatric indications. This outcome may be due to the low incidence of VT in the population at large (< 1%), and small number of events and effect size37 Recent observation cohort study of over 140,000 patients has confirmed a strong association between lamotrigine and risk of VT38 Importantly, patients treated with LMG were at risk of VT, independent of SHD.
Limitations
Interpreting our findings, readers should be mindful of several limitations. First, VT is correlated with mortality,39–40 but we were unable to link mortality from the claim records to death certificates. Thereby, detected VT could be subject to the survivalship bias that needs to be determined and many patients who already have VT might not have a chance to claim the condition, which likely had a significant impact on the measure of association. Second, our study is subject to event size. To determine the statistical significance of the LTG versus CTR and its interaction with baseline characteristics, a sufficient number of events and absolute difference is required. Since our VT cumulative incidence estimates were less than 1% across the three diagnosis criteria, statistical interpretation is limited by the small number of events and effect size. Third, our study included a large number of regression variables, which raises concern on the potential interactions and collinearity among independent variables. Although this would not significantly change the magnitude or direction of the LTG-outcome association, future studies need to focus on the other risk factors that could modify the measure of association. Lastly, any interpretation should be considered considering the limitations of insurance claims. Our study findings and conclusions may be subject to miscoding and unobserved confounders. In addition, the study findings have limited generalizability to the commercially insured population. The validity of using diagnosis codes to determine VT event will be another theme of future research.
Conclusions
This study provides a general insight into the positive real-world association between LTG use and VT. The LTG-VT association was clinically and statistically significant in adult BPD participants. Although limited statistical significance, the positive LTG-VT association is ubiquitous across epileptic conditions. SHD has a notable increased effect on the incidence on the onset of VT.
Data availability
The datasets generated during the current study are available from the corresponding author on request.
Abbreviations
- ACEi:
-
Angiotensin-converting enzyme inhibitor
- ARB:
-
Angiotensin receptor blocker
- BB:
-
ßeta-adrenergic blocker
- BPD:
-
Bipolar 1 disorder
- CTR:
-
Control group
- FDA:
-
U.S. food and drug administration
- GTSZ:
-
Generalized tonic-clonic seizure
- HR:
-
Hazard ratio
- INa :
-
Voltage-gated sodium channels.
- LTG:
-
Lamotrigine
- NDHP-CCB:
-
Non-dihydropyridine calcium channel blocker
- PSZ:
-
Partial seizure
- PS:
-
Propensity score
- SHD:
-
Structural heart disease
- SPL:
-
Spironolactone
- SMD:
-
Standard mean difference
- QRS:
-
QRS complex
- QT:
-
QT interval
- VT:
-
Ventricular tachycardia
References
Kane, S. P. Lamotrigine - Drug usage statistics. Accessed 06/13/2024 https://clincalc.com/DrugStats/Drugs/Lamotrigine
Nolan, S. J., Tudur Smith, C., Weston, J. & Marson, A. G. Lamotrigine versus carbamazepine monotherapy for epilepsy: an individual participant data review. Cochrane Database Syst Rev. ;11(11):CD001031. doi: 10.1002/14651858.CD001031.pub3. Update in: Cochrane Database Syst Rev. 2018;6:CD001031. (2016). https://doi.org/10.1002/14651858.CD001031.pub4. Database Syst Rev. 2016;11(11):Cd001031. 10.1002/14651858.CD001031.pub3.
Ng, F., Hallam, K., Lucas, N. & Berk, M. The role of lamotrigine in the management of bipolar disorder. Neuropsychiatr Dis. Treat. 3 (4), 463–474 (2007).
Costa, B. & Vale, N. Understanding Lamotrigine’s role in the CNS and possible future evolution. Int. J. Mol. Sci. 24 (7), 6050. https://doi.org/10.3390/ijms24076050 (2023).
Xie, X., Lancaster, B., Peakman, T. & Garthwaite, J. Interaction of the antiepileptic drug lamotrigine with Recombinant rat brain type IIA Na + channels and with Na + channels in rat hippocampal neurones. Pfluegers Arch. Eur. J. Physiol. 430, 437–446 (1995).
Kuo, C. C. & Lu, L. Characterization of lamotrigine Inhibition of Na + channels in rat hippocampal neurones. Br. J. Pharmacol. 121 (6), 1231–1238. https://doi.org/10.1038/sj.bjp.0701221 (1997).
Herold, T. J. Lamotrigine as a possible cause of QRS prolongation in a patient with known seizure disorder. CJEM 8 (5), 361–364. https://doi.org/10.1017/s1481803500014093 (2006).
Chavez, P., Casso Dominguez, A. & Herzog, E. Evolving electrocardiographic changes in lamotrigine overdose: A case report and literature review. Cardiovasc. Toxicol. 15 (4), 394–398. https://doi.org/10.1007/s12012-014-9300-0 (2015).
Dream, A., Holmgren, H., Merrill, R. & Simon, E. L. Refractory ventricular fibrillation in patient taking lamictal. Am. J. Emerg. Med. 36 (7), 1324e. 1-1324.e2 (2018). Epub 2018 Apr 5.
Omer, H. et al. Unmasking of Brugada syndrome by lamotrigine in a patient with pre-existing epilepsy: A case report with review of the literature. Front. Cardiovasc. Med. 9, 1005952. https://doi.org/10.3389/fcvm.2022.1005952 (2022).
Contreras Vite, J. A., Vega Valle, C., Biekeu Mbem, H., Boivin, S-M. & Dumaine, R. Epilepsy-Induced high affinity blockade of the cardiac sodium current INa by lamotrigine; A potential for acquired arrythmias. Pharmaceuticals. 15(10)1208. INa by lamotrigine; A potential for acquired arrhythmias. Pharmaceuticals 2022;15:1208. (2022).
Xiaolei Meng, D. R. K. & Przemyslaw Radwanski LTG Promotes Heterogenous Cardiac Conduction Slowing and Reentrant Ventricular Tachycardia. American Epilepsy Society (2023). https://aesnet.org/abstractslisting/transcriptome-wide-survey-via-rna-seq-identifies-alterations-in-splicesosome-gene-expression-in-lymphoblast-cell-lines-from-dravet-syndrome-patients Accessed 02/20/2024.
French, J. A. et al. FDA safety warning on the cardiac effects of LTG: an advisory from the ad hoc ILAE/AES task force. Epilepsy Curr. 21 (3), 1535759721996344. https://doi.org/10.1177/1535759721996344 (2021).
Aboukaoud, M., Wilf-Yarkoni, A. & Maor, E. Investigation of cardiac arrhythmia events in patients treated with lamotrigine: FDA adverse event reporing system analysis. Epilepsia 64 (9), 2322–2329. https://doi.org/10.1111/epi.17696 (2023). Epub 2023 Jul 2.
Chistensen, J., Trabjerg, B. B. & Dreier, J. W. Cardiac morbidity and mortality associated with the use of lamotrigine. Epilepsia 63, 1271–1280. https://doi.org/10.1111/epi.17339 (2022).
Glasheen, W. P. et al. Charlson Comorbidity Index: ICD-9 Update and ICD-10 Translation. Am. Health Drug Benefits. 12 (4) 188–197. (2019).
Quan, H. et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med. Care. 43 (11), 1130–1139. https://doi.org/10.1097/01.mlr.0000182534.19832.83 (2005).
Husein, N., Thijs, R. D., Bunschoten, J. W., Keezer, M. R. & Sander, J. W. Concerns about lamotrigine. Lancet Neurol. 20 (6) 418–419. https://doi.org/10.1016/S1474-4422(21)00132-0. PMID: 34022162. (2021).
Bipolar Disorder. National Insitute of Menal Health. https://www.nimh.nih.gov/health/statistics/bipolar-disorder Accessed 06/13/2024.
Orts, L., Flumian, C. & Montastruc, F. Lamotrigine and risk of arrhythmias: A global pharmacovigilance analysis. J. Neurol. Sci. 448, 120644. https://doi.org/10.1016/j.jns.2023.120644 (2023).
Lamotrigine Prescribine Information. GlaxoSMithKline. Research Triangle Park. 10/12/2020.
Ingleby-Talecki, L. et al. Cardiac sodium channel inhibition by lamotrigine: in vitro characterization and clinical implications. Clin. Transl Sci. 2022 15 (8) 1978–1989. https://doi.org/10.1111/cts.13311. Epub 2022.
Goto, A. et al. Reverse translational analysis of clinically reported, lamotrigine-induced cardiovascular adverse events using the halothane-anesthetized dogs. Heart Vessels. 36 (3), 424–429. https://doi.org/10.1007/s00380-020-01716-8 (2021). Epub 2020.
Jia, L., Verkerk, A. O. & Tan, H. L. The Anti-Epileptic drugs lamotrigine and valproic acid reduce the cardiac sodium current. Biomedicines 11 (2), 477. https://doi.org/10.3390/biomedicines11020477 (2023).
Miller, J. N. & Black, D. W. Bipolar disorder and suicide: a review. Curr. Psychiatry Rep. 22 (2), 6. https://doi.org/10.1007/s11920-020-1130-0 (2020).
Dixon, R. et al. LTG does not prolong QTc Ina thorough QT/QTc study in health subjects. Brit J. Clin. Pharmacol. 66 (3), 396–404. https://doi.org/10.1111/j.1365-2125.2008.03250.x (2008).
Restrepo, J. A. et al. The assessment of cardiac risk in patients taking lamotrigine; a systematic review. Gen. Hosp. Psychiatry. 78, 14–27 (2022). Epub 2022 Jun 22.
Macdonald, R. L. & Kelly, K. M. Antiepileptic drug mechanisms of action. Epilepsia. 36 2 S2-12. (1995). https://doi.org/10.1111/j.1528-1157. 1995.
Radwański, P. B., Greer-Short, A. & Poelzing, S. Inhibition of Na + channels ameliorates arrhythmias in a drug-induced model of Andersen-Tawil syndrome. Heart Rhythm. 10 (2) 255 – 63 https://doi.org/10.1016/j.hrthm.2012.10.005. Epub. PMID: 23041575. 2012.
Cruciani, V. & Mikalsen, S. O. The vertebrate connexin family. Cell. Mol. Life Sci. 63 (10), 1125–1140. https://doi.org/10.1007/s00018-005-5571-8 (2006).
Severs, N. J. et al. Gap junction alterations in human cardiac disease. Cardiovasc. Res. 62(2), 368–377. https://doi.org/10.1016/j.cardiores.2003.12.007 (2004).
Burnham, M. P. et al. Investigation of connexin 43 uncoupling and prolongation of the cardiac QRS complex in preclinical and marketed drugs. Br J Pharmacol. 171(21) 4808-19. (2014). https://doi.org/10.1111/bph.12554. 2014.
Veeraraghavan, R. et al. Sodium channels in the Cx43 gap junction perinexus may constitute a cardiac ephapse: an experimental and modeling study. Pflugers Arch. ;467(10):2093 – 105. (2015). https://doi.org/10.1007/s00424-014-1675-z. Epub 2015. atrioventricular junction and atrioventricular node in the rat. Circulation. 2006;114(13):1360-71. doi: 10.1161/CIRCULATIONAHA.106.613182. Epub 2006.
Bunschoten, J. W. et al. Sudden death and cardiac arrythmia with lamotrigine: A rapid systematic review. Neurology 26 98 (17), e1748–e1760 (2022).
Christensen, J., Trabjerg, B. B. & Dreier, J. W. Cardiac morbidity and mortality are associated with the use of lamotrigine. Epilepsia 63 (9), 2371–2380 (2022).
Nightscales, R. et al. Risk of sudden unexpected death in epilepsy (SUDEP) with lamotrigine and other sodium channel-modulating Antiseizure medications. Epilepsia Open. 8 (2), 334–345 (2023).
Biehl, A. et al. Lamotrigine use and potential for adverse cardiac effects: A retrospective evaluation in a veteran population. Epilepsy Behav. EB. 149, 109496 (2023).
Sirichand, S. et al. Incidence of idiopathic ventricular arrhythmias: A population-based study. Circ. Arrhythm. Electrophysiol. 10 (2), e004662 (2017).
Kim, S. et al. Association of ventricular arrhythmias with lamotrigine: an observational cohort study. MedRxiv Prepr Serv. Health Sci. 2024 2024.09.10.24313446.
Foth, C., Gangwani, M. K., Ahmed, I. & Alvey, H. Ventricular tachycardia. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; (2024).
Acknowledgements
We express our gratitude to Marco Bortalato, M.D., Ph.D., Professor of Pharmacology and Toxicology, University of Utah for his expertise and counsel towards this manuscript.
Funding
National Institutes of Health from grants R01HL155378 and R01 NS121234 to Dr. Radwanski.
Author information
Authors and Affiliations
Contributions
All authors fully contributed to the content of this manuscript, including meeting the four criteria of the Internal Committee of Medical Journal Editors. All authors had full access to all the data in the study and took full responsibility for integrity of the work and accuracy of the data analysis, from inception to published article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kim, S., Welch, L., De Los Santos, B. et al. Association of ventricular arrhythmias with lamotrigine: an observational cohort study. Sci Rep 15, 19542 (2025). https://doi.org/10.1038/s41598-025-01321-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-01321-7