Abstract
Despite progress in HIV control, gaps persist in understanding the prevalence, drug resistance, and associated factors in urban Ethiopia. This cross-sectional study analyzed data from 19,136 consenting participants (aged 15–64 years) for interview and blood testing in the 2017–2018 Population-based HIV Impact Assessment (PHIA) survey to assess HIV-1 prevalence, drug resistance mutations, and associated factors. Participants confirmed to be living with HIV (PLHIV) provided additional samples for CD4 + T-cell counts, viral load testing, and drug resistance analysis. Forty-three samples were analyzed for viral subtyping and drug resistance profiling, of which 42 samples successfully amplified. Data were analyzed using descriptive statistics and logistic regression. The overall HIV-1 prevalence was 3%. Among genotyped samples, 79.1% harbored drug resistance mutations, with high rates of resistance to Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs) (78.8%) and Nucleoside Reverse Transcriptase Inhibitors (NRTIs) (66.7%). Regarding associated factors, females had 1.7 times higher odds of HIV infection than males (AOR = 1.7). Participants aged 35–44 and 45–54 had significantly higher odds of infection (AOR = 6.7 for both) compared to those aged 15–24. Lower educational attainment (primary school: AOR = 2.5; secondary school: AOR = 2.2) was associated with increased HIV prevalence compared to post-secondary education. Previously married individuals had higher infection odds than never-married participants (AOR = 2.7). Geographically, residents of Gambella showed significantly higher odds (AOR = 2.8) compared to those in Tigray. This study identified a high HIV-1 prevalence in Ethiopia, along with elevated virological failure rates, a significant proportion of PLHIV unaware of their status, and widespread drug resistance. Sex, age, education level, marital status, and region were the identified factors associated with HIV-1 prevalence. To improve treatment outcomes and curb transmission, Ethiopia should strengthen HIV prevention strategies focused on vulnerable populations, enhance testing and linkage to care, and implement routine drug resistance monitoring.
Similar content being viewed by others
Background
The Human Immunodeficiency Virus (HIV) infections remain a major public health issue in Ethiopia, particularly in urban areas1,2. It can be classified as either recently acquired (within 6–12 months), characterized by high viral loads (≥ 1000 copies/mL) and distinct antibody maturation markers, or long-term infection (persisting beyond this period)3,4. Across Africa, HIV prevalence varies widely, ranging from 0.54% in Niger to 23.98% in Lesotho5. In Ethiopia, HIV-1 is the predominant strain, accounting for over 95% of HIV cases6, with its prevalence showing significant regional variation, reaching 3.4% in Addis Ababa city and 4.8% in the Gambella region7.
The emergence of drug resistance mutations (DRMs) is a growing concern, significantly compromising the efficacy of antiretroviral therapy (ART)8,9,10. Mutations such as M184V and K103N11,12 are increasingly reported, with cross-class resistance13. Previous studies highlighted a high resistance to protease inhibitors, including atazanavir/ritonavir (28.6%) and lopinavir/ritonavir (14.3%)14, and 9.8% for Non-Nucleoside Reverse Transcriptase Inhibitor (NNRTI) mutations in the ART-naïve population15. For patients on ART, the existence of dual resistance to Nucleoside/Nucleotide Reverse Transcriptase Inhibitors (NRTIs) and NNRTIs is alarmingly prevalent (65.3%)16, with mutations including K65R and M184V having been frequently observed in first-line ART failures17,18.
As previous studies showed, HIV transmission in Ethiopia is driven by a combination of behavioral, socioeconomic, and structural factors19,20. The key behavioral factors include inconsistent condom use1,21,22,23, multiple sexual partners1,22,23,24,25, transactional sex23, early sexual debut24, and substance use21,25,26. Socioeconomic disparities further exacerbate the epidemic, including poverty1,22,27,28,29, low education levels1,22,30, limited healthcare access27,30, urban residence28,29,31, and the lack of media exposure28.
While previous studies have provided valuable insights, their small-scale designs limit generalizability to Ethiopia’s urban population. By analyzing the PHIA dataset, this study generated evidence on HIV-1 prevalence, drug resistance, and associated factors in urban HIV dynamics that can inform targeted interventions, optimize ART regimens, and guide national HIV policies to curb the epidemic effectively.
Methods
Survey setting and population characteristics
The survey was conducted in Ethiopia, a country divided into nine regional states and two city administrations. The study targeted urban areas across Ethiopia, focusing on participants aged 15 to 64 years. Data collection took place from October 2017 to April 2018 as part of the EPHIA survey. The survey was designed to generate comprehensive national and subnational estimates of HIV-1 prevalence, viral load suppression, and other key indicators among urban residents.
Sampling strategy and study population
The survey utilized a multi-stage stratified sampling design. In the first stage, 393 enumeration areas (EAs) were selected. From each EA, an average of 30 households were randomly chosen, resulting in a total of 11,810 households. Overall, 19,136 study participants aged 15 to 64 consented to participate in the interview and HIV-1 testing (Fig. 1).
Data collection procedures
The data collection process was conducted through face-to-face interviews using tablet-based data capture systems. Trained interviewers gathered information at the household level across all urban administrative regions of the country. The survey collected data on demographic characteristics, socioeconomic factors, HIV testing history, knowledge about HIV/AIDS, sexual behavior and practices, HIV prevention methods, access to healthcare services, ART usage among people living with HIV (PLWH), and blood samples for biomarker analysis.
HIV screening, viral subtyping analysis, and drug resistance
Blood samples from consenting participants underwent on-site HIV-1 screening using the national testing algorithm (Wantai HIV 1/2 test, Uni-Gold HIV 1/2™ confirmatory test, and Vikia HIV 1/2 tie-breaker test). Additional samples from PLWH were collected for CD4 + T-cell counts, viral load testing, drug resistance analysis, and recent infection assessment. CD4 + T-cell counts were measured in the field for all PLWH participants and a 2% random sample of HIV-negative individuals using a Pima™ Analyzer and Cartridge by trained staff. Viral load testing was performed on plasma (Roche COBAS®) and dried blood spots (DBS; Abbott m2000), both employing automated RNA extraction, purification, amplification, and detection. Roche’s 1 mL plasma protocol and Abbott’s open-mode DBS protocol (for low-volume samples) were applied32. ART coverage was evaluated by analyzing samples from all PLWH participants for ARVs (efavirenz, lopinavir, and tenofovir) using LC-MS/MS. These drugs were chosen based on their common use in treatment regimens and extended half-lives, though broader ARV profiling was limited by cost32. Drug resistance mutations in the protease and reverse transcriptase genes were analyzed using Stanford University’s Genotypic Resistance Interpretation Algorithm or Calibrated Population Resistance Tool33. Transmitted resistance was assessed by comparing recently infected PLWH with long-term infection cases, with mutations detected in the pol gene via TaqMan® SNP Genotyping Assay (Applied Biosystems)32. Viral RNA extraction was performed using NucliSens easyMAG (bioMérieux), followed by sequencing of ~ 1.1-kilobase amplicons on an Applied Biosystems Genetic Analyzer33.
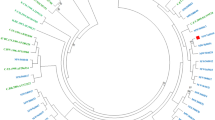
The study analyzed 43 samples (all 5 confirmed recent infections and 38 from long-term infections) for drug resistance and viral subtyping, with successful amplification achieved in 42 samples32. The HIV pol gene was amplified by a one-step reverse transcription polymerase chain reaction (RT-PCR), followed by nested PCR. Each sample was subtyped using the REGA HIV-1 & 2 Automated Subtyping Tool34,35.
Recent and long-term infections classification
Recent and long-term HIV infections were classified using two algorithms combining HIV-1 LAg avidity, viral load (VL), and antiretroviral (ARV) detection. Recent infections were defined by a median normalized optical density (ODn) ≤ 1.5 on the LAg avidity test (confirmed through triplicate testing) coupled with VL ≥ 1,000 copies/mL; cases meeting these criteria but with no detected ARVs were also classified as recent, reflecting active viral replication in untreated individuals. Long-term infections were identified through an initial LAg avidity ODn > 2.0; a median ODn > 1.5 upon repeat testing if the initial result was ≤ 2.0; a median ODn ≤ 1.5 with VL < 1,000 copies/mL (indicating viral suppression); or cases with median ODn ≤ 1.5 and VL ≥ 1,000 copies/mL but with detected ARVs (suggesting treatment failure or non-adherence rather than recent infection). This multi-parameter approach enhances diagnostic accuracy by distinguishing true recent infections from long-standing ones with similar biomarker profiles due to treatment effects or natural viral control.
Variable selection and category
The primary outcome variables in this study were HIV-1 prevalence and the presence of drug-resistant mutations. Independent variables, selected based on a review of existing literature, included demographic factors (age, sex, education level, marital status, region, and household wealth index), behavioral factors (alcohol and non-prescribed drug use, sexual history including age at first sex, condom use, number of sex partners in the last 12 months, relationship with the most recent partner, and condom use consistency in non-marital or non-cohabiting relationships; HIV testing history such as ever being tested and self-reported HIV status; and STI history), and laboratory variables (HIV-1 infection duration, viral load suppression, CD4 count, ART status [self-reported and ARV testing], viral load, ARV detection, duration of ART, time since ART initiation, and drug-resistant mutations). These variables were used to evaluate the health status of PLWH and/or ART effectiveness.
Operational definitions
Virological failure A confirmed HIV viral load of ≥ 1,000 copies/mL in PLWH who are on ART32.
Viral load suppression An HIV viral load of < 1,000 copies/mL in PLWH who are on ART, indicating effective ART response32.
Early sexual initiation (sexual debut) The initiation of sexual activity before the age of 18 years. This threshold was selected based on established literature linking early sexual initiation to increased HIV risk and its relevance to adolescent health policies in Ethiopia36,37,38.
Data management and analysis
All potential variables were selected based on existing literature. Data were recoded and analyzed using STATA version 17.0 (StataCorp, College Station, TX, USA). A complete case analysis approach was employed, excluding participants with missing values for any variables in the multivariable logistic regression model to ensure the reliability of estimates and the validity of statistical inferences. The statistical analysis also accounted for the complex survey design by incorporating weighting adjustments to address the sampling strategy and non-response. Descriptive statistics were used to present means, frequencies, and proportions. The bivariable logistic regression analyses were performed between each independent variable and the primary outcome (HIV-1 prevalence) to identify potential candidate variables for the multivariable model. Variables showing a suggestive association with HIV-1 prevalence (p < 0.25) in the bivariable analyses were considered for inclusion in the multivariable analysis to avoid prematurely excluding potentially important variables. In the final multivariable model, variables with p < 0.05 were considered statistically significant independent predictors of HIV-1 prevalence.
Ethics approval
The survey protocol was approved by the Institutional Review Boards (IRBs) of the Ethiopian Public Health Institute (EPHI), the Centers for Disease Control and Prevention (CDC), and Columbia University. Written informed consent was obtained from emancipated minors (aged 15–17) and adult participants (aged 18–64) for both interview and biomarker components. The IRBs waived the requirement for parental/guardian consent for emancipated minors. For non-emancipated minors (aged 15–17), written consent was obtained from a parent or guardian, accompanied by the participant’s assent. All methods were performed in accordance with the relevant guidelines and regulations.
Results
Demographic, socioeconomic, and behavioral characteristics of study participants
The study analyzed data from 19,136 participants, the majority of whom were female (60.6%). The mean participant age was 29.95 ± 11.92 years (mean ± standard deviation). The age distribution indicated a predominantly young adult population, with 39.4% aged 15–24 and 29.6% aged 25–34 years. Regarding educational attainment, 35.7% of participants had completed primary school, while 28.8% had completed secondary school. In terms of sexual behavior, most participants (65.7%) initiated sexual activity at age 18 or older. Furthermore, 16.2% reported engaging in non-marital or non-cohabiting sexual relationships in the 12 months preceding the survey. Among this subgroup, only 34.9% reported using condoms (Table 1).
HIV-1 prevalence and distribution
The overall HIV-1 prevalence was 3.0% (614/18,522; 95% CI: 2.6–3.5). Among PLWH, 15.3% (88/614; 95% CI: 12.2–18.9) reported being HIV-negative, while 10.5% (59/614; 95% CI: 7.9–13.7) were unaware of their status. Prevalence was higher among females (4.1%) than males (2.0%). Age-stratified analysis revealed an inverted U-shaped trend in HIV-1 prevalence, with rates increasing with age, peaking at 6.2% among individuals aged 35–44 years, and subsequently declining to 3.4% in the 55–64 age group. The prevalence decreased with higher educational levels, ranging from 5.2% (no formal education) to 1.0% (post-secondary education). Similarly, HIV-1 prevalence was higher among those who initiated sex before age 18 (5.1%) compared to those who began at 18 or older (3.2%). Additionally, participants reporting consistent condom use or abstinence in the 12 months before the survey had a higher prevalence (4.9%), while those with inconsistent use (2.0%) or no condom use (2.2%) showed lower rates.
HIV-1 infection duration, ART adherence, and treatment outcomes
Among the study participants, 0.5% (95% CI: 0.2–1.5) had acquired HIV-1 recent infections. Of those receiving ART, 12.4% (95% CI: 9.4–16.3) showed virological failure. Regarding immune status, 35.8% (221/614; 95% CI: 31.7–40.1) had CD4 T-cell counts below 350 cells/mm³. Approximately three-fourths (76.7%; 95% CI: 72.3– 80.7) of participants were confirmed to be on ART, based on a combination of blood testing and self-reported data. Blood tests for specific ARVs revealed that 46.7% (95% CI: 41.8–51.6) tested positive for Efavirenz, 0.2% (95% CI: 0.0–1.6) for Lopinavir, and 26.0% (95% CI: 21.7– 30.7) for Nevirapine (Table 2).
HIV-1 drug resistance mutations and genotyping outcomes
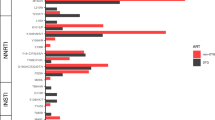
All successfully genotyped samples (100%, 42/42) were subtype C. The most prevalent resistance mutation was K103N, observed in 45.2% of samples (19/42), followed by V106M in 21.4% (9/42) for NNRTIs. For NRTIs, M184V was the most frequent mutation at 57.1% (24/42), followed by K65R at 21.4% (9/42) (Supp. Table 1). In most samples, 78.8% (33/42) showed resistance to at least one NNRTI, while 66.7% (28/42) showed mutations impacting at least one NRTI. Mutations conferring resistance to protease inhibitors (PIs) were found in only one sample. Furthermore, co-occurring resistance mutations for NNRTI and NRTI classes were present in 64.3% (27/42) of samples. Co-occurrence of mutations conferring resistance to all three ARV classes was infrequent, observed in only one individual (Fig. 2).
Factors associated with HIV-1 prevalence
The study identified multiple factors associated with HIV-1 prevalence. Females had significantly higher odds of HIV-1 infection than males (AOR: 1.7, 95% CI: 1.26–2.26). Regarding age, participants aged 35–44 and 45–54 years had the highest odds ratios (AOR: 6.7, 95% CI: 4.02–10.54 and AOR: 6.7, 95% CI: 3.88–11.52, respectively) compared to those aged 15–24 years. The study also found that education level significantly influenced HIV-1 prevalence; participants with primary education (AOR: 2.5, 95% CI: 1.64–3.71) and secondary school (AOR: 2.2, 95% CI: 1.43 − 3.36) had a higher likelihood of infection than those with post-secondary education. Marital status was another determinant; previously married individuals had significantly higher odds of infection (AOR: 2.7, 95% CI: 1.47–4.95) compared to those never married. Regional variation was also evident; residents of Gambella had significantly higher odds of infection (AOR: 2.8, 95% CI: 1.52–5.31) than those in Tigray (the reference region). Participants whose last partner was neither marital nor cohabiting had increased odds of HIV-1 infection (AOR: 1.7, 95% CI: 1.08– 2.67) compared to those in marital or cohabiting relationships. Additionally, individuals with a history of STIs had the highest odds ratio (AOR: 3.3, 95% CI: 1.85– 5.78) compared to those without such a history (Table 3).
Discussion
Despite significant progress in prevention and treatment programs in Ethiopia, HIV remains a major public health challenge. In this study, the overall HIV-1 prevalence was found to be 3% (95% CI: 2.6–3.5), which aligns with the national estimates (2.9–6.0%)1,7 and falls within the range reported in Zambia (4.2–23.5%)39. However, this result is much lower than the prevalence among female sex workers in Ethiopia, which ranges from 18.5% nationally40 to 19.9% in Hawassa41 and 28.2% in Bahir Dar42. This variation may stem from the heightened vulnerability of female sex workers due to factors such as inconsistent condom use40, stigma, discrimination43, and limited access to healthcare services40,43,44.
Cross-country comparisons further highlight regional variations. For instance, HIV prevalence in Lesotho (25.6%)45 and South Africa (25.9–28.0%)46 far exceeds Ethiopia’s rates. These differences likely reflect variations in health system infrastructure, socioeconomic conditions, and cultural contexts that influence sexual behavior and HIV risk.
HIV-1 prevalence was significantly higher among women (4.1%) than among men (2.0%), representing a more than twofold difference. Several factors may contribute to this difference. Biologically, women are more susceptible to HIV acquisition during heterosexual intercourse due to anatomical differences that facilitate male-to-female transmission. Additionally, gender inequalities and cultural norms often limit women’s ability to negotiate safe sex practices, while financial dependence may further heighten vulnerability, potentially leading to engagement in transactional sex. However, this prevalence is lower than the 24.1% reported in South Africa47, likely reflecting broad differences between the two countries. These include variations in national HIV program effectiveness, socioeconomic conditions, health system infrastructures, and cultural norms governing sexual behavior and HIV risk. The HIV-1 incidence rate in this study was 0.5%, lower than the 1.5% reported in Lesotho45, suggesting that new infections are being effectively prevented in the current study’s context. Similarly, age-stratified analysis showed that HIV-1 prevalence peaked among adults aged 35–44 (6.2%) and 45–54 (6.1%). This result likely reflects cumulative exposure risk over time, as older individuals may have had more sexual partners or prolonged periods of untreated infection. Delayed diagnosis due to stigma and discrimination could further exacerbate transmission in these age groups, as fear of social repercussions may deter testing and treatment-seeking.
The study found that 12.4% of PLWH on ART experienced virological failure, indicating persistent challenges in treatment adherence and effectiveness. This finding is consistent with studies from Ethiopia, which reported virological failure rates of 15.9% and 15.95%48,49. However, our result is higher than those reported in other African regions (7.3–9%)50,51, suggesting potential regional variations in treatment outcomes. Even higher rates of virological failure have been documented in Ethiopia (18.0%)52 and Asia (17.9%)53, indicating that suboptimal viral suppression remains a significant concern in multiple high-burden settings.
In the current study, a significant discrepancy was observed between self-reported HIV-1 status and laboratory-confirmed results, with 15.3% of PLWH incorrectly reported as negative status. This mismatch might be due to gaps in service delivery, including inadequate patient education, poor linkage to care, or insufficient counseling during HIV testing. It also highlights critical deficiencies in HIV-1 status awareness, which may hinder treatment initiation and retention in care.
Though Ethiopia has made significant progress toward the UNAIDS 90-90-90 targets, with 97.1% of diagnosed HIV patients receiving ART and 87.6% achieving viral suppression54, the emerging drug resistance poses a critical challenge, particularly in patients requiring second-line therapy. Our analysis of 42 HIV-positive samples revealed a high prevalence of DRMs, with NNRTIs and NRTIs being the most affected. NNRTI resistance was prevalent, observed in 76.7% of samples, with K103N (44.2%) and V106M (23.3%) being the most frequent mutations. The high prevalence of K103N is concerning, as it confers resistance to key first-line NNRTIs, including efavirenz and nevirapine, potentially undermining treatment efficacy. Similarly, NRTI resistance was detected in 65.1% of samples, with M184V (55.8%) and K65R (20.9%) being the predominant mutations. While M184V is associated with resistance to lamivudine and emtricitabine, it also increases susceptibility to tenofovir and zidovudine, which can be leveraged in regimen optimization55. The NADIA trial further supports the efficacy of tenofovir-containing regimens even in the presence of M184V, demonstrating high virological suppression rates when combined with dolutegravir56. These findings suggest that a substantial proportion of individuals in the study population may be experiencing treatment failure due to ineffective drug regimens, necessitating a transition to second-line therapies. On the other hand, resistance to PIs was low, with only a single (M46I) mutation identified. This indicates that PIs remain a viable and effective alternative for patients who resist NNRTIs and NRTIs. Given the high resistance rates to first-line regimens, enhanced resistance monitoring, optimized ART regimens, and timely switches to second-line therapies are essential to maintaining treatment success and preventing further transmission of resistant strains.
The current study identified multiple factors associated with HIV-1 prevalence in Ethiopia. Female participants were 1.7 times more likely to be HIV-1 positive than male participants. This finding is supported by a previous study from Ethiopia57. The variation may be due to gender inequality that significantly contributes to higher rates of HIV infection among women57. In terms of age, participants aged 35–44 and 45–54 years each 6.7 times more likely to be HIV-1 positive compared to those aged 15–24 years. A similar pattern was observed in China, where older age groups faced an increased HIV risk58. This difference could reflect factors such as prolonged exposure to risk behaviors, lower testing rates, or delayed diagnosis in older populations. The study also found that participants with primary or secondary school education were 2.5 and 2.2 times more likely, respectively, to have HIV-1 compared to participants with post-secondary education. This suggests that higher education may protect against HIV infection. Consistent with this, studies in Africa have also linked higher education levels to lower HIV prevalence59. Regarding marital status, participants who were previously married (divorced, separated, or widowed) were 2.7 times more likely to contract HIV-1, while currently married/cohabiting participants were 1.5 times more likely, respectively, compared to never-married individuals. This aligns with a Nigerian study where previously married individuals had60. Their vulnerability may stem from disrupted social networks, economic instability, or engagement in transactional sex, as seen in Malawi and Uganda61,62. Geographically, residents of the Gambella region were 2.8 times more likely to be HIV-1 positive than those in Tigray. This may reflect inadequate government focus on HIV prevention, including the discontinuation of community programs, which reduced adherence to prevention63. Additionally, individuals with an STI history had a 3.3-fold higher likelihood of HIV-1 infection. STIs, especially those causing genital ulcers, increase susceptibility by damaging mucosal barriers. For example, syphilis, chlamydia, or gonorrhea elevate risk due to inflammation-driven immune responses64.
Conclusions
This study identified a high prevalence of HIV-1 in Ethiopia. The findings showed major challenges to treatment effectiveness, including a high virological failure rate, a significant proportion of people living with HIV being unaware of or misreporting their status, and widespread drug resistance to common first-line NNRTI and NRTI antiretrovirals. Multiple factors were associated with HIV-1 prevalence, including sex, age, education level, marital status, region, and history of STIs. To improve treatment outcomes and curb transmission, Ethiopia should strengthen HIV prevention strategies focused on vulnerable populations, enhance testing and linkage to care, and implement routine drug resistance monitoring.
Data availability
The datasets used or analyzed for this study are available and can be accessed with the necessary access credentials from the PHIA program (https://phia-data.icap.columbia.edu/datasets?country_id=12).
References
Adal, M. Systematic review on HIV situation in addis Ababa, Ethiopia. BMC Public. Health. 19, 1544 (2019).
Mitiku, A. A., Dimore, A. L. & Gach, W. Trends of HIV infection and progress towards the 95-95-95 targets in Gambella regional state from 2019 to 2023, Southwest Ethiopia. HIV AIDS (Auckl). 16, 193–201 (2024).
Msukwa, M. et al. Characterising persons diagnosed with HIV as either recent or long-term using a cross-sectional analysis of recent infection surveillance data collected in Malawi from September 2019 to March 2020. BMJ Open. 12. (2022).
Mohloanyane, T., Olivier, D., Labhardt, N. & Amstutz, A. Recent HIV infections among newly diagnosed individuals living with HIV in rural Lesotho: secondary data from the VIBRA cluster-randomized trial. PLOS ONE 17. (2022).
Gedefie, A. et al. Prevalence and determinants of HIV among reproductive-age women (15–49 years) in Africa from 2010 to 2019: a multilevel analysis of demographic and health survey data. Front. Public. Health. 12, 1376235 (2024).
Nure, M. E., Weldegebreal, F., Tebeje, F., Sime, A. & Regassa, L. D. Prevalence and associated factors of recent HIV-1 infection among newly identified HIV-positive individuals tested with the Asante rapid recency assay in Harari region, Eastern Ethiopia. Front. Epidemiol. 5, 1443148 (2025).
Kibret, G. D. et al. Trends and Spatial distributions of HIV prevalence in Ethiopia. Infect. Dis. Poverty. 8, 90 (2019).
Vandepitte, J. et al. HIV and other sexually transmitted infections in a cohort of women involved in high-risk sexual behavior in Kampala, Uganda. Sex. Transm. Dis. 38, 316–323 (2011).
Reepalu, A. et al. Drug resistance in HIV-Positive adults during the initial year of antiretroviral treatment at Ethiopian health centers. Open. Forum Infect. Dis. 8. (2021).
Arimide, D. A. et al. High level of HIV drug resistance and virologic nonsuppression among female sex workers in Ethiopia: A nationwide Cross-Sectional study. JAIDS J. Acquir. Immune Defic. Syndr. 89. (2022).
Coetzee, J. et al. HIV-1 viraemia and drug resistance amongst female sex workers in Soweto, South Africa: A cross sectional study. PLOS ONE. 12, e0188606 (2017).
Ndahimana, J. A. et al. Drug resistance mutations after the first 12 months on antiretroviral therapy and determinants of virological failure in Rwanda. Tropical Med. Int. Health. 21, 928–935 (2016).
Namale, G. et al. Sustained virological response and drug resistance among female sex workers living with HIV on antiretroviral therapy in Kampala, Uganda: A cross-sectional study. Sex. Transm. Infect. 95, 405–411 (2019).
Tufa, T. B. et al. Characterization of HIV-1 drug resistance among patients with failure of second‐line combined antiretroviral therapy in central Ethiopia. HIV Med. 23, 159–168 (2022).
Kiros, M. et al. Increased HIV-1 pretreatment drug resistance with consistent clade homogeneity among ART-naive HIV-1 infected individuals in Ethiopia. Retrovirology 17. (2020).
Tadesse, B. T. et al. High Levels of Dual-Class Drug Resistance in HIV-Infected Children Failing First-Line Antiretroviral Therapy in Southern Ethiopia. In Viruses, vol. 10; (2018).
Steegen, K. et al. HIV-1 antiretroviral drug resistance patterns in patients failing NNRTI-based treatment: Results from a National survey in South Africa. J. Antimicrob. Chemother. 72, 210–219 (2016).
Gupta, R., Hill, A., Sawyer, A. W. & Pillay, D. Emergence of drug resistance in HIV type 1-Infected patients after receipt of First-Line highly active antiretroviral therapy: A systematic review of clinical trials. Clin. Infect. Dis. 47, 712–722 (2008).
Foka, F. E. T. & Mufhandu, H. Current arts, virologic failure, and implications for AIDS management: A systematic review. Viruses 15. (2023).
Gulick, R. & Glesby, M. Managing HIV treatment failure: Time to REVAMP? Ann. Intern. Med. 174, 1753–1754 (2021).
Wondmeneh, T. G. & Wondmeneh, R. G. Risky Sexual Behaviour among HIV-Infected Adults in Sub‐Saharan Africa: A Systematic Review and Meta‐Analysis. BioMed Research International 2023:6698384. (2023).
Yilema, S. A., Shiferaw, Y. A., Belay, A. T. & Belay, D. B. Mapping the Spatial disparities of HIV prevalence in Ethiopian zones using the generalized additive model. Sci. Rep. 14, 6215 (2024).
Phiri, M., Lemba, M., Chomba, C. & Kanyamuna, V. Examining differentials in HIV transmission risk behaviour and its associated factors among men in Southern African countries. Humanit. Social Sci. Commun. 9, 295 (2022).
Mulugeta, S. S. & Wassihun, S. G. Prevalence of HIV/AIDS infection among sexually active women in Ethiopia: Further analysis of 2016 EDHS. AIDS Research and Treatment 2022:8971654. (2022).
Damtie, Y. et al. HIV risk behavior and associated factors among people living with HIV/AIDS in Ethiopia: A systematic review and meta-analysis. PLOS ONE. 17, e0269304 (2022).
Corneil, T. A. et al. Unstable housing, associated risk behaviour, and increased risk for HIV infection among injection drug users. Health Place. 12, 79–85 (2006).
Doyle, K. E. et al. High prevalence of disability and HIV risk among low socioeconomic status urban adults, 17 U.S. Cities. Disabil. Health J. 13, 100834 (2020).
Negesse, Y., Mankelkl, G., Setegn, M. & Fetene, G. Multilevel analysis of factors associated with HIV among women of reproductive age (15–49 years old) in Ethiopia: Bayesian approach. Women’s Health. 17, 17455065211067638 (2021).
Lakew, Y., Benedict, S. & Haile, D. Social determinants of HIV infection, hotspot areas and subpopulation groups in Ethiopia: evidence from the National demographic and health survey in 2011. BMJ Open. 5, e008669 (2015).
Brawner, B. M. et al. A systematic review of Neighborhood-Level influences on HIV vulnerability. AIDS Behav. 26, 874–934 (2022).
Gelibo, T. et al. Spatial distribution and determinants of HIV prevalence among adults in urban Ethiopia: findings from the Ethiopia Population-based HIV impact assessment survey (2017–2018). PLOS ONE. 17, e0271221 (2022).
Ethiopian Public Health Institute (EPHI). Ethiopia Population-based HIV Impact Assessment (EPHIA) 2017–2018: Final Report. Addis Ababa: EPHI. (2020). Available from https://phia.icap.columbia.edu/ethiopia-final-report/
Zhou, Z. et al. Optimization of a low cost and broadly sensitive genotyping assay for HIV-1 drug resistance surveillance and monitoring in resource-limited settings. PLOS ONE. 6, e28184 (2011).
Yang, C. et al. Development and application of a broadly sensitive dried-bloodspot-based genotyping assay for global surveillance of HIV-1 drug resistance. J. Clin. Microbiol. 48, 3158–3164 (2010).
Alcantara, L. C. et al. A standardized framework for accurate, high-throughput genotyping of Recombinant and non-recombinant viral sequences. Nucleic Acids Res. 37, W634–642 (2009).
Diress, G., Adane, S., Linger, M., Merchaw, A. & Mengist, B. Individual and community-level determinates of risky sexual behaviors among sexually active unmarried men: A multilevel analysis of 2016 Ethiopian demographic and health survey. PLOS ONE. 17, e0270083 (2022).
Baru, A., Adeoye, I. A. & Adekunle, A. O. Risky sexual behavior and associated factors among sexually-active unmarried young female internal migrants working in Burayu town, Ethiopia. PLOS ONE. 15, e0240695 (2020).
He, J. et al. Association between early sexual debut and new HIV infections among adolescents and young adults in 11 African countries. AIDS Behav. 28, 2444–2453 (2024).
Mweemba, C., Hangoma, P., Fwemba, I., Mutale, W. & Masiye, F. Estimating district HIV prevalence in Zambia using small-area Estimation methods (SAE). Popul. Health Metrics. 20, 8 (2022).
Rameto, M. A. et al. Prevalence and factors associated with inconsistent condom use among female sex workers in Ethiopia: findings from the National biobehavioral survey, 2020. BMC Public. Health. 23, 2407 (2023).
Alemu, G. H., Gidebo, D. D. & Ali, M. M. Magnitude of HIV infection and associated factors among female sex workers at Hawassa, Ethiopia. Ethiop. J. Health Sci. 32, 261–268 (2022).
Abdella, S., Demissie, M., Worku, A., Dheresa, M. & Berhane, Y. HIV prevalence and associated factors among female sex workers in Ethiopia, East Africa: A cross-sectional study using a respondent-driven sampling technique. eClinicalMedicine 51. (2022).
Gebreyesus, S. H. & Mariam, D. H. Assessment of HIV/AIDS related risks among men having sex with men (MSM) in addis Ababa, Ethiopia. J. Public. Health Policy. 30, 269–279 (2009).
Arimide, D. A. et al. High level of HIV drug resistance and virologic nonsuppression among female sex workers in Ethiopia: A nationwide Cross-Sectional study. J. Acquir. Immune Defic. Syndr. 89, 566–574 (2022).
Schwitters, A. et al. High HIV prevalence and associated factors in Lesotho: results from a population-based survey. PLOS ONE. 17, e0271431 (2022).
Kharsany, A. B. M. et al. Community-based HIV prevalence in KwaZulu-Natal, South Africa: Results of a cross-sectional household survey. Lancet HIV. 5, e427–e437 (2018).
Mabaso, M. et al. HIV prevalence in South Africa through gender and Racial lenses: Results from the 2012 population-based National household survey. Int. J. Equity Health. 18, 1–11 (2019).
Fentie Wendie, T. & Workneh, B. D. Prevalence and predictors of virological failure among adults living with HIV in South Wollo zone, Northeast Ethiopia: A retrospective cohort study. HIV/AIDS - Res. Palliat. Care. 12, 393–402 (2020).
Aytenew, T. M. et al. Virological failure and associated factors among patients receiving anti-retroviral therapy in Ethiopia: A systematic review and meta-analysis. BMJ Open. 14, e087569 (2024).
Agegnehu, C. D., Techane, M. A., Mersha, A. T. & Atalell, K. A. Burden and associated factors of virological failure among people living with HIV in Sub-Saharan Africa: A systematic review and Meta-Analysis. AIDS Behav. 26, 3327–3336 (2022).
Kiweewa, F. et al. HIV virologic failure and its predictors among HIV-infected adults on antiretroviral therapy in the African cohort study. PLOS ONE. 14, e0211344 (2019).
Melku, M. et al. Immunological status and virological suppression among HIV-infected adults on highly active antiretroviral therapy. Environ. Health Prev. Med. 25, 43 (2020).
Mu, W. et al. Early and late virologic failure after virologic suppression in HIV-Infected Asian children and adolescents. J. Acquir. Immune Defic. Syndr. 80, 308–315 (2019).
Bayisa, L., Keno, T. S., Ayana, G. E., Abera, T. & Akuma, A. O. Out-of-Catchment area antiretroviral therapy initiation and its factors among HIV positive people on lifelong therapy in Western Ethiopia: implementation of decentralized ART services. J. Int. Assoc. Provid AIDS Care. 24, 23259582241307467 (2025).
Puertas, M. C. et al. Impact of intensification with raltegravir on HIV-1-infected individuals receiving monotherapy with boosted PIs. J. Antimicrob. Chemother. 73, 1940–1948 (2018).
Paton, N. I. et al. Efficacy and safety of dolutegravir or Darunavir in combination with lamivudine plus either Zidovudine or Tenofovir for second-line treatment of HIV infection (NADIA): week 96 results from a prospective, multicentre, open-label, factorial, randomised, non-inferiority trial. Lancet HIV. 9, e381–e393 (2022).
Girum, T. et al. Gender disparity in epidemiological trend of HIV/AIDS infection and treatment in Ethiopia. Archives Public. Health. 76, 51 (2018).
Chen, C. et al. Comparison of healthspan-related indicators between adults with and without HIV infection aged 18–59 in the united States: a secondary analysis of NAHNES 1999-March 2020. BMC Public. Health. 23, 814 (2023).
Hargreaves, J. R., Davey, C., Fearon, E., Hensen, B. & Krishnaratne, S. Trends in socioeconomic inequalities in HIV prevalence among young people in seven countries in Eastern and Southern Africa. PLOS ONE. 10, e0121775 (2015).
Fagbamigbe, A. F., Adebayo, S. B. & Idemudia, E. Marital status and HIV prevalence among women in Nigeria: ingredients for evidence-based programming. Int. J. Infect. Dis. 48, 57–63 (2016).
Fedor, T. M., Kohler, H. P. & Behrman, J. R. The impact of married individuals learning HIV status in Malawi: divorce, number of sexual partners, and condom use with spouses. Demography 52, 259–280 (2015).
Matovu, J. K., Todd, J., Wanyenze, R. K., Wabwire-Mangen, F. & Serwadda, D. Correlates of previous couples’ HIV counseling and testing uptake among married individuals in three HIV prevalence strata in Rakai, Uganda. Glob Health Action. 8, 27935 (2015).
Qanche, Q. et al. Factors contributing to high HIV prevalence in Majang zone, Southwest Ethiopia: what Lies beneath the tip of the iceberg?? J. Multidiscip Healthc. 14, 3273–3283 (2021).
Wall, K. M. et al. Etiologies of genital inflammation and ulceration in symptomatic Rwandan men and women responding to radio promotions of free screening and treatment services. PLOS ONE. 16, e0250044 (2021).
Acknowledgements
The authors gratefully acknowledge the Population-based HIV Impact Assessment (PHIA) program managers for generously providing the data utilized in this research.
Funding
No financial support was received for this study.
Author information
Authors and Affiliations
Contributions
All authors participated in formulating the research question(s) and designing the study. TD contributed to the data analysis and interpreting the results, to writing the original draft, and to reviewing and editing. MG contributed to the supervision of the project, interpretation of findings, and to reviewing and editing. AB and AW contributed to the interpretation of the findings and to reviewing and editing. All authors read and approved the final version of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Deress, T., Worede, A., Birhanu, A. et al. HIV-1 prevalence, drug resistance, and associated factors in the urban Ethiopian population. Sci Rep 15, 17216 (2025). https://doi.org/10.1038/s41598-025-02122-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-02122-8