Abstract
Melanoma remains one of the leading causes of cancer-related mortality worldwide, necessitating advanced imaging techniques for early and accurate detection. This study assesses the dosimetry, safety, and imaging performance of a novel 68Ga-labeled α-melanocyte-stimulating hormone ([68Ga]Ga-αMSH) derivative for targeting melanocortin 1 receptors (MC1Rs) in metastatic melanoma. In this first-in-human, prospective, open-label clinical trial, 11 patients with histologically confirmed metastatic melanoma underwent whole-body PET/CT imaging following intravenous administration of the radiolabeled compound (150 ± 10 MBq). Tumor uptake, biodistribution, pharmacokinetics, and radiation dosimetry were evaluated at 60 and 120 min post-injection. Organ and tumor uptake values were measured as standardized uptake values. Radiation dose estimates were calculated using the MIRD methodology and S-values obtained from OLINDA/EXM software. Safety evaluations included monitoring adverse events, biochemical parameters, and vital signs. The radiopharmaceutical demonstrated rapid and selective uptake in metastatic melanoma lesions, achieving high tumor-to-background contrast within 60 min. Quantitative analysis showed substantial tumor uptake, with sustained activity at 120 min. High tumor-to-blood and tumor-to-muscle ratios ensured excellent lesion detectability. The kidneys exhibited the highest absorbed dose (0.0948 ± 0.0425 mSv/MBq), attributed to renal excretion, whereas the brain received the lowest dose (0.0012 ± 0.0007 mSv/MBq). Comparisons with [18F]FDG and other tracers demonstrated superior dosimetry profiles, minimizing radiation exposure and enabling repeat imaging. Also, safety monitoring revealed no serious adverse events. [68Ga]Ga-αMSH analogue exhibits excellent imaging properties, favorable pharmacokinetics, and a strong safety profile, supporting its clinical utility for PET imaging of metastatic melanoma. Its high tumor specificity and minimal off-target accumulation address limitations associated with [18F]FDG.
Similar content being viewed by others
Introduction
Melanoma is an aggressive form of skin cancer with a high global mortality rate1,2. Although the incidence of some cancers has decreased in recent years, melanoma, which is among the top 10 cancers, has shown an increasing incidence in recent years3. In the five stages of melanoma, the 0 stage is melanoma without metastases, and the IV stage is spread melanoma in the body that moves to other regions, including the brain, liver, lymph nodes, lungs, and bones4. Therefore, timely and accurate detection using advanced imaging modalities, such as positron emission tomography (PET) can effectively control the death rate worldwide2,5.
PET accurately identifies clusters of tumor cells that spread and metastasize to various organs or tissues. While [18F]FDG PET/CT remains the standard imaging modality for melanoma staging and monitoring, due to its ability to detect metabolically active tumors6, it has several notable limitations. FDG uptake is non-specific and can occur in inflamed or infected tissues, potentially leading to false-positive results and unnecessary interventions7. Furthermore, FDG’s diagnostic accuracy can be compromised in diabetic patients or those with elevated blood glucose levels8. The technique also shows limited specificity in detecting early locoregional lymph node metastases, making it less reliable in initial staging9,10. Additionally, a phenomenon known as pseudoprogression, frequently observed in patients receiving immune checkpoint inhibitors, may cause increased FDG uptake due to immune cell infiltration rather than true tumor growth. These limitations highlight the need for more specific radiotracers that reduce off-target uptake and improve lesion detectability, particularly in complex clinical scenarios. In this context, radiopharmaceuticals targeting tumor-specific biomarkers such as melanocortin-1 receptors (MC1Rs) offer a promising alternative11.
The physical characteristics of Gallium-68 (68Ga), including its short half-life of 67.71 min and high positron emission yield of 88%, make it an ideal radionuclide for PET imaging, offering high-quality images with minimal radiation burden to patients12. 68Ga is conveniently available from a Germanium-68/Gallium-68 (68Ge/68Ga) generator, which is easily accessible and relatively inexpensive13. The recent development of easily labeled small peptides with 68Ga has unequivocally enabled the production of radiopharmaceuticals for PET imaging. Moreover, 68Ga is increasingly used in combination with lutetium-177 (177Lu) as part of theranostic approaches, enabling both diagnostic and therapeutic applications14,15.
Alpha-melanocyte-stimulating hormone (α-MSH) is a known peptide hormone that regulates melanogenesis and binds to the melanocortin 1 receptor (MC1R) on melanocytes. α-MSH influences skin pigmentation by stimulating the production of melanin and is available in linear and cyclic analog forms16. However, several α-MSH motifs, including CCZ01047, CCZ01056, and CCZ0104817 have been developed and preclinically evaluated. Among these, CCZ01048, with the sequence of His-Phe-Arg-Trp and DOTA-4-amino-(1-carboxymethyl) piperidine (Pip)-Nle-CycMSHhex,18, has shown promising results in labeling with 68Ga and 177Lu19,20, including high stability, great binding affinity21, and high tumor uptake characteristics22.
Dosimetry plays a pivotal role in both diagnostic and therapeutic nuclear medicine, particularly in first-in-human and early-phase clinical studies, where it provides a quantitative assessment of the radiation absorbed by the patient. Dosimetry evaluations have been mandated by regulatory authorities such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) to document radiation exposure to critical organs and the whole body during the approval process for new radiopharmaceuticals. Furthermore, compliance with the International Commission on Radiological Protection (ICRP) and the Medical Internal Radiation Dosimetry (MIRD) standards makes this measurement beneficial, accurate, and consistent23.
Given the limited availability of effective radiopharmaceuticals for the diagnosis of metastatic melanoma, the development of novel targeted agents remains a critical need in nuclear medicine. In this context, proper α-MSH analogues labeled with 68Ga using PET modalities may have unique properties in the imaging metastatic melanoma.
The [68Ga]Ga-CCZ01048 radiotracer introduced in this study represents a promising strategy for imaging metastatic melanoma through selective targeting MC1Rs, which are overexpressed in melanoma cells. In contrast to conventional PET tracers such as [18F]FDG, this tracer enhances tumor detection while minimizing non-specific uptake in healthy or inflamed tissues. Structurally, it is a modified α-MSH analogue with DOTA-based modifications which improves receptor-binding affinity, metabolic stability, and clearance from non-target tissues. The DOTA moiety ensures strong radiochemical stability and efficient chelation of 68Ga (Fig. 1), making the tracer highly stable and suitable for clinical use.
To the best of our knowledge, 68Ga-labeled α-melanocyte-stimulating hormone (αMSH) analogues have rarely been evaluated in clinical studies. One prior first-in-human study reported by Yang et al.24 demonstrated the feasibility of such compounds in melanoma imaging. However, no clinical study to date has evaluated [68Ga]Ga-CCZ01048 specifically, nor compared its performance with [18F]FDG in a head-to-head setting, as done in the present work. Furthermore, this study includes a direct comparison of the new radiopharmaceutical with the standard-of-care tracer [18F]FDG to evaluate its diagnostic performance. Specifically, [68Ga]Ga-CCZ01048, a modified α-MSH analogue designed for selective binding to MC1Rs, was evaluated. It is important to note that [68Ga]Ga-CCZ01048 and [68Ga]Ga-αMSH refer to the same radiopharmaceutical compound throughout this manuscript.
Methods
Study design
This prospective, first-in-human, open-label clinical study was designed to evaluate the dosimetry and safety of a novel PET radiopharmaceutical in patients with metastatic melanoma. The study was conducted between 2023 and 2024 in accordance with the Declaration of Helsinki and approved by the Research Ethics Committee of the Tehran University of Medical Science (Approval No.: IR.TUMS.IKHC.REC.1402.245). Written informed consent was obtained from all participants prior to enrollment.
Patients and eligibility criteria
A total of 11 patients (7 women and 4 men), with a mean age of 55 ± 12 years and mean weight of 69 ± 14 kg, were enrolled in this study. 5 patients were enrolled for initial staging, aimed at assessing the primary status of melanoma and determining the extent of disease spread throughout the body. This stage focused on identifying the disease stage and evaluating its dissemination in various organs. 6 patients were enrolled for restaging, having previously undergone different treatments. The purpose of the PET/CT scan in these patients was to reassess the disease status and evaluate its progression or response to treatment. All participants had histologically confirmed metastatic melanoma, were 18 years of age or older, and demonstrated adequate organ function at baseline. Exclusion criteria included overweight (> 225 kg), prior systemic therapy within four weeks, severe organ dysfunction, or any contraindication to PET imaging agents. All relevant medical records, including pathology reports and imaging data, were reviewed prior to enrollment.
Preparation and quality control of the radiopharmaceutical
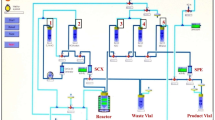
The CCZ01048 peptide was procured from MedChemExpress LLC (USA). The novel radiopharmaceutical, [68Ga]Ga-CCZ01048, was prepared via a chelation-based radiolabeling method with 68Ga obtained from an in-house developed 68Ge/68Ga generator in sterile conditions according to the previously published procedure22. Radiochemical purity (> 99%) was verified using high-performance liquid chromatography (HPLC) and instant thin-layer chromatography (ITLC). The final product was passed through a 0.22 µm Millipore filter (MilliporeSigma, USA) and used for injection.
Imaging study Protocol
Patients received intravenous (i.v.) administrations of [68Ga]Ga-CCZ01048 (150 ± 10 MBq) and [18F]FDG (303 ± 59 MBq) for comparative imaging. Whole-body PET/CT scans were performed at 60- and 120-min post-injection using a Discovery IQ PET/CT scanner (GE Healthcare, Waukesha, WI, USA) to assess tracer distribution and uptake. The exact acquisition time per bed position was 180 Sec. Finally, PET images were reconstructed using standard corrections for dead time, random events, attenuation (via low-dose CT), and scatters. The 60- and 120-min imaging time points were selected based on preclinical studies with the same compound, [68Ga]Ga-CCZ01048, which demonstrated sustained high tumor uptake and favorable tumor-to-background contrast at both time points18.
All detected lesions in each scan were described and compared. Additionally, SUVs were calculated for each lesion, specifically, SUVmax of each lesion was normalized to SUVmean of the liver, which served as the reference organ. This comparison enhanced the differential diagnostic capability and sensitivity of the novel radiotracer relative to [18F] FDG. Data analyses were conducted using SPSS software (version 25).
Dosimetry study
Dosimetry calculations were conducted to estimate radiation absorbed doses to organs based on time-activity curves (TACs) and time-integrated activity coefficients (TIACs). The SUVs were calculated after imaging in each patient using Eq. 1, from which the cumulated activity is determined, and the volume (ml) of the body obtained by references25,26,27:
Regions of interest (ROIs) were manually delineated over critical organs and tumor lesions. TIACs were derived from PET/CT imaging data and served as input for subsequent dosimetry modeling. The Cumulative activity (Ã) for each organ was calculated using the SUVmean, which offers greater reproducibility and minimizes variability compared to SUVmax-based approaches. As illustrated in Fig. 2, cumulative activity was computed using Eq. (2)28,29, based on a mathematical extrapolation of the TACs. The procedure involved the following steps: (1) extraction of SUVmean values from defined ROIs, (2) conversion of SUV to MBq/mL using organ volume and patient weight, and (3) integration of the TACs over time to estimate total activity accumulation in each organ.
where A(t) represents the measured activity in each organ over time.
Schematic representation of TACs used for the calculation of cumulative activity (Ã). Points A and B represent activity measurements derived from patient PET data for each source organ. At time zero, activity in all source organs (excluding blood) is assumed to be zero. From point B, the curve is extrapolated to infinity using a mono-exponential decay function with the physical decay constant of 68Ga. The shaded area under the curve corresponds to the cumulative activity for the respective organ.
To estimate the cumulative activity for each source organ, the activity at time zero was assumed to be zero. Activity measurements at 60- and 120-min post-injection were connected using linear interpolation to form the initial segment of the time–activity curve. Subsequently, the activity at the 120-min point was extrapolated to infinity by fitting a mono-exponential decay function based on the physical decay constant of 68Ga30,31. The area under the resulting curves, from zero to infinity, was calculated and used to represent the cumulative activity (Ã) for each organ.
Subsequently, the absorbed dose is estimated using à and S-values according to the MIRD method (Eq. 3):
The S-values for 68Ga radionuclide had been pre-computed using the OLINDA/EXM software (version 2.1)32.
Endpoints and safety monitoring
The Primary endpoints of this study included the assessment of tumor uptake, pharmacokinetic profiles, and organ-specific radiation dose estimates following administration of [68Ga]Ga-CCZ01048. Secondary endpoints focused on evaluating the safety profile of the radiopharmaceutical, including the incidence of adverse events, and preliminary efficacy in lesion detection. All patients were monitored up for adverse events to 7 days post-injection, and routine biochemical analyses were performed to assess renal and hepatic function.
Statistical analysis
Descriptive statistics were employed to summarize and analyze the biodistribution, pharmacokinetics, dosimetry, and safety profiles of [68Ga]Ga-CCZ01048 in patients with metastatic melanoma. Specifically, the mean, standard deviation (SD), range, and median were calculated for key variables such as SUVs, organ-specific radiation dose estimates, and pharmacokinetic parameters. For comparative analysis, paired t-tests were conducted to assess differences in tracer uptake between tumor and non-tumor tissues, with statistical significance set as p < 0.05. For dosimetry calculations, organ-specific absorbed doses were derived from TACs and TIACs, using the MIRD formalism and S-values obtained via the OLINDA/EXM software.
Results
Preparation and quality control of the radiopharmaceutical
The radiolabeled compound was prepared under optimized conditions (pH = 3, temperature = 95 °C, time = 15 min, and peptide amount = 15 µg), achieving a RCP greater than 99%, as confirmed by analytical methods (Fig. 3).
Tumor uptake and imaging performance
In this study, the diagnostic confirmation of lesions was primarily based on PET/CT imaging findings. Since all patients had confirmed metastatic melanoma, imaging results were compared with clinical data and patient history. In patients who underwent biopsy, radiotracer uptake observed on PET/CT correlated well with histopathological findings. A high concordance was observed between the radiotracer uptake sites of [68Ga]Ga-αMSH and biopsy-confirmed lesions, indicating the high sensitivity of this radiotracer for detecting metastatic melanoma. To provide a comprehensive overview of the study cohort, Table 1 summarizes the demographic characteristics, TNM staging, and histopathological findings of all included patients.
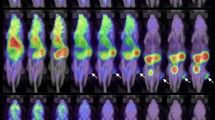
PET/CT images were acquired after injection of the [68Ga]Ga-CCZ01048 and [18F]FDG radiotracers. The novel PET radiopharmaceutical exhibited rapid and selective uptake in metastatic melanoma lesions, achieving high tumor-to-background contrast as early as 60 min post-injection (Figs. 4 and 5). The radiotracer demonstrated fast clearance from non-target tissues, with the majority of the radioactivity excreted via the urinary system within the first hour. Renal uptake was the most prominent among non-target organs, primarily due to physiological excretion rather than receptor-specific binding. These findings highlight the favorable pharmacokinetic profile of the tracer, characterized by minimal off-target accumulation and efficient systemic clearance. PET/CT imaging effectively visualized metastatic melanoma lesions, confirming the diagnostic potential of the radiotracer. The high-resolution imaging capability facilitated clear delineation of tumor margins and supported its utility for detecting lesions in complex anatomical regions.
A 48-year-old female with a history of rectal melanoma referred for restaging. (A, B) [68Ga]Ga-CCZ01048 PET/CT images; (C, D) [18F]FDG PET/CT images. The patient was diagnosed with mucosal melanoma of the perineal region, with evidence of local recurrence and extensive metastatic spread. PET/CT revealed two large tumor masses in the perineal and perirectal regions extending to the mesorectum and perisacral areas. Additionally, at least six pulmonary nodules were identified in the right lung, indicative of pulmonary metastases. Multiple mesorectal lymph nodes were also involved, suggesting lymphatic dissemination of the disease.
PET/CT images of a 63-year-old female patient with histologically confirmed stage IV metastatic melanoma. The patient was referred for evaluation of suspected disease progression. (A) Image acquired at 60 min post-injection of [68Ga]Ga-CCZ01048; (B) image acquired at 120 min post-injection. The scan revealed widespread metastatic involvement, including the brain, lungs, stomach, lymph nodes, and subcutaneous tissues. Notable findings include a hypermetabolic lesion near the fourth ventricle, multiple pulmonary metastases, focal gastric wall thickening along the greater curvature, extensive lymphadenopathy in the left iliac chain, and tumor nodules in the subcutaneous fat of the foot.
A comparative imaging analysis between [68Ga]Ga-αMSH and [18F]FDG demonstrated that [68Ga]Ga-αMSH exhibited significantly higher uptake in metastatic melanoma lesions, while [18F]FDG showed uptake in both malignant and non-specific metabolically active tissues. Table 2 summarizes the mean SUVmax values across different organs, as well as the tumor-to-liver SUVmean ratios obtained 60 min post-injection of each radiotracer. On average, the SUVmax of metastatic lesions was higher with [68Ga]Ga-αMSH than with [18F]FDG, reflecting the superior target specificity of [68Ga]Ga-αMSH for MC1R-expressing tumors. In contrast, the relatively non-specific accumulation of [18F]FDG in inflamed and normal tissues often led to reduced lesion contrast and potential false-positive findings. These findings underscore the potential of [68Ga]Ga-αMSH as a more selective and reliable PET agent for the detection of metastatic melanoma. To assess the statistically significant difference of radiotracer uptake between tumor and non-tumor regions, statistical comparisons were conducted using the paired t-test. The results demonstrated that the uptake of [68Ga]Ga-αMSH in tumor regions was significantly higher than in the non-tumor areas. Specifically, the SUVmax lesion-to-SUVmean liver ratio was assessed as a differentiation index, and the differences were statistically significant at both 60- and 120-min post-injection (p < 0.005).
Radiation dose estimates and dosimetry
Table 3 summarizes the organ-specific absorbed radiation doses following the administration of [68Ga]Ga-CCZ01048. Dosimetry analysis revealed that the highest radiation burden occurred in the kidneys, consistent with the renal excretion pathway of the tracer. The estimated effective doses remained well within established diagnostic reference levels, supporting the feasibility of repeated imaging when clinically indicated. Additionally, the calculated cumulative activity values confirmed the tracer’s efficient clearance, thereby minimizing concerns regarding prolonged radiation exposure. Overall, these findings affirm the favorable dosimetric profile and clinical safety of [68Ga]Ga-CCZ01048 for use in serial PET imaging studies.
Safety and adverse events
The radiopharmaceutical was well tolerated, with no serious adverse events reported during the study. Comprehensive safety monitoring, including laboratory tests, vital sign assessments, and electrocardiographic evaluations, revealed no clinically significant abnormalities in any participant.
Mild and transient side effects, such as headache and nausea, were observed in a small subset of patients; however, these were considered unlikely to be related to the radiopharmaceutical administration. Overall, the favorable safety profile supports the feasibility of further clinical development and broader application of [68Ga]Ga-CCZ01048 in patients with metastatic melanoma.
Discussion
Melanoma is a highly aggressive malignancy and remains among the leading causes of cancer-related mortality worldwide. Early and accurate detection is critical for improving patient outcomes, particularly prior to the development of widespread metastases. PET has emerged as a pivotal imaging modality in this context, owing to its ability to noninvasively detect metabolically or receptor-active tumor lesions with high sensitivity. This study investigates [68Ga]Ga-CCZ01048, a novel PET radiopharmaceutical targeting MC1Rs, which are overexpressed in melanoma cells.
A prior first-in-human study by Yang et al. evaluated a different 68Ga-labeled α-MSH analogue and demonstrated promising uptake and dosimetric characteristics24. While their findings confirmed the feasibility of MC1R-targeted PET imaging, our study expands this work by evaluating [68Ga]Ga-CCZ01048, a compound with improved pharmacokinetic properties and high receptor affinity. Notably, this is the first clinical study to conduct a direct intra-patient comparison with [18F]FDG, the current standard of care, thereby enabling a comprehensive evaluation of tumor-to-background contrast.
[18F]FDG PET/CT remains the most widely utilized modality for melanoma imaging, primarily targeting glucose metabolism33. However, a key limitation of FDG lies in its non-specific uptake, particularly in inflammatory or immune-activated tissues, which often results in false-positive findings and subsequent diagnostic uncertainty. Moreover, its effectiveness may be compromised in melanoma subtypes with low metabolic activity, where tumor visualization becomes challenging. In contrast, [68Ga]Ga-CCZ01048 selectively targets MC1Rs, significantly enhancing tumor specificity and minimizing uptake in non-target tissues. This receptor-based targeting translated into markedly higher tumor-to-liver uptake ratios (Table 2), thereby improving lesion contrast and detectability, especially in metastatic settings where [18F]FDG may yield suboptimal performance.
Quantitative image analysis demonstrated that [68Ga]Ga-CCZ01048 exhibits greater sensitivity and specificity than [18F]FDG, driven by more favorable biodistribution characteristics. Specifically, the mean SUVmax in normal tissues was substantially lower for [68Ga]Ga-CCZ01048 (1.7) compared to [18F]FDG (6.1), while the tumor-to-liver SUVmean ratio was significantly higher (6.4 vs. 2.9), yielding improved tumor-to-background contrast. Notably, [68Ga]Ga-CCZ01048 exhibited minimal physiological uptake in the brain (SUVmax = 0.05), compared to the high FDG uptake in cerebral tissue (SUVmax = 6.5), underscoring its suitability for detecting brain metastases. Additionally, its reduced uptake in muscle and lung tissues further enhances the signal-to-noise ratio for detecting metastatic lesions in these areas. Collectively, these results confirm the diagnostic superiority of [68Ga]Ga-CCZ01048, particularly in clinical contexts where [18F]FDG’s physiological uptake may obscure tumor visualization.
Another promising melanoma-specific PET tracer, 18F-P3BZA, targets melanin, a characteristic pigment in melanomas34. Prior studies have shown that 18F-P3BZA exhibits high tumor affinity and selectivity, making it a viable alternative to [18F]FDG. However, our comparative analysis revealed indicates that 18F-P3BZA has a notable physiological uptake in the liver and stomach, potentially affecting diagnostic clarity. In contrast, [68Ga]Ga-CCZ01048 exhibited a more favorable biodistribution with reduced hepatic accumulation, which may be beneficial in cases where liver metastases are suspected.
Table 4 presents a comparative overview of the organ-specific absorbed doses for [68Ga]Ga-CCZ01048, [18F]FDG, 18F-P3BZA, and [68Ga]Ga-DOTATATE. The absorbed doses for major organs are similar across all tracers, suggesting comparable safety profiles. However, one notable advantage of fluorine-18-based tracers is their lower positron range compared to gallium-68, leading to better spatial resolution. Although gallium-68 has a higher positron yield, its more extended positron range can result in reduced imaging resolution, particularly for detecting small lesions. This factor may be a limitation for [68Ga]Ga-CCZ01048 compared to 18F-based tracers like 18F-P3BZA.
A notable practical advantage of [68Ga]Ga-CCZ01048 is its production via 68Ge/68Ga generators enabling on-site synthesis without the need for a cyclotron, unlike 18F-based radiotracers. This logistical advantage makes gallium-68-labeled compounds particularly useful in centers without direct access to fluorine-18 production facilities. Additionally, the rapid systemic clearance of [68Ga]Ga-CCZ01048 reduces radiation exposure and allows for repeat imaging when necessary, which further underscores its suitability for routine clinical use36.
Although the renal absorbed dose for [68Ga]Ga-CCZ01048 (0.0945 mSv/MBq) exceeds that of [18F]FDG (0.017 mSv/MBq), this difference remains clinically acceptable and does not pose a limitation for diagnostic use, as evidenced by the routine application of other 68Ga-labeled agents34. Importantly, the renal dose is only marginally higher than that of [68Ga]Ga-DOTATATE (0.0921 mSv/MBq), a tracer approved by the FDA in 2016, underscoring its safety profile within established clinical thresholds23. A key advantage of [68Ga]Ga-CCZ01048 over FDG PET is its superior tumor-to-background contrast, particularly in tissues where high physiological FDG uptake can obscure lesion detection. Its higher tumor-to-liver SUVmean ratio (6.4 vs. 2.9 for [18F]FDG) enhances lesion detectability, particularly in the liver, muscle, and brain, where the non-specific uptake of [18F]FDG may compromise imaging accuracy. Additionally, studies indicate that 68Ga -labeled tracers generally have a favorable safety profile and acceptable dosimetry compared to other PET tracers. Despite the higher renal dose, this is not a limiting factor due to the diagnostic nature of the study and the well-established clinical use of 68Ga -based tracers such as DOTATATE. Therefore, [68Ga]Ga-CCZ01048 presents a valuable alternative to FDG PET, offering improved imaging contrast and enhanced lesion detectability in specific clinical applications.
Our findings suggest that [68Ga]Ga-CCZ01048 is a promising PET radiotracer for melanoma imaging, offering advantages over [18F]FDG and complementing the capabilities of 18F-P3BZA. While [18F]FDG remains the standard for metabolic imaging, receptor-targeted approaches such as [68Ga]Ga-CCZ01048 provide enhanced specificity, particularly in patients undergoing immunotherapy or in cases of low-metabolic-activity tumors. Further studies with larger patient cohorts are necessary to validate these findings and explore potential theranostic applications by pairing [68Ga]Ga-CCZ01048 with therapeutic isotopes like 177Lu37. These advancements could pave the way for more personalized melanoma diagnostics and treatment strategies, ultimately improving patient management and clinical outcomes.
Conclusion
This first-in-human evaluation of [68Ga]Ga-CCZ01048 demonstrates its clinical feasibility and diagnostic potential as a PET radiopharmaceutical for metastatic melanoma. The agent exhibited high tumor specificity, rapid systemic clearance, and an acceptable dosimetric profile, supporting its safe and effective use in clinical imaging protocols. By addressing key limitations of [18F]FDG, [68Ga]Ga-CCZ01048 provides a more targeted and informative alternative, particularly in patients undergoing immunotherapy or with low-metabolism tumors. Additionally, its physicochemical and biological characteristics make it a strong candidate for theranostic applications, potentially enabling pairing with therapeutic isotopes such as 177Lu for personalized management of melanoma. These promising findings warrant further validation in larger clinical cohorts to support regulatory advancement and widespread adoption in oncological nuclear medicine.
Data availability
"The datasets used and/or analyzed during the current study are available from the corresponding author, H. Yousefnia (hyousefnia@aeoi.org.ir), on reasonable request."
References
Ahmed, B., Qadir, M. I. & Ghafoor, S. Malignant melanoma: Skin cancer− diagnosis, prevention, and treatment. Crit. Rev. Eukaryot. Gene Exp. 30, 4. https://doi.org/10.1615/CritRevEukaryotGeneExpr.2020028454 (2020).
Schadendorf, D. et al. Melanoma. Lancet 392, 971–984 (2018).
Siegel, R. L., Giaquinto, A. N. & Jemal, A. Cancer statistics, 2024. CA Cancer J. Clin. 74, 12–49. https://doi.org/10.3322/caac.21820 (2024).
Sundararajan, S., Thida, A. M., Yadlapati, S., Mukkamalla, S. K. & Koya, S. Metastatic melanoma. In StatPearls [Internet] (StatPearls Publishing, 2024).
Zamani-Siahkali, N., Mirshahvalad, S. A., Pirich, C. & Beheshti, M. Diagnostic performance of [18F] F-FDG positron emission tomography (PET) in non-ophthalmic malignant melanoma: A Systematic review and meta-analysis of more than 10,000 melanoma patients. Cancers 16, 215. https://doi.org/10.3390/cancers16010215 (2024).
Kumar, R., Mavi, A., Bural, G. & Alavi, A. Fluorodeoxyglucose-PET in the management of malignant melanoma. Radiol. Clin. North Am. 43, 23–33 (2005).
Fletcher, J. W. et al. Recommendations on the use of 18F-FDG PET in oncology. J. Nucl. Med. 49, 480–508 (2008).
Rabkin, Z., Israel, O. & Keidar, Z. Do hyperglycemia and diabetes affect the incidence of false-negative 18F-FDG PET/CT studies in patients evaluated for infection or inflammation and cancer?. J. Nucl. Med. 51, 1015–1020. https://doi.org/10.2967/jnumed.109.074294 (2010).
Kudura, K. et al. Malignancy rate of indeterminate findings on FDG-PET/CT in cutaneous melanoma patients. Diagnostics 11, 883. https://doi.org/10.3390/diagnostics11050883 (2021).
Cazzato, G. et al. GLUT1, GLUT3 expression and 18FDG-PET/CT in human malignant melanoma: What relationship exists? New insights and perspectives. Cells 10, 3090. https://doi.org/10.3390/cells10113090 (2021).
Rivas, A. et al. 18FDG PET assessment of therapeutic response in patients with advanced or metastatic melanoma treated with first-line immune checkpoint inhibitors. Cancers 14, 3190. https://doi.org/10.3390/cancers14133190 (2022).
Shanehsazzadeh, S. et al. Preclinical evaluation of 68Ga -MAA from commercial available 99mTc-MAA kit. Iran. J. Pharm. Res. 16, 1415 (2017).
Rabie, A. et al. Preparation, quality control and biodistribution study of 68Ga -BPAMD: Optimized production with an in-house 68Ge-68Ga generator. Iran. J. Nucl. Med. 26, 82–86 (2018).
Sharifi, M. et al. Preparation and biodistribution assessment of 68Ga-DKFZ-PSMA-617 for PET prostate cancer imaging. Nucl. Sci. Tech. 27, 1–9. https://doi.org/10.1007/s41365-016-0134-2 (2016).
Sharifi, M. et al. Production, quality control, biodistribution and imaging studies of 177Lu-PSMA-617 in breast adenocarcinoma model. Radiochim. Acta 106, 507–513. https://doi.org/10.1515/ract-2017-2873 (2018).
Zhang, C. et al. 18F-labeled cyclized α-melanocyte-stimulating hormone derivatives for imaging human melanoma Xenograft with positron emission tomography. Sci. Rep. 9, 13575. https://doi.org/10.1038/s41598-019-50014-5 (2019).
Zhang, C., Lin, K. S. & Bénard, F. Molecular imaging and radionuclide therapy of melanoma targeting the melanocortin 1 receptor. Mol. Imaging 16, 1536012117737919. https://doi.org/10.1177/1536012117737919 (2017).
Zhang, C. et al. Preclinical melanoma imaging with 68Ga-labeled α-melanocyte-stimulating hormone derivatives using PET. Theranostics 7, 805–813. https://doi.org/10.7150/thno.17117 (2017).
Zhang, C. et al. Human melanoma imaging with 68Ga -and 18F-labeled alpha-melanocyte-stimulating hormone derivatives using positron emission tomography. Theranostics 7, 805–813 (2017).
Zhang, C. et al. Targeting the melanocortin-1 receptor with 177Lu-labeled alpha-melanocyte stimulating hormone derivatives: Increased tumor uptake using an albumin binder. ACS Omega 5, 10767–10773 (2020).
Zhang, C. et al. Selective cyclized α-melanocyte-stimulating hormone derivative with multiple N-methylations for melanoma imaging with positron emission tomography. ACS Omega 5, 10767–10773. https://doi.org/10.1021/acsomega.0c00310 (2020).
. Shafiei, S. et al. Preparation and preclinical study of [68Ga] Ga-(Pip)-Nle-CycMSH hex: Optimized production with an in-house 68 Ge/68Ga generator. Iran. J. Nucl. Med. 32, (2024). https://doi.org/10.22034/IRJNM.2024.129486.1590
Stokke, C. et al. EANM guidance document: Dosimetry for first-in-human studies and early phase clinical trials. Eur. J. Nucl. Med. Mol. Imaging 51, 1268–1286. https://doi.org/10.1007/s00259-024-06640-x (2024).
Yang, J. et al. 68Ga -DOTA-GGNle-CycMSHhex targets the melanocortin-1 receptor for melanoma imaging. Sci. Transl. Med. 10, 466. https://doi.org/10.1126/scitranslmed.aau4445 (2018).
Schmitt, V. et al. Quantitative SPECT imaging and biodistribution point to molecular weight independent tumor uptake for some long-circulating polymer nanocarriers. RSC Adv. 8, 5586–5595. https://doi.org/10.1039/C7RA09183D (2018).
Drake, R., Vogl, A. W. & Mitchell, A. W. Gray’s anatomy for students (Elsevier Health Sciences, 2009).
Frank, H. & Netter, M. D. Atlas of human anatomy (Elsevier Inc, 2019).
Vaez-Tehrani, M., Zolghadri, S., Yousefnia, H. & Afarideh, H. Estimation of human absorbed dose for Ho-166-PAM: Comparison with Ho-166-DOTMP and Ho-166-TTHMP. Br. J. Radiol. 89, 1066. https://doi.org/10.1259/bjr.20160153 (2016).
Yousefnia, H. & Zolghadri, S. Estimated human absorbed dose of a new 153Sm bone-seeking agent based on biodistribution data in mice: Comparison with 153Sm-EDTMP. Phys. Med. 31, 714–719. https://doi.org/10.1016/j.ejmp.2015.05.015 (2015).
Shanehsazzadeh, S. et al. Estimated human absorbed dose for 68Ga-ECC based on mice data: Comparison with 67Ga-ECC. Ann. Nucl. Med. 29, 475–481. https://doi.org/10.1007/s12149-015-0967-5 (2015).
Shanehsazzadeh, S. et al. Comparison of estimated human dose of 68Ga-MAA with 99mTc-MAA based on rat data. Ann. Nucl. Med. 29, 745–753. https://doi.org/10.1007/s12149-015-0997-z (2015).
Stabin, M. G., Sparks, R. B. & Crowe, E. OLINDA/EXM: The second-generation personal computer software for internal dose assessment in nuclear medicine. J. Nucl. Med. 46, 1023–1027 (2005).
Adeleye, B. & Chetty, N. Radiation dose from 18F-FDG PET/CT procedures: Influence of specific CT model and protocols. Radioprotection 53, 107–113. https://doi.org/10.1051/radiopro/2018009 (2018).
Ma, X. et al. Biodistribution, radiation dosimetry, and clinical application of a melanin-targeted PET probe, 18F–P3BZA, in patients. J. Nucl. Med. 60, 16–22. https://doi.org/10.2967/jnumed.118.209643 (2019).
Walker, R. C. et al. Measured human dosimetry of 68Ga -DOTATATE. J. Nucl. Med. 54, 855–860. https://doi.org/10.2967/jnumed.112.114165 (2013).
Sciuto, R. et al. The role of dosimetry and biological effects in metastatic castration–resistant prostate cancer (mCRPC) patients treated with 223Ra: First in human study. J. Exp. Clin. Cancer Res. 40, 281. https://doi.org/10.1186/s13046-021-02056-9 (2021).
Ballal, S. et al. First-in-human results on the biodistribution, pharmacokinetics, and dosimetry of [177Lu]Lu-DOTA.SA.FAPi and [177Lu]Lu-DOTAGA(SA.FAPi)2. Pharmaceuticals 14, 1212. https://doi.org/10.3390/ph14121212 (2021).
Author information
Authors and Affiliations
Contributions
S. Zolghadri performed the main parts of the study. M. Bakhshi Kashi, N. Vahidfar, and S. Farzanefar conducted the clinical part of the study. A. Karimian and M.H. Mohammadi Ashnani performed the dosimetry calculations, the safety evaluations, and helped in different parts of the study. H. Yousefnia conducted the whole of the study, analyzed the data, and wrote the manuscript. All authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zolghadri, S., Bakhshi Kashi, M., Vahidfar, N. et al. First-in-human dosimetry and safety evaluation of 68Ga-αMSH derivative for PET imaging of melanoma. Sci Rep 15, 17748 (2025). https://doi.org/10.1038/s41598-025-02334-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-02334-y
This article is cited by
-
Assessment of human organs absorbed dose of 113mIn-BBN: extrapolation from mice biodistribution data using different phantom models
Journal of Radioanalytical and Nuclear Chemistry (2026)