Abstract
A novel acidic deep eutectic solvent (ADES) has been developed as an efficient and sustainable alternative to traditional solvents and catalysts in organic synthesis. The ADES was synthesized through a simple one-pot procedure involving benzyl chloride, 2-(dimethylamino)ethanol, and p-toluenesulfonic acid, yielding a homogeneous liquid. Structural and compositional characterization was performed using FTIR and NMR spectroscopy. The catalytic performance of the synthesized ADES was evaluated in the Pechmann condensation for the synthesis of coumarins. The reaction proceeded under mild conditions using commercially available starting materials, with reaction times ranging from 5 to 200 min and isolated yields between 72% and 97%. The process is operationally simple and readily scalable, indicating its suitability for industrial implementation. Importantly, the ADES demonstrated excellent recyclability, maintaining catalytic activity over five consecutive cycles without significant loss of efficiency which makes it an environmentally friendly and economically viable option for large-scale synthesis.
Similar content being viewed by others
Introduction
Deep eutectic solvents (DESs) are an emerging class of environmentally benign solvents formed by combining a hydrogen bond acceptor (HBA), such as choline chloride or metal salts, to a hydrogen bond donor (HBD), including organic acids, sugars, or urea. This combination results in a eutectic mixture to a melting point significantly lower than that of the individual components, primarily due to strong hydrogen bonding interactions between the constituents1,2. The synthesis of DESs is straightforward, typically involving gentle heating and mixing until a homogeneous liquid forms3. Structurally, DESs exhibit disrupted crystalline lattices, imparting distinctive solvation properties and enabling tunable physicochemical characteristics4,5. Since their initial introduction by Abbott et al. in 2003, DESs have attracted considerable interest across a wide range of applications, including catalysis, metal extraction, electrochemistry, biomass processing, and pharmaceutical formulations6,7,8,9. Their low toxicity, biodegradability, ease of preparation, and capacity to dissolve a broad array of compounds, make them attractive alternatives to conventional solvents10,11,12,13,14,15. A subset of DESs, known as acidic deep eutectic solvents (ADESs), are specifically tailored to function as acidic media. These are formed by combining HBAs such as choline chloride or metal salts with acidic HBDs, including organic acids like oxalic acid, citric acid, formic acid, lactic acid, or p-toluenesulfonic acid16,17,18. The resulting ADESs feature strong hydrogen bonding networks, tunable acidity, and high solubility for polar substrates. These characteristics render them especially useful in acid-catalyzed transformations, metal processing techniques and green chemistry applications such as CO₂ capture and environmental remediation19,20,21,22,23.
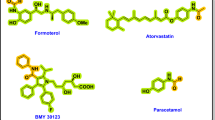
Coumarin is an aromatic organic compound found abundantly in various plant sources such as sweet clover, cinnamon, lavender, and tonka beans24,25,26,27,28. Coumarin is characterized by a sweet odor and serves as the structural core for numerous bioactive derivatives29,30,31,32. These derivatives such as warfarin, dicoumarol, scopoletin, umbelliferone, and aesculetin exhibit a wide range of pharmacological activities including anticoagulant, antioxidant, anti-inflammatory, antibacterial, and anticancer effects (Fig. 1)33,34,−35.In addition to pharmaceutical applications, coumarins are also utilized in the fragrance and cosmetics industries36. Over the years, several synthetic strategies have been developed for the preparation of coumarin derivatives, including the Pechmann condensation, Knoevenagel condensation, and Wittig reaction37,38,39,40,41,42,43. Recent research has increasingly focused on green chemistry approaches, such as the use of DESs, ionic liquids, and microwave-assisted synthesis, to enhance sustainability and reduce environmental impact. Notably, a DES system based on choline chloride and L-(+)-tartaric acid has been previously applied in the Pechmann condensation for coumarin synthesis44. While effective, this approach required relatively large quantities of DES, extended reaction times, and harsher reaction conditions, thereby limiting its scalability and practical utility.
In recent years, our research is focused on advancing the principles of green chemistry in organic synthesis through the development of sustainable methodologies, including aqueous systems, solvent-free conditions, and DESs45,46,47. In this context, we have designed and synthesized a novel acidic deep eutectic solvent (ADES), composed of benzyl chloride, 2-(dimethylamino)ethanol, and p-toluenesulfonic acid. The synthesized ADES was characterized using various spectroscopic techniques, which confirms its composition and structure. This novel ADES serves as an effective and versatile catalytic medium, facilitating the construction of structurally diverse coumarin frameworks employing various substituted phenols and salicylaldehyde in combination with different β-ketoesters.
The experimental
Materials and methods
All reagents, including phenol derivatives, salicylaldehydes, β-keto esters, benzyl chloride, 2-(dimethylamino)ethanol, and p-toluenesulfonic acid, were purchased from Sigma-Aldrich and used without further purification. Melting points were determined using a Büchi 535 melting point apparatus. Fourier-transform infrared (FT-IR) spectra were recorded on an AVATAR spectrometer. Nuclear magnetic resonance (NMR) spectra of ADES were recorded in DMSO-d6 using a Bruker Fourier 300 MHz spectrometer (Germany).
Preparation of ADES
A mixture of benzyl chloride (200 mmol) and 2-(dimethylamino)ethanol (200 mmol) was placed in a round-bottom flask equipped with a magnetic stirrer and heated to 80 °C under a nitrogen atmosphere. The reaction mixture was stirred continuously for 8 h. Upon completion of the initial stage, p-toluenesulfonic acid (200 mmol) was added to the reaction mixture and stirring was continued at the same temperature for an additional 2 h. The reaction mixture gradually transformed into a viscous liquid, indicating the successful formation of the acidic deep eutectic solvent (ADES), with a viscosity of 680 cP, density of 1.26 g/mL, and a pH of 2.3 (100 mM solution, 25 °C). (Fig. 2).
Typical procedure for preparation of 7-hydroxy-4-methylcoumarin derivatives
In a typical experiment, resorcinol (1.0 mmol) and ethyl acetoacetate (1.0 mmol) were dissolved in the synthesized ADES (0.5 mL) in a test tube equipped with a magnetic stir bar and sealed with a septum. The reaction mixture was stirred vigorously at 100 °C for 80–200 min. Upon completion, the mixture was allowed to cool to room temperature, followed by the addition of distilled water (15 mL). The resulting precipitate was isolated by centrifugation and purified either by recrystallization from ethanol or tetrahydrofuran, or by column chromatography using a petroleum ether/ethyl acetate mixture as the eluent, affording pure 7-hydroxy-4-methylcoumarin derivatives. All synthesized compounds were previously reported in the literature. Their identities were confirmed by comparison of melting points with those of authentic samples or previously published data.
Typical procedure for the preparation of 3-ethoxycarbonylcoumarin derivatives
In a test tube with magnetic stirring bar, a mixture of salicylaldehyde (1.0 mmol) and dimethyl malonate (1.0 mmol) was added to water (5 mL) containing DES (0.2 mL), and the reaction mixture was stirred vigorously at room temperature for 5–15 min. Upon completion of the reaction, the resulting crystalline product was isolated by simple filtration to afford 3-ethoxycarbonylcoumarin. The crude solid obtained was further purified by recrystallization from ethanol to give the pure product. All synthesized compounds were crystalline, and their melting points were determined and compared with those reported in the literature, confirming their identity and purity.
Results and discussion
Characterizations
Characterization of ADES by NMR and FTIR analysis
After the preparation of ADES, characterization was carried out using ¹H NMR and FTIR spectroscopy. In the ¹H NMR spectrum recorded in DMSO-d₆ solvent, the methyl protons of p-toluenesulfonic acid (PTSA) appeared as a singlet at 2.29 ppm. The methylene group (CH₂) attached to the hydroxyl group (–OH) was observed at 3.01 ppm. The CH₂ and CH₃ groups attached to the nitrogen atom (–N) appeared at 3.39 ppm and 3.91 ppm, respectively. The benzylic CH₂ protons were found at 4.59 ppm. The aromatic protons corresponding to both phenyl rings were detected in the range of 7.12–7.59 ppm (Fig. 3).
Furthermore, the ¹³C NMR spectrum of ADES confirmed the structure of the synthesized ADES. The methyl carbon of p-toluenesulfonic acid was observed at 21.2 ppm. The methylene carbon (CH₂) attached to the hydroxyl group appeared at 50.2 ppm. The methyl groups attached to the nitrogen atom were detected at 55.3 ppm, while the methylene carbon attached to the nitrogen was observed at 65.3 ppm. The benzylic methylene carbon (CH₂–Ph) was found at 67.9 ppm. Finally, the aromatic carbons for both phenyl rings were observed in the range of 125.9–145.8 ppm (Fig. 4).
The chemical structures of the ADES system were further analyzed based on FT-IR spectroscopy. As shown in Fig. 5, ADES exhibited a broad absorption peak around 3362 cm⁻¹, which corresponds to the O–H stretching vibration, indicating the presence of hydroxyl groups in the ADES structure as well as absorbed moisture from the environment. A broad peak observed at approximately 2123 cm⁻¹ is attributed to strong interactions between the salt and p-toluenesulfonic acid. Additional characteristic peaks were observed at 1637 cm⁻¹ and 1481 cm⁻¹, corresponding to the bending vibration and stretching vibration of the N–C bond in the amino group, respectively. The peak at 860 cm⁻¹ is ascribed to the stretching vibration of the S–OH group. Furthermore, two distinct peaks at 1085 cm⁻¹ and 1217 cm⁻¹ were assigned to the antisymmetric and symmetric stretching vibrations of the S = O bonds, respectively.
Catalytic evaluation of DES in the synthesis of coumarin derivatives
Following the preparation and characterization of ADES composed of p-toluenesulfonic acid (PTSA) and benzyl dimethyl (2-hydroxyethyl) ammonium chloride (BDC), their catalytic activity was evaluated in the synthesis of coumarin derivatives. The reaction between resorcinol (1.0 mmol) and ethyl acetoacetate (1.0 mmol) served as a model, and various DES systems and conditions were screened. The results are summarized in Table 1. Among the DES system tested, the PTSA–BDC system demonstrated the highest catalytic efficiency, yielding the target coumarin derivative 3a in excellent yield. First, the model reaction was carried out at room temperature for 200 min, resulting in negligible product formation. (Table 1, Entry 1). To improve the reaction yield, elevated temperatures were investigated. Although a slightly higher yield was obtained at 120 °C compared to 100 °C, the difference was not significant. Consequently, 100 °C was selected as the optimal reaction temperature for subsequent experiments (Table 1, Entry 5). To further evaluate the catalytic potential of alternative DES systems, the model reaction was carried out under optimized conditions (100 °C, 120 min) using various DES combinations, including oxalic acid: BDC (Entry 7), SnCl₂:BDC (Entry 8), ZnCl₂:BDC (Entry 9), SnCl₂–ChCl (Entry 10), ZnCl₂–ChCl (Entry 11), PTSA–ChCl (Entry 12), oxalic acid–ChCl (Entry 13), and urea–ChCl (Entry 14). However, none of these systems outperformed the PTSA–BDC combination in terms of product yield, thereby confirming its superior catalytic efficiency.
a Isolated yields. ChCl choline chloride, BDC benzyl dimethyl (2-hydroxyethyl) ammonium chloride, OX Oxalic acid.
Under the optimized reaction conditions, the scope of the reaction was extended to various phenols and β-ketoester derivatives, and the results summarized in Table 2. This environmentally benign procedure provides an efficient and general approach to the synthesis of coumarin derivatives, accommodating a wide range of functional groups including chloro, methoxy, methyl, and hydroxy on the phenolic ring. Both ortho-, meta-, and para-substituted phenols successfully underwent the Pechmann condensation, reacting smoothly with different β-ketoesters under operationally simple and mild conditions. The method proved to be broadly applicable, furnishing a diverse set of substituted coumarins in good to excellent yields, thereby demonstrating the versatility and efficiency of the PTSA/BDC based DES catalytic system48.
To further evaluate the reliability and versatility of the developed DES catalytic system, the scope of the reaction was extended to include salicylaldehyde and β-ketoester derivatives. As a model reaction, salicylaldehyde (1.0 mmol) and dimethyl malonate (1.0 mmol) were selected for the synthesis of 3-ethoxycarbonylcoumarin (6a). In the initial attempt, the reaction was performed in the PTSA–BDC-based DES system (Table 3, Entry 1) and this system afforded negligible yields. Consequently, a series of experiments were conducted under varied reaction conditions using different DES systems. The results of these screening experiments are summarized in Table 3. Following extensive trial-and-error optimization, it was discovered that the Knoevenagel condensation of salicylaldehyde (4) and diethyl malonate (5) proceeded efficiently and rapidly in water in the presence of DES at room temperature. The screening of various deep eutectic solvents (DESs) in aqueous media for the Pechmann condensation reaction revealed that a piperidine/acetic acid-based DES was the most effective, offering excellent product yield and cost-efficiency under room temperature (Table 3, entry 15). Interestingly, further increasing the amount of DES, prolonging the reaction time, or elevating the temperature to 60 °C did not result in any noticeable improvement in yield, highlighting the efficiency of the optimized conditions. In an effort to identify the most suitable reaction medium, several solvents were examined, including choline chloride–urea DES, polyethylene glycol (PEG), tetrahydrofuran (THF), dichloromethane (DCM), and ethanol. Among these, water was found to be the most effective solvent, significantly enhancing the yield of the desired coumarin derivatives under mild and environmentally friendly conditions. This observation is strongly supported by the experimental data presented in Table 3 confirming the advantage of using water as the reaction medium in combination with the piperidine/acetic acid DES system.
Under the optimized reaction conditions, various salicylaldehyde and ketoester derivatives were subjected to the optimized DES system, and the results are summarized in Table 4. The piperidine/acetic acid-based DES proved to be highly effective for all tested starting materials, showing excellent reactivity. A variety of salicylaldehyde derivatives, including 5-bromosalicylaldehyde and 4-hydroxy salicylaldehyde, along with ketoesters such as ethyl acetoacetate, methyl acetoacetate, malonitrile, and phenylacetoacetate, were successfully converted their corresponding coumarin products with good to excellent yields. The reactions proceeded rapidly, typically completing in less than 15 min, with clean formation of the desired products. Notably, the reaction system allowed for easy isolation of the products, with the precipitated coumarins being formed upon addition of additional water at the reaction mixture. This approach minimized the need for volatile organic solvents in the workup process. In many cases, purification was unnecessary, as the products obtained after direct filtration of the precipitate were reasonably pure. Importantly, compound 6a was synthesized on a larger scale (5 g, 97% yield) in just 10 min under similar conditions, demonstrating the scalability and efficiency of the method.
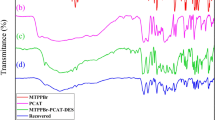
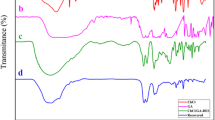
The reusability of the DES system was thoroughly investigated for the synthesis of compound 6a in a model reaction. The results revealed that the DES system remained highly effective over five consecutive reaction cycles without any significant decrease in yield. After each reaction, the solid product was separated by centrifugation, and the aqueous phase containing the DES was directly reused in the subsequent run. Figure 6 illustrates the stability of the system, with yields remaining consistent across all five cycles. This demonstrates the high catalytic efficiency and recyclability of the DES, which key advantage for large-scale industrial applications, aligning with the principles of green chemistry by minimizing waste and solvent use while maintaining excellent performance.
Conclusion
In summary, this study explored the use of novel acidic deep eutectic solvents as both catalysts and reaction media for the synthesis of coumarin under mild conditions. The ADES developed in this study offers several significant advantages over traditional solvents and catalysts. It provides a sustainable, environmentally friendly alternative by eliminating the need for hazardous reagents while maintaining high catalytic efficiency. The ADES system enables coumarin synthesis under mild conditions, thereby reducing energy consumption and minimizing waste generation. Moreover, the solvent demonstrated excellent recyclability, maintaining its catalytic activity for at least five consecutive runs with no significant loss in performance, making it both cost-effective and suitable for industrial-scale applications. Its dual function as both solvent and catalyst further simplifies the reaction system, enhancing the overall efficiency of the process.
Data availability
All data generated during this study are included in this published article.
References
Smith, E. L., Abbott, A. P. & Ryder, K. S. Deep eutectic solvents (DESs) and their applications. Chem. Rev. 114 (21), 11060–11082 (2014).
Hansen, B. B. et al. Deep eutectic solvents: A review of fundamentals and applications. Chem. Rev. 121 (3), 1232–1285 (2020).
El Achkar, T., Greige-Gerges, H. & Fourmentin, S. Basics and properties of deep eutectic solvents: a review. Environ. Chem. Lett. 19, 3397–3408 (2021).
Paiva, A. et al. C. Natural deep eutectic solvents–solvents for the 21st century. ACS Sustain. Chem. Eng. 2 (5), 1063–1071 (2014).
Perna, F. M., Vitale, P. & Capriati, V. Deep eutectic solvents and their applications as green solvents. Curr. Opin. Green. Sustainable Chem. 21, 27–33 (2020).
Liu, Y. et al. Natural deep eutectic solvents: properties, applications, and perspectives. J. Nat. Prod. 81 (3), 679–690 (2018).
Zhang, Q., Vigier, K. D. O., Royer, S. & Jérôme, F. Deep eutectic solvents: syntheses, properties and applications. Chem. Soc. Rev. 41 (21), 7108–7146 (2012).
Omar, K. A. & Sadeghi, R. Physicochemical properties of deep eutectic solvents: A review. J. Mol. Liq. 360, 119524 (2022).
Tang, B. & Row, K. H. Recent developments in deep eutectic solvents in chemical sciences. Monatshefte Für Chemie-Chemical Monthly. 144, 1427–1454 (2013).
Hayyan, M. et al. Saheed, O. K. Are deep eutectic solvents benign or toxic? Chemosphere 90 (7), 2193–2195 (2013).
Alonso, D. A. et al. Deep eutectic solvents: the organic reaction medium of the century. Eur. J. Organic Chem., 612–632 (2016). (2016)(4).
Liu, P., Hao, J. W., Mo, L. P. & Zhang, Z. H. Recent advances in the application of deep eutectic solvents as sustainable media as well as catalysts in organic reactions. RSC Adv. 5 (60), 48675–48704 (2015).
Zhang, M., Liu, Y. H., Shang, Z. R., Hu, H. C. & Zhang, Z. H. Supported molybdenum on graphene oxide/Fe3O4: an efficient, magnetically separable catalyst for one-pot construction of spiro-oxindole dihydropyridines in deep eutectic solvent under microwave irradiation. Catal Commun. 88, 39–44 (2017).
Gu, Y. & Jérôme, F. Bio-based solvents: an emerging generation of fluids for the design of eco-efficient processes in catalysis and organic chemistry. Chem. Soc. Rev. 42 (24), 9550–9570 (2013).
Gu, Y. Multicomponent reactions in unconventional solvents: state of the Art. Green Chem. 14 (8), 2091–2128 (2012).
Qin, H. et al. Overview of acidic deep eutectic solvents on synthesis, properties and applications. Green. Energy Environ. 5 (1), 8–21 (2020).
Cui, Y. et al. Design, synthesis and properties of acidic deep eutectic solvents based on choline chloride. J. Mol. Liq. 236, 338–343 (2017).
Chen, W., Jiang, J., Lan, X., Zhao, X. & Mou, H. Mu, T. A strategy for the dissolution and separation of rare Earth oxides by novel Brønsted acidic deep eutectic solvents. Green Chem. 21 (17), 4748–4756 (2019).
Ramón, D. J. & Guillena, G. (eds). Deep eutectic solvents: synthesis, properties, and applications. (2020).
Martínez, R., Berbegal, L., Guillena, G. & Ramón, D. J. Bio-renewable enantioselective aldol reaction in natural deep eutectic solvents. Green Chem. 18 (6), 1724–1730 (2016).
Tian, D. et al. Acidic deep eutectic solvents pretreatment for selective lignocellulosic biomass fractionation with enhanced cellulose reactivity. Int. J. Biol. Macromol. 142, 288–297 (2020).
Liu, W. et al. One-pot oxidative desulfurization of fuels using dual-acidic deep eutectic solvents. Fuel 265, 116967 (2020).
Morais, E. S., Freire, M. G., Freire, C. S., Coutinho, J. A. & Silvestre, A. J. Enhanced conversion of Xylan into furfural using acidic deep eutectic solvents with dual solvent and catalyst behavior. ChemSusChem 13 (4), 784–790 (2020).
Zak, J. et al. Establishing a flow process to Coumarin-8-Carbaldehydes as important synthetic scaffolds. Chemistry 18, 9901–9910 (2012).
Prousis, K. C., Avlonitis, N., Heropoulos, G. A. & Calogeropoulou, T. FeCl3-catalysed ultrasonic-assisted, solvent-free synthesis of 4-substituted coumarins. A useful complement to the Pechmann reaction. Ultrason. Sonochem. 21, 937–942 (2014).
Albadi, J., Shirini, F., Abasi, J., Armand, N. & Motaharizadeh, T. A green, efficient and recyclable Poly (4-vinylpyridine)-supported copper iodide catalyst for the synthesis of coumarin derivatives under solvent-free conditions. C. R. Chim. 16 (5), 407–411 (2013).
Sugino, T. & Tanaka, K. Solvent-free coumarin synthesis. Chem. Lett. 30, 110–111 (2001).
Bose, D. S., Rudradas, A. P. & Babu, M. H. The indium (III) chloride-catalyzed von Pechmann reaction: a simple and effective procedure for the synthesis of 4-substituted coumarins. Tetrahedron Lett. 43 (50), 9195–9197 (2002).
Gupta, M. et al. Regioselective, dehydrogenative cross‐coupling between formamides/aldehydes and coumarins by C–H functionalization. Eur. J. Org. Chem. 896–900 (2018).
Yuan, H. J., Wang, M., Liu, Y. J., Liu, Q. & Copper II)-Catalyzed C- C Bond‐Forming reactions of α‐Electron‐Withdrawing Group‐Substituted ketene S, S‐Acetals with carbonyl compounds and a facile synthesis of coumarins. Adv. Synth. Catal. 351 (1‐2), 112–116 (2009).
Hasan, A. H. et al. Novel thiophene Chalcones-Coumarin as acetylcholinesterase inhibitors: design, synthesis, biological evaluation, molecular docking, ADMET prediction and molecular dynamics simulation. Bioorg. Chem. 119, 105572 (2022).
Rehuman, N. A. et al. Halogenated coumarin–chalcones as multifunctional monoamine oxidase-B and butyrylcholinesterase inhibitors. ACS Omega. 6 (42), 28182–28193 (2021).
Sarothiya, D., Bhawale, R. T. & Kshirsagar, U. A. Organic-dye-catalyzed visible-light-mediated regioselective C-3 alkoxycarbonylation of imidazopyridines by carbazates. J. Org. Chem. 87 (21), 14915–14922 (2022).
Keshavarzipour, F. & Tavakol, H. The synthesis of coumarin derivatives using choline chloride/zinc chloride as a deep eutectic solvent. J. Iran. Chem. Soc. 13, 149–153 (2016).
Sinhamahapatra, A., Sutradhar, N., Pahari, S., Bajaj, H. C. & Panda, A. B. Mesoporous zirconium phosphate: an efficient catalyst for the synthesis of coumarin derivatives through Pechmann condensation reaction. Appl. Catal. A. 394 (1–2), 93–100 (2011).
Khan, D., Mukhtar, S., Alsharif, M. A., Alahmdi, M. I. & Ahmed, N. PhI (OAc) 2 mediated an efficient Knoevenagel reaction and their synthetic application for coumarin derivatives. Tetrahedron Lett. 58 (32), 3183–3187 (2017).
Bouasla, S. et al. Coumarin derivatives solvent-free synthesis under microwave irradiation over heterogeneous solid catalysts. Molecules 22 (12), 2072–2078 (2017).
Ramalingam, M., Mani, C., Manickam, S. & Srinivasalu, K. R. N-Doped carbon wrapped polyoxometalate derived from POM‐IL hybrid: A heterogeneous catalyst for the synthesis of coumarin derivatives under Solvent‐Free conditions. Eur. J. Inorg. Chem. 2019 (14), 1904–1910 (2019).
Li, S., Qi, X. & Huang, B. Synthesis of 7-hydroxy-4-methylcoumarin via the Pechmann reaction with PVP-supported phosphotungstic acid catalyst. Catal. Today. 276, 139–144 (2016).
Shi, X. L., Xing, X., Lin, H. & Zhang, W. Synthesis of polyacrylonitrile Fiber-Supported Poly (ammonium methanesulfonate) s as active and recyclable heterogeneous Brønsted acid catalysts. Adv. Synthesis Catal. 356 (10), 2349–2354 (2014).
Rajabi, F., Feiz, A. & Luque, R. An efficient synthesis of coumarin derivatives using a SBA-15 supported Cobalt (II) nanocatalyst. Catal. Lett. 145, 1621–1625 (2015).
Gu, Y., Zhang, J., Duan, Z. & Deng, Y. Pechmann reaction in non-chloroaluminate acidic ionic liquids under solvent‐free conditions. Adv. Synth. Catal. 347 (4), 512–516 (2005).
Zhang, Y. et al. Cholinium ionic liquids as cheap and reusable catalysts for the synthesis of coumarins via Pechmann reaction under solvent-free conditions. RSC Adv. 4 (44), 22946–22950 (2014).
Rather, I. A. & Ali, R. An efficient and versatile deep eutectic solvent-mediated green method for the synthesis of functionalized coumarins. ACS Omega. 7 (12), 10649–10659 (2022).
Saidi, M. R., Azizi, N. & Zali-Boinee, H. A simple one-pot three-component reaction for Preparation of secondary amines and amino esters mediated by lithium perchlorate. Tetrahedron 57 (31), 6829–6832 (2001).
Azizi, N. & Edrisi, M. Deep eutectic solvent immobilized on SBA-15 as a novel separable catalyst for one-pot three-component Mannich reaction. Microporous Mesoporous Mater. 240, 130–136 (2017).
Azizi, N., Yadollahy, Z. & Rahimzadeh-Oskooee, A. An atom-economic and odorless thia-Michael addition in a deep eutectic solvent. Tetrahedron Lett. 55 (10), 1722–1725 (2014).
Uroos, M. et al. Green synthesis of coumarin derivatives using Brønsted acidic pyridinium based ionic liquid [MBSPy][HSO 4] to control an opportunistic human and a devastating plant pathogenic fungus Macrophomina phaseolina. RSC Adv. 12 (37), 23963–23972 (2022).
Author information
Authors and Affiliations
Contributions
Material preparation, data collection, and analysis were performed by Fatemeh Mohammad. Najmedin Azizi supervised and Writing- review & editing. The first draft of the manuscript was written by Zohreh Mirjafari. Javad Mokhtari: Formal analysis, Investigation, Software, Writing – review & editing. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mohammad, F., Azizi, N., Mirjafari, Z. et al. A novel acidic deep eutectic solvent for sustainable and efficient coumarin synthesis. Sci Rep 15, 23482 (2025). https://doi.org/10.1038/s41598-025-07279-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-07279-w