Abstract
This study aimed to investigate the changes in the free amino acid profile, mineral content, phenolic compounds, and antioxidant activity of three mulberry species white (Morus alba), red (Morus rubra), and black (Morus nigra) following different drying methods (shade drying, and controlled drying at 55 and 65 °C). The results indicated that drying temperature significantly affected the retention of bioactive compounds. In the amino acid analyses, the highest proline content was recorded in white mulberry dried under shade conditions, reaching 834.80 mg/100 g. The GABA content was found to be 336.17 mg/100 g in red mulberry. In the mineral analyses, calcium content was determined as 4474 mg/kg in white mulberry and 10,660 mg/kg in red mulberry. Regarding phenolic content, shade-dried red mulberry showed the highest level at 10.34 mg GAE/g, followed by black mulberry at 9.69 mg GAE/g and white mulberry at 2.86 mg GAE/g. The highest antioxidant activity was observed in red mulberry, with a value of 47.68%. Overall, the findings suggest that low-temperature drying, particularly shade drying, is the most effective method for preserving the bioactive components of mulberry fruits.
Similar content being viewed by others
Introduction
Fruits and vegetables are rich sources of bioactive compounds, including vitamins, minerals, phenolic compounds, and amino acids, all of which are vital for human health. These components help neutralize free radicals, thereby reducing oxidative stress and contributing to the prevention of various chronic diseases such as cardiovascular diseases, cancer, and diabetes1,2,3.
Among these, mulberry (Morus spp.) is distinguished by its rich bioactive compound profile and long-standing use in traditional medicine. Belonging to the Moraceae family, the mulberry plant is widely cultivated around the world and is primarily classified into three main species: white (Morus alba), red (Morus rubra), and black (Morus nigra)4,5.
Different mulberry species demonstrate notable variations in their nutritional and bioactive compound compositions. White mulberry is characterized by its sweet flavor and widespread consumption as a fresh fruit, while red and black mulberries are particularly valued for their high anthocyanin content, which contributes to their deep red and black coloration. These anthocyanins provide significant health benefits due to their potent antioxidant properties6,7. Additionally, mulberries are rich in phenolic acids, flavonoids, and other antioxidant compounds that play a crucial role in cellular protection and health promotion8,9.
Traditionally, mulberry fruits have been recognized for their anti-inflammatory, antimicrobial, and neuroprotective effects. White mulberries are especially noted for their immune-supporting and blood sugar-regulating properties, while red and black mulberries offer strong oxidative stress-reducing effects attributed to their high polyphenol and anthocyanin contents10,11. Moreover, all mulberry species are significant sources of essential minerals such as potassium, calcium, magnesium, and iron, making them valuable contributors to the nutritional needs of different age groups12,13.
In modern food technology, extending the shelf life of foods while maintaining their nutritional and functional properties is a major challenge. Drying is one of the most widely used methods for preserving fruits and vegetables; however, the drying temperature and method can significantly impact the retention of bioactive compounds. High-temperature drying often leads to the degradation of heat-sensitive components such as phenolic compounds and vitamins, whereas low-temperature and controlled drying methods are more effective at preserving these constituents14,15. Although sun drying remains a traditional, low-cost method, it is highly dependent on environmental conditions and may result in variable product quality16.
Therefore, this study aims to investigate the effects of different drying methods (shade drying, controlled drying at 55 °C and 65 °C) on the total phenolic content, antioxidant activity, amino acid profile, and mineral composition of white, red, and black mulberries. The objective is to determine optimal drying conditions that minimize bioactive compound losses and to explore the potential application of mulberries in the development of functional food products. Given the limited information in the literature regarding the impact of drying on the bioactive components of mulberries, this study is expected to contribute valuable insights to both scientific research and food processing technologies.
Materials and methods
Materials
Three different mulberry species white (Morus alba), red (Morus rubra), and black (Morus nigra) were used in this study. Fully ripe fruits were harvested from mature trees located on the Harran University Osmanbey Campus (Şanlıurfa, Türkiye), which are part of long-established, semi-domesticated plantations.
Ripeness was determined based on species-specific indicators. Morus alba fruits were harvested when they turned translucent creamy-white, had a soft texture, and measured 1.5–2.0 cm in length, with a °Brix value of 17.5 ± 0.3. Morus rubra fruits were picked at a deep red to purplish-red stage, 2.0–2.5 cm in length, and ~ 19.2 ± 0.4 °Brix. Morus nigra fruits were considered ripe when they reached a dark purple to black color, soft texture, 2.0–2.8 cm in size, and a °Brix value of 20.5 ± 0.5.
Harvesting took place during the early morning hours of May 2024, under ambient temperatures of 22–26 °C and relative humidity of 50–55%. To preserve the native bioactive compound profile, fruits were transported under shaded conditions in ventilated crates and processed within 2 h of harvest.
After washing with clean water to remove debris (leaves, soil, etc.), samples were classified by size and color to create homogeneous experimental groups. Three drying methods were applied:
Shade drying: Conducted in a naturally ventilated area protected from direct sunlight. Ambient temperature ranged between 30 and 35 °C with ~ 60 ± 5% relative humidity. Fruits were laid in a single layer on perforated trays and turned twice daily. Drying lasted 72–96 h, depending on fruit variety and size.
Hot-air drying (55 °C and 65 °C): Conducted in a membrane-type hot air circulation oven. The drying chamber was maintained at ± 1 °C, with an air velocity of ~ 1.2 m/s and relative humidity < 20%. Fruits were placed in single layers on mesh trays and turned periodically. Drying times were approximately 30–36 h at 55 °C and 20–24 h at 65 °C, until a constant weight was reached (weight change < 0.1%).
The choice of 55 °C and 65 °C was based on literature-reported values commonly used in fruit dehydration studies, selected to compare moderate and elevated temperatures with respect to bioactive compound preservation and drying efficiency in mulberries.
After drying, all samples were allowed to cool at ambient conditions and then stored in airtight, opaque containers at room temperature (22–24 °C) to prevent light-induced degradation and moisture uptake. The final moisture content of the dried mulberries was determined using the gravimetric constant weight method and ranged between 11 and 14% (wet basis) depending on the drying temperature and fruit type. Although water activity (aw) was not directly measured, the moisture levels fall within acceptable ranges for shelf-. All drying treatments and subsequent analyses were performed in triplicate (n = 3). Analytical results were reported on a fresh weight basis, as the relatively narrow final moisture range helped to minimize variability across samples. Future studies may incorporate dry matter (DM) normalization to improve cross-study comparability.
Methods
Amino acid composition
Free amino acid profiles were analyzed using LC-MS/MS (Jasem Laboratory Systems) following hydrolysis of 0.5 g dried sample with 4 mL Reagent-2 at 110 °C for 24 h. After centrifugation (4000 rpm, 5 min), the supernatant was diluted, re-centrifuged, and 50 µL of the extract was mixed with internal standard and Reagent-1 for injection.
Chromatographic separation was achieved using an Agilent 1260 Infinity HPLC system with a Jasem amino acid column at 30 °C. The 3 µL injection volume and 0.7 mL/min flow rate enabled analysis within 7.5 min.
The method was validated via calibration curves (0.1–100 µg/mL, R2 > 0.998), limits of detection (0.02–0.10 µg/mL), and precision (intra-/inter-day RSD < 5%), aligning with literature data17.
Mineral composition
Samples (0.5 g) were digested with 6 mL HNO₃ and 2 mL H₂O₂ using microwave digestion. The digest was filtered and diluted for ICP-OES (Perkin Elmer Optima 2100 DV) analysis. Phosphorus, potassium, calcium, magnesium, sodium, iron, copper, manganese, zinc, and boron were quantified. Calibration standards (0.01–50 mg/L) yielded R² > 0.995; LODs were 0.005–0.05 mg/L. Accuracy (± 10% of CRM values) and precision (RSD < 5%) were verified18.
Total phenolic content and antioxidant capacity
Extraction was performed by mixing the dried mulberry powder with 70% methanol at a 1:10 (w/v) ratio. The mixture was homogenized using a mechanical homogenizer and sonicated for 10 min at room temperature, followed by shaking for 15 min and centrifugation at 26,000 × g for 20 min at 4 °C. The residue was re-extracted under the same conditions, and the supernatants were combined.
Total phenolic content (TPC) was determined using the Folin–Ciocalteu colorimetric method as described by Singleton and Rossi (1965), with gallic acid as the calibration standard. Absorbance was measured at 760 nm using a UV–Vis spectrophotometer (Shimadzu UV-1800, Kyoto, Japan). Results were expressed as mg gallic acid equivalents per gram of dry matter (mg GAE/g DM)19.
Antioxidant activity was evaluated by the DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging assay following the method of Brand-Williams et al. (1995). A 150 µL extract was mixed with 2850 µL of 0.1 mM DPPH solution in methanol and incubated for 1 h in the dark. Absorbance was measured at 515 nm using the same UV–Vis spectrophotometer (Shimadzu UV-1800, Kyoto, Japan)20.
All centrifugation steps were carried out using a refrigerated high-speed centrifuge (Hettich Rotina 420R, Tuttlingen, Germany). All analyses were conducted in triplicate (n = 3) for each sample and treatment to ensure accuracy and reproducibility.
Statistical analysis
Data were analyzed using SPSS 27.0 and MetaboAnalyst 6.0. Normality was assessed via Shapiro–Wilk test. One-way ANOVA with LSD post hoc was used for normal data; Kruskal–Wallis test with pairwise comparisons for non-normal data. Results were expressed as mean ± SD; significance was set at p < 0.05.
Results and discussion
Amino acid profile
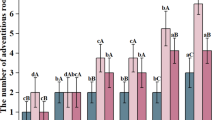
The free amino acid profiles of white, red, and black mulberries (Morus alba L., Morus rubra L., and Morus nigra L.) were analyzed following shade drying and controlled drying at 55 °C and 65 °C. The results are visualized in Fig. 1 as a heatmap, which highlights the variation in amino acid concentrations across different mulberry species and drying treatments.
In white mulberries, shade drying resulted in the highest levels of proline (834.80 mg/100 g), glutamic acid (303.47 mg/100 g), serine (447.10 mg/100 g), as well as elevated levels of tyrosine and threonine. Red mulberries dried at 55 °C showed increases in alanine (644.76 mg/100 g), leucine (363.70 mg/100 g), and tyrosine (525.77 mg/100 g), while shade-dried samples exhibited high proline content (513.09 mg/100 g). In black mulberries, alanine (967.10 mg/100 g) and proline (468.34 mg/100 g) were most concentrated at 55 °C, with additional increases in glutamic acid, serine, and valine.
GABA was notably abundant in shade-dried red (336.17 mg/100 g) and white (167.41 mg/100 g) mulberries.
In general, the lowest amino acid levels were recorded at higher drying temperatures, particularly 65 °C. However, some essential amino acids, including leucine, isoleucine, and valine, remained comparatively stable at 55 °C.
These results confirm that drying temperature significantly affects the preservation of bioactive amino acids. Shade drying (~ 45 °C) was most effective in retaining compounds such as proline, alanine, and GABA, while high-temperature drying at 65 °C led to degradation, underscoring the importance of low-temperature techniques for maintaining nutritional quality.
Heatmap visualization of free amino acid concentrations (mg/100 g) in white, red, and black mulberries subjected to shade drying, 55 °C, and 65 °C treatments. The color gradient reflects relative abundance, with darker tones indicating higher concentrations. Notable retention of amino acids such as proline, GABA, and alanine was observed under.
Comparative evaluation with previous studies confirmed the consistency of these findings. Taşova et al.21 reported that low-temperature oven drying (50 °C) was more favorable for maintaining product quality and preserving bioactive compounds in black mulberries. Similarly, Krzykowski et al.22 demonstrated that freeze-drying provided superior retention of phenolic compounds and ascorbic acid in white mulberries compared to air drying. In addition, Wang et al.23 indicated that fruit ripeness stages influenced drying quality and amino acid stability when using heat pump drying in black mulberries.
Studies on fruits with similar structural characteristics further support our findings. Martín-Gómez et al.24 reported reductions in amino acid and phenolic compound contents during hot air drying of grapes (Vitis vinifera), emphasizing that while fruit perforation accelerated drying, it exacerbated quality deterioration. Likewise, Michalczyk et al.25 showed that air drying caused significant losses of amino acids and polyphenols in small fruits such as strawberries, raspberries, and blueberries, whereas freeze-drying substantially mitigated these losses.
The substantial retention of bioactive amino acids, particularly proline and GABA, in the shade-dried mulberry samples highlights the potential of this method for the production of functional food products. Furthermore, the higher concentrations of essential amino acids (leucine, isoleucine, valine) observed in both shade-dried and 55 °C oven-dried samples underscore the critical role of low-temperature applications in preserving amino acid stability.
Beyond the influence of drying methods, intrinsic biochemical differences among mulberry species were clearly observed. For instance, red mulberries consistently demonstrated higher concentrations of GABA, tyrosine, and alanine compared to white and black varieties, regardless of the drying conditions. These findings are in agreement with recent characterizations of regional Morus genotypes, which have revealed significant intervarietal differences in amino acid biosynthesis and metabolite accumulation6. Furthermore, the markedly elevated proline levels observed in black mulberries following hot-air drying may indicate a stronger osmotic adjustment capacity and stress resilience specific to this genotype. These varietal distinctions highlight the importance of genetic background in shaping the nutritional and functional profiles of mulberries, even under identical postharvest treatments.
In conclusion, shade drying and moderate-temperature oven drying (55 °C) were found to be more suitable methods for preserving free amino acids in mulberry fruits, whereas high-temperature drying (65 °C) led to increased amino acid losses.
The degradation of specific amino acids at elevated drying temperatures (e.g., 65 °C) can be attributed to their thermolabile nature and the physicochemical transformations induced by heat. Amino acids such as proline and glutamic acid are particularly susceptible to thermal denaturation, oxidation, and structural modifications when exposed to prolonged heating, resulting in a reduction in their detectable concentrations26,27. In addition, the structural integrity of amino acids possessing polar side chains may be compromised due to thermal denaturation and pH fluctuations within the fruit matrix during the drying process26,27. These findings highlight the importance of gentle drying techniques in preserving the stability of heat-sensitive bioactive compounds. Therefore, when the use of mulberry fruits in functional foods and nutraceutical products is intended, it is recommended to employ drying methods conducted at low temperatures and under controlled conditions.
Mineral content
The effects of drying conditions on the mineral composition of white, red, and black mulberries are illustrated in Fig. 2. The logarithmic bar chart enables clear comparison across minerals with varying abundance levels. Overall, shade drying preserved the highest concentrations of both major (e.g., calcium, potassium) and trace minerals (e.g., iron, copper, zinc) across all mulberry species. In contrast, increasing drying temperatures, particularly 65 °C, resulted in significant reductions in mineral content, with red and black mulberries exhibiting relatively better retention compared to white mulberries.
Calcium (Ca), potassium (K), magnesium (Mg), manganese (Mn), copper (Cu), and iron (Fe) contents consistently decreased as drying temperature increased. For instance, Ca levels dropped from 4474 to 3339 mg/kg in white mulberries, and from 10,660 to 9238 mg/kg in red mulberries between shade drying and 65 °C. Similar trends were observed for Fe (e.g., 47.58 → 22.64 mg/kg in white mulberries) and K (e.g., 14010 → 10930 mg/kg).
Magnesium and manganese showed the greatest losses at 65 °C, particularly in black mulberries (e.g., Mg: 2235 → 1322 mg/kg; Mn: 21.95 → 11.49 mg/kg). Sodium (Na) content also decreased in white and black varieties, whereas red mulberries maintained relatively stable Na levels. Zinc (Zn) content fluctuated slightly in white mulberries but was largely unaffected in red and black varieties. These findings clearly demonstrate that higher drying temperatures negatively affect mineral retention in mulberries. Shade drying was the most effective method for preserving mineral quality.
Bar chart of mineral content (mg/kg) in white, red, and black mulberries under different drying conditions (shade, 55, 65 °C) displayed on a logarithmic scale. This visualization allows simultaneous comparison of high- and low-abundance minerals, highlighting the thermal sensitivity of elements like Fe, Cu, and Zn, and the relative stability of Ca and K.
When compared with the literature, the Fe content reported by Sun et al.25 in the Ji’an variety (467.32 mg/kg) was considerably higher than the levels observed in this study25. Our findings showing higher Ca and Mg contents in red mulberries are consistent with Saeed et al. (2023), who reported black mulberries as richer in these minerals compared to white ones28. These results suggest that mineral composition in mulberries varies depending on genetics, cultivation, and environmental factors.
Statistical analyses revealed significant effects of drying temperature on Cu, Fe, K, Mn, and Na levels29. In particular, the notable decrease in Cu from 45 °C to 65 °C highlights the sensitivity of this element to thermal degradation30. Similar losses were seen for Fe, K, Mn, and Na, which are critical in oxidative stress management, immune function, and electrolyte balance31,32,33.
High temperatures reduce the stability of oxidation-sensitive minerals like Cu, Fe, and Mn, contributing to losses during drying34. In our study, Cu content declined from 5.49 to 4.28 mg/kg, while K decreased from 14,730 to 10,763 mg/kg. Similar reductions have been reported in grapes and strawberries35.
In addition to thermal degradation, soluble minerals such as K and Na may migrate to the surface and be lost via evaporation. Overall, trace elements (Cu, Fe, Mn) are affected by oxidation, while K and Na are more prone to surface loss. Freeze-drying is known to be more effective in preserving mineral contents compared to hot-air drying methods36,37.
The Fe content in black mulberries decreased from 58.36 to 35.02 mg/kg with temperature rise, mirroring trends observed in other thermally processed products35. While Ca showed relative thermal stability, Na and K were more affected38. In our study, Ca did not vary significantly (p = 0.077), confirming its resistance to heat. Meanwhile, Na content decreased from 85 to 70–73 mg/kg, aligning with similar findings in carrots and grapes39.
Mineral losses may also result from surface-level heat-induced reactions such as the Maillard reaction, which can affect mineral binding and availability38. For example, Mn content dropped from 20.73 to 13.03 mg/kg, in agreement with previous reports on thermal mineral degradation.
In conclusion, key factors contributing to mineral loss during drying include oxidation, evaporation, drying method, mineral thermal stability, and surface chemical reactions. Low-temperature methods (e.g., shade drying, freeze-drying) are more effective for mineral retention. Future studies should focus on optimizing these techniques for enhanced nutritional quality.
Phenolic compounds and antioxidant activity
The total phenolic contents and antioxidant activities of white, red, and black mulberries under different drying methods are illustrated in (Fig. 3). This dual-axis bar chart enables a simultaneous comparison of both parameters, revealing that shade drying consistently resulted in higher retention of phenolic compounds and antioxidant capacity across all mulberry species, particularly in red and black varieties.
Shade-dried white mulberries exhibited the highest total phenolic content (2.86 mg GAE/g) and antioxidant activity (6.13%). These values declined significantly at 55 °C (1.63 mg GAE/g and 4.75%, respectively), with a partial increase observed at 65 °C.
In red mulberries, the highest phenolic content (10.34 mg GAE/g) and antioxidant activity (47.68%) were also found in shade-dried samples. Both parameters dropped markedly at 55 °C, followed by a slight increase at 65 °C.
A similar pattern was observed in black mulberries, where shade drying preserved the highest levels of phenolics (9.69 mg GAE/g) and antioxidant activity (43.50%). Values decreased at 55 °C and partially recovered at 65 °C.
Overall, shade drying consistently enabled greater retention of phenolic compounds and antioxidant activity across all mulberry species. These findings highlight that low-temperature drying, particularly shade drying, is more effective in preserving phenolics and enhancing antioxidant potential.
The findings of this study are consistent with previously reported data in the literature. For instance, Krzykowski et al.22 demonstrated that freeze-drying was superior to air drying in preserving phenolic compounds and ascorbic acid in white mulberry fruits22. Similarly, Martín-Gómez et al. (2019) showed that low-temperature drying (below 40 °C) minimized phenolic degradation in grape fruits24. In a study by Michalczyk et al.40, freeze-drying resulted in better retention of phenolics and antioxidant activity compared to hot-air drying at 60–70 °C in berries such as strawberries, raspberries, and blueberries40.
Additionally, Taşova et al.21 reported that drying black mulberries at 50 °C using a conventional oven preserved higher amounts of total phenolics and antioxidant activity than drying at higher temperatures such as 65 °C21. Likewise, Wang et al. (2020) showed that drying black mulberries at 55 °C using a heat pump dryer provided better amino acid and phenolic compound stability compared to 70 °C23. These studies support the current findings, which highlight that shade drying (~ 30–35 °C) and moderate-temperature drying at 55 °C are more effective in preserving bioactive compounds than drying at 65 °C.
Comparative bar chart showing total phenolic content (left axis, mg GAE/g dry matter) and antioxidant activity (right axis, %) in mulberries by species and drying method. GAE: gallic acid equivalents. Antioxidant activity was determined using the DPPH radical scavenging assay. Bars represent mean values; shade drying yielded the highest retention of both parameters.
The findings of this study are consistent with results reported in the literature. Krzykowski et al. (2023) reported that freeze-drying was superior to air drying in preserving phenolic compounds and ascorbic acid in white mulberry fruits22.
Similarly, Martín-Gómez et al.24 demonstrated that low-temperature drying minimized phenolic compound losses in grape fruits24. In a study conducted by Michalczyk et al. (2009) on strawberries, raspberries, and blueberries, freeze-drying was also found to be more effective than hot air drying in preserving phenolic content and antioxidant activity40.
These findings highlight the sensitivity of phenolic compounds to increasing temperatures and demonstrate that controlling the drying temperature is a critical factor for the preservation of bioactive compounds. Furthermore, although some losses were observed at 55 °C, the partial recovery of phenolic content and antioxidant activity at 65 °C could be attributed to potential enzyme inactivation, leading to greater stability of phenolic compounds.
In conclusion, shade drying appears to be the most suitable method for preserving the functional properties of mulberry fruits, while drying at moderate temperatures (55–65 °C) should be carefully optimized to minimize the loss of phenolic compounds.
Conclusion
In this study, the free amino acid profiles, phenolic contents, antioxidant activities, and mineral compositions of Morus alba, Morus rubra, and Morus nigra were analyzed under different drying conditions. Shade drying generally preserved higher levels of bioactive compounds, particularly phenolics and certain amino acids such as proline and GABA. Red mulberry exhibited the highest phenolic content (10.34 mg GAE/g) and antioxidant activity (47.68%), while white mulberry showed elevated proline levels (834.80 mg/100 g), and red mulberry had the highest GABA and calcium contents.
These results suggest that low-temperature drying especially shade drying can enhance the nutritional retention of mulberries. However, considering industrial scalability, hot-air drying at 55 °C may serve as a more practical compromise. The distinct biochemical profiles observed among mulberry species may also support targeted applications in functional food development. Future studies including dry matter normalization and biological activity assays are encouraged to further validate these findings.
Data availability
All data generated or analysed during this study are included in this published article.
References
Wallace, T. et al. Fruits, vegetables, and health: A comprehensive narrative, umbrella review of the science and recommendations for enhanced public policy to improve intake. Crit. Rev. Food Sci. Nutr. 60, 2174–2211 (2020).
Shabbir, U. et al. Curcumin, quercetin, catechins and metabolic diseases: the role of gut microbiota 13 Nutrients ( 2021).
Dhalaria, R. et al. Bioactive compounds of edible fruits with their anti-aging properties: A comprehensive review to prolong human life 9 Antioxidants (2020).
Wani, M. et al. The phenolic components extracted from mulberry fruits as bioactive compounds against cancer: A review. Phytother. Res. 37, 1136–1152 (2023).
He, X. et al. Structures, bioactivities and future prospective of polysaccharides from Morus alba (white mulberry): A review. Food Chem. 245, 899–910 (2018).
Uyak, C., Ağlar, E., Ozturk, B., Doğan, A. & Tekin, O. Biochemical characterization of mulberry (Morus spp.) genotypes from the Türkiye (Hizan, Bitlis): A comprehensive analysis of fruit properties and bioactive compounds. Food Sci. Nutr. 126425–6441 (2024).
Chen, T. et al. Evaluation of the chemical composition and antioxidant activity of mulberry (Morus Alba L.) fruits from different varieties in China. Molecules 27 (2022).
Abreu-Naranjo, R. et al. Bioactive compounds, phenolic profile, antioxidant capacity and effectiveness against lipid peroxidation of cell membranes of mauritia flexuosa L. fruit extracts from three biomes in the ecuadorian amazon. 6 Heliyon (2020).
Macedo, C., Costa, P. & Rodrigues, F. Bioactive compounds from Actinidia arguta fruit as a new strategy to fight glioblastoma. Food Res. Int. 175, 113770 (2023).
Yu, J., Lim, S., Lee, S., Choi, C. & Kim, K. Antioxidant and Anti-Inflammatory effects of white mulberry (Morus Alba L.) fruits on lipopolysaccharide-stimulated RAW 264.7 macrophages. Molecules 26 (2021).
Jung, S., Lee, M., Choi, A., Kim, C. & Kim, Y. Anti-Inflammatory effects of high hydrostatic pressure extract of mulberry (Morus alba) fruit on LPS-Stimulated RAW264.7 cells. Molecules 24 (2019).
Redha, A. et al. Comparison of fatty acid profile and mineral content of black mulberry (Morus nigra), white mulberry (Morus alba) and red mulberry (Morus rubra) grown in Bahrain. Appl. Food Res. (2023).
Kattil, A., Dash, H., Shams, K., Sharma, S. & R., & Nutritional composition, phytochemical extraction, and pharmacological potential of mulberry: A comprehensive review. Future Foods (2024).
Elgamal, R. et al. Thermal degradation of bioactive compounds during drying process of horticultural and agronomic products. Comprehen. Overview (2023).
Vidinamo, F., Vidinamo, F., Fawzia, S. & Karim, M. Effect of drying methods and storage with agro-ecological conditions on phytochemicals and antioxidant activity of fruits: a review. Crit. Rev. Food Sci. Nutr. 62, 353–361 (2020).
Goel, V. et al. Solar drying systems for domestic/Industrial purposes: A state-of-art review on topical progress and feasibility assessments. Solar Energy (2024).
Wang, P. et al. A rapid and efficient method of microwave-assisted extraction and hydrolysis and automatic amino acid analyzer determination of 17 amino acids from mulberry leaves. Indust. Crops Prod. (2022).
Göncü, A. & Celik, I. Investigation of some properties of gluten-free Tarhanas produced by red, green and yellow lentil whole flour. Food Sci. Technol. 40, 574–581 (2020).
Singleton, V., Orthofer, R. & Lamuela-Raventós, R. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Methods Enzymol. 299, 152–178 (1999).
Thaipong, K., Boonprakob, U., Crosby, K., Cisneros-Zevallos, L. & Byrne, D. H. Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J. Food Compos. Anal. 19 (6–7), 669–675 (2006).
Taşova, S., Ceylan, F. D. & Demir, M. K. Effects of different drying methods on the quality attributes and bioactive components of black mulberry (Morus Nigra L.). Food Sci. Technol., 42 (1), e65320. (2021).
Krzykowski, M., Mitek, M. & Nowacka, M. Freeze-drying as a method of preservation of white mulberry (Morus Alba L.)—Impact on phenolic compounds and antioxidant activity. Foods 12 (6), 1275 (2023).
Wang, K. et al. Freeze-drying as a method of preservation of white mulberry (Morus alba L.)—Impact on phenolic compounds and antioxidant activity. Food and Bioprocess Technol. (2023).
Martín-Gómez, J., Varo, M. Á., Mérida, J. & Serratosa, M. P. The influence of berry perforation on grape drying kinetics and total phenolic compounds. J. Sci. Food. Agric. 99 (9), 4260–4266 (2019).
Sun, B., Zhao, Y., Qiu, Y. & Wang, L. Quality determination and correlation analysis of mulberry fruits during different development stages. J. Berry Res. (2023).
Wen, P. et al. Effects of different dry heating temperatures on the Spatial structure and amino acid residue side-chain oxidative modification of soybean isolated proteins. 405 Pt A, 134795 https://doi.org/10.1016/j.foodchem.2022.134795 Food Chem. (2022).
Jiang, G., Townsend, W. & Ren, S. Analysis of seed amino acids in vegetable soybeans dried by freeze and thermal drying. Agronomy https://doi.org/10.3390/agronomy13020574 (2023).
Saeed, A., Kauser, S., Hussain, A. & Nadeem, A. A. Comparative variability of nutrients, minerals, phenolics and anthocyanins with antioxidant potentials during fruit development stages in five Mulberry (Morus) cultivars. J. Berry Res. (2023).
Thuy, N. et al. Optimization of mulberry extract Foam-Mat drying process parameters. Molecules 27 (2022).
Wang, L., Wen, H., Yang, N. & Li, H. Effect of vacuum freeze drying and hot air drying on dried mulberry fruit quality. 18 PLoS One (2023).
Li, M. et al. Convenient use of low field nuclear magnetic resonance to determine the drying kinetics and predict the quality properties of mulberries dried in hot-blast air. Lwt - Food Sci. Technol. 137, 110402 (2021).
Chen, Q. et al. Effect of hybrid drying methods on physicochemical, nutritional and antioxidant properties of dried black mulberry. Lwt - Food Sci. Technol. 80, 178–184 (2017).
Dharmarajan, K. & Minaker, K. Water, potassium, sodium, and chloride in nutrition. Geriatr. Gastroenterol. (2021).
Yildiz, G. Color, microstructure, physicochemical, textural and sensory properties with the retention of secondary metabolites in convective-, microwave- and freeze-dried Carrot (Daucus carota) slices. Br. Food J. 123 (13), 4221–4237 (2021).
Çoklar, H. & Akbulut, M. Effect of sun, oven and Freeze-Drying on anthocyanins, phenolic compounds and antioxidant activity of black grape (Ekşikara) (Vitis vinifera L). S. Afr. J. Enol. Vitic. 38 (2), 264–272 (2017).
Vega-Gálvez, A. et al. Drying kinetic modeling and assessment of mineral content, antimicrobial activity, and potential α-Glucosidase activity Inhibition of a green seaweed (Ulva spp.) subjected to different drying methods. ACS Omega. 7 (40), 34230–34238 (2022).
Uluocha, P. Effects of different drying methods on the micronutrients of four leafy vegetables traditionally consumed by some Clans in Izzi and Unwana, Ebonyi State, Nigeria. World J. Adv. Res. Rev. (2023).
Chen, Y. S., Phang, S. & Shuib, A. A preliminary study of chemical property and thermal stability of potassium chloride encapsulated in starch-alginate matrix. J. Phys. Conf. Ser. 2523 (1), 012035. (2023).
Multari, S., Marsol-Vall, A., Keskitalo, M., Yang, B. & Suomela, J. Effects of different drying temperatures on the content of phenolic compounds and carotenoids in quinoa seeds (Chenopodium quinoa) from Finland. J. Food Composit. Anal. (2018).
Michalczyk, M., Macura, R. & Matuszak, I. The effect of air-drying, freeze-drying and storage on the quality and antioxidant activity of some selected berries. J. Food Process. Preserv. 33 (1), 11–21 (2009).
Acknowledgements
I gratefully acknowledge the Harran University Center for Science and Technology Application and Research (HÜBTAM) for providing the laboratory infrastructure and technical assistance throughout the analytical procedures of this study.
Funding
The author did not receive any assistance or funding from any organization for the work presented.
Author information
Authors and Affiliations
Contributions
The sole author, Tuğba Gül Dikme, was responsible for the conception and design of the study, data collection, analysis and interpretation of the results, drafting and critical revision of the manuscript, and approval of the final version for submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The plant materials used in this study were collected and utilized in full compliance with institutional, national, and international guidelines and regulations. No specific permissions were required for the sampling of mulberry fruits from the Harran University Osmanbey Campus, as the trees are non-endangered and not subject to restricted access or collection policies.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gül Dikme, T. Preservation of bioactive compounds in mulberry species (Morus spp.) under different drying conditions. Sci Rep 15, 31398 (2025). https://doi.org/10.1038/s41598-025-08455-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-08455-8