Abstract
We characterized all ceftazidime-avibactam-resistant KPC-producing K. pneumoniae (KPC-Kp) isolates recovered from both patients and environmental samples at the ICU of our hospital in 2020, during the COVID-19 pandemic initiation. Antimicrobial susceptibility testing (Sensititre-EUMDROXF; disk-diffusion) and WGS analysis (Illumina-Novaseq/Miseq; Oxford Nanopore®-MinION) were performed. Ten patients (16% of ICU patients) were colonized/infected by a ceftazidime-avibactam-resistant KPC-Kp isolate (March-December), six of them during/after treatment with ceftazidime-avibactam. Two ceftazidime-avibactam-resistant KPC-Kp were also recovered from two ICU sinks (July-September). All isolates belonged to ST307 clone and had identical resistance gene content. Six KPC-variants were detected in patient isolates (KPC-62, KPC-92, KPC-150, KPC-66, KPC-53, KPC-46). KPC-92 and KPC-66 variants were also detected in sink isolates. Regardless of the origin (patients or sinks), KPC-92-, KPC-150 and KPC-62-Kp isolates combining altered porin proteins also exhibited increased/resistant MIC values to cefiderocol, cefepime-taniborbactam, aztreonam-avibactam, meropenem-vaborbactam and/or imipenem-relebactam. A cgMLST analysis demonstrated the clonal spread of KPC-ST307-Kp within the ICU, between patients and the hospital environment. Clustering was observed mainly by KPC variants (KPC-92, KPC-62). A variant calling analysis and plasmid characterization showed possible transmission between patients and sinks. Our results suggest that the patient care environment likely contributed to persistence and spread of last-line antibiotics-resistant KPC-Kp within the ICU during the COVID-19 pandemic.
Similar content being viewed by others
Introduction
Klebsiella pneumoniae is a significant nosocomial pathogen, particularly in intensive care units (ICUs), where infections caused by carbapenem-resistant strains have become a critical concern. These strains are often associated with the production of carbapenemases, such as Klebsiella pneumoniae carbapenemase (KPC), which confer resistance to a wide range of β-lactam antibiotics, including carbapenems, and involve defects of outer membrane porins that reduce permeability of β-lactams1,2. The emergence and spread of KPC-producing K. pneumoniae (KPC-Kp) has posed a serious challenge to infection control practices in healthcare settings, especially due to their ability to colonize patients, cause outbreaks and persist in hospital settings3,4. Currently, high-risk clones (HiRCs), such as ST307, play a major role in the spread of resistance, as they possess genetic characteristics that may provide an advantage in adaptation to the human host, but also to the hospital environment5,6.
Ceftazidime-avibactam is a combination of a third-generation cephalosporin with a β-lactamase inhibitor, that was introduced in 2015 as a novel therapeutic option for treating infections caused by carbapenem-resistant Enterobacterales, including KPC-Kp7. Despite its success, reports of resistance to ceftazidime-avibactam have emerged associated with mutations in the KPC enzyme and changes in outer membrane porins, particularly among KPC-Kp strains8,9,10.
Between 2019 and 2020, KPC-3 was the predominant carbapenemase among all K. pneumoniae clinical isolates collected in our hospital11. In 2020, the COVID-19 pandemic had a major impact on epidemiology, antimicrobial prescribing and resistance to novel antimicrobials in our institution, especially in patients with severe infections and admitted to critical units12,13. In the present study, we aimed to investigate the occurrence and characteristics of ceftazidime-avibactam resistant KPC-Kp isolates recovered from patients and sinks of a large ICU ward in our hospital during the COVID-19 early pandemic period (year 2020). We conducted microbiological and molecular analyses to identify resistance mechanisms to ceftazidime-avibactam and to assess the genetic relatedness of clinical and environmental KPC-Kp isolates.
Results
Bacterial isolates, patient`s data and ceftazidime-avibactam resistance
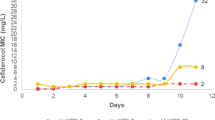
During 2020, 1,110 carbapenemase-producing K. pneumoniae isolates (390 clinical and 720 from rectal samples) were detected in 292 patients admitted to our hospital. Up to 64 of these patients (64/292, 21.9%) were admitted at the UCI and in 41 of them (41/64, 64.1%), a KPC-Kp isolate was detected. KPC-Kp isolates resistant or with decreased susceptibility to ceftazidime-avibactam (MICCZA: 8->16 mg/L) were detected in 10 ICU patients (10/64, 15.6%) between March and December 2020 (Table 1; Fig. 1). These isolates were recovered from rectal colonization samples in nine patients (9/10, 90%) and from clinical samples (three bronchial aspirates, two urines, one catheter and one blood) in five patients (5/10, 50%). The median length of stay (LOS) of these patients was 89 days (range, 26 to 129 days), and the median LOS at the ICU was 61 days (range, 2 to 76 days). Seven of these 10 patients had COVID-19 infection during their ICU stay. Infection control measures were applied to both patients and surfaces immediately after identification of each new case of colonization and/or infection.
Timeline of events in the ICU of our hospital during the year 2020. KPC-Kp (susceptible and resistant to ceftazidime-avibactam) recovered from patient between January and December are represented horizontally. KPC-Kp (resistant to ceftazidime-avibactam) recovered from environmental samples collected between July and September are represented in vertical (green zone). Length of stay (LOS) and LOS at the ICU of patients are indicated with a blue line and a dotted blue line, respectively. The ICU room where the patients stayed and where the environmental samples were collected is indicated by the framed number (on the left and below, respectively). Time of treatment with ceftazidime-avibactam (dose 2/0.5 g /8 h iv) is represented with a pink bar. The type of clinical sample from which KPC-Kp was isolated is represented by colored symbols (rectal, green hexagon; urine, yellow triangle; blood, red square; bronchial aspirate, blue cross; and catheter, purple circle). Patients involved in the KPC-62-Kp outbreak are framed in the red box.
Notably, ceftazidime-avibactam consumption in the ICU increased in 2020, with a DDD/100 patient-days value rising from 0.39 in 2019 to 0.95 in 2020, resulting in a year-on-year variation of 1.41. Among the 64 ICU-admitted patients in 2020, 46 (71.9%) received ceftazidime-avibactam during their stay. In six of these patients (6/46, 13%), ceftazidime-avibactam-resistant KPC-Kp isolates were recovered during or after treatment with ceftazidime-avibactam, and in four of them, a previous KPC-3-Kp isolate susceptible to ceftazidime-avibactam (MICCZA: 1–2 mg/L) had been isolated before starting treatment. In these patients, the median days of treatment with ceftazidime-avibactam until the detection of a ceftazidime-avibactam resistant KPC-Kp isolate was 11 days (range, 5 to 24 days) (Table 1). The four patients with a ceftazidime-avibactam resistant KPC-Kp who had not been treated with ceftazidime-avibactam were admitted to the ICU between November and December and were part of a previously characterized outbreak of KPC-62-K. pneumoniae resistant to both ceftazidime-avibactam and cefiderocol14.
Between July-2020 and September-2020, 350 environmental samples were also collected from abiotic surfaces, including sinks (drain, siphon and basin) and rooms surfaces (control-breather screen and bed rails), from the ICU of our Hospital as part of the MicroCarbaFlux Project (Ref. CC23140547)15. Up to 407 K. pneumoniae isolates with a different colony morphotype and antibiotic susceptibility phenotype were recovered. During this screening, two ceftazidime-avibactam resistant KPC-Kp isolates were detected from two samples collected in a drain and a siphon from two sinks in two different ICU rooms (Table 1; Fig. 1).
Description of cases and antimicrobial susceptibility testing
Ceftazidime-avibactam KPC-Kp isolates were recovered from ten patients admitted to 8 different ICU rooms (Fig. 1). Up to six KPC variants were detected in isolates from patients, some of them in more than one patient (4 KPC-62, 2 KPC-92, 1 KPC-150, 1 KPC-66, 1 KPC-53 and 1 KPC-46). Two of these KPC variants (KPC-92 and KPC-66) were also identified in KPC-Kp isolates from sink samples. A KPC-66-Kp was collected from a drain of a sink from room 7, 44 days after the collection of a clinical KPC-53-Kp isolate from patient 2 in the same room, and 17 days after the collection of a KPC-66-Kp isolate from patient 3, in the adjacent room (room 6). A KPC-92-Kp was recovered from a siphon of a sink in room 10, 77 days after the isolation of a clinical KPC-92-Kp in patient 4 (room 2), and one day before the detection of another KPC-92-Kp in patient 6 (room 12) (Fig. 1).
Ceftazidime-avibactam resistant isolates that carried variants KPC-66 (from both patient and sink), KPC-92 (from patient), KPC-46 and KPC-53, showed collateral sensitivity to carbapenems (MICIMP ≤ 1 mg/L; MICMER ≤ 0.12 mg/L). Isolates carrying KPC-150, KPC-62 and KPC-92 (from the sink), displayed the highest MIC values to ceftazidime-avibactam (MICCZA: 16->16 mg/L), decreased susceptibility to carbapenems (MICIMP: 2–8 mg/L; MICMER: 4–16 mg/L), and were resistant to cefiderocol (MICFDC: 4->8 mg/L). Moreover, these isolates also showed increased MIC values or were eventually associated with resistance to other novel beta-lactam/beta-lactamase inhibitor combinations (Table 1).
Molecular typing and resistance gene content
All ceftazidime-avibactam resistant KPC-Kp isolates, from both patients and sinks, belonged to the HiRC ST307. An identical acquired-resistance gene content affecting different antimicrobial groups was found: β-lactams (blaCTX−M−15, blaKPC, blaOXA−1, blaOXA−9−like [W112X], blaSHV−11, blaTEM−1 A, blaTEM−1B), aminoglycosides [aac(3)-IIa, aac(6’)-Ib-cr, aac(6’)-Ib-cr, aph(3’’)-Ib, aph(6)-Id], quinolones (oqxA5, oqxB19, qnrB1), sulfonamides (sul2), fosfomycin (fosA6), chloramphenicol (catB3) and trimethoprim (dfrA14). Disinfectant resistant genes were not found.
Porin proteins suggested to be unfunctional were present in the clinical KPC-150-ST307-Kp (truncated OmpK36 and OmpK35-Q350X) and the sink KPC-92-ST307-Kp (OmpK36-Ins179_180GE, Ins287_293EVVAQYQ) isolates, both resistant to cefiderocol and cefepime-taniborbactam and with increased MIC values to other novel antimicrobial combinations. In addition, all cefiderocol resistant KPC-62-ST307-Kp isolates carried mutated OmpA (S138F) and OmpR/EnvZ (Y52C, Q103L) genes. Mutations in other genes previously found to be involved in cefiderocol resistance were not detected. The remaining KPC-ST307-Kp isolates, susceptible to carbapenems, cefiderocol and other novel combinations showed intact and identical porin proteins (Table 1). Additionally, all KPC-ST307-Kp isolates carried the conserved ParC-S80I and GyrA-S83I fluoroquinolone resistance-associated mutations.
cgMLST analysis
A cgMLST scheme was created and validated using the ST307-K. pneumoniae NCTN00000000 as the reference genome. Nine KPC-3-ST307-Kp isolates with susceptible MIC values to ceftazidime-avibactam recovered during the environmental sampling were also included in this analysis. Of a total of 5,217 loci searched, 4,655 loci were present in the genome of all analyzed KPC-ST307-Kp isolates, which diverged in distance from 0 to 23 loci. The minimum spanning tree showed that isolates from the same patient clustered together (range 0–4 loci), irrespective of whether they were susceptible (KPC-3 producers) or resistant to ceftazidime-avibactam (other KPC producers). Overall, most of the KPC-3-ST307-Kp sink isolates showed a high genetic relationship with patient isolates (≤ 10 loci). Patients were grouped according to the KPC variant they produced (KPC-92 or KPC-62), regardless of the ICU room to which they were admitted. All KPC-62-ST307-Kp resistant to both ceftazidime-avibactam and cefiderocol involved in the outbreak were grouped together and showed a low genetic distance (range 0–3 loci) (Fig. 2).
Minimum spanning tree (MST) constructed based on the cgMLST of all KPC-Kp (susceptible and resistant to ceftazidime-avibactam) collected from patients (year 2020) and from environmental samples (July-September 2020) at the ICU of our hospital. KPC-Kp isolates collected from environmental samples are indicated with green circles (totally green, susceptible to ceftazidime-avibactam; red-filled, resistant to ceftazidime-avibactam). The NLV (N locus variant) graph (green line) was calculated between patients and environmental isolates producing the same KPC variant (KPC-92-Kp and KPC-66-Kp).
The KPC-92-ST307-Kp sink isolate (recovered in room 10) was genetically related to clinical KPC-3- and KPC-92-ST307-Kp isolates from patients 6 and 4 (6 loci of difference from each other), despite being collected in different ICU rooms (room 12A and room 2, respectively). However, the KPC-66-ST307-Kp sink isolate was more genetically similar to clinical isolates recovered from patient 7 (KPC-3- and KPC-53-ST307-Kp; range, 2–4 loci), who had been admitted to the same ICU room where the environmental sample was collected (room 7), than with the clinical isolates KPC-3- and KPC-66-ST307-Kp from patient 3 (room 6) (range, 7–8 loci) (Fig. 2).
Variant calling analysis and plasmid characterization
A cgSNP based phylogenetic tree was constructed with all KPC-ST307-Kp isolates using the ST307-K. pneumoniae NCTN00000000 as the reference genome (Figure S1). A phylogenetic analysis of the KPC-92-Kp sink isolate (used as reference genome) and the KPC-3- and KPC-92-ST307-Kp clinical isolates from patients 4 and 6, respectively, was also performed using hybrid genome assemblies from the combination of short- and long-reads. The core genome ranged from 5,501,653 pb to 5,511,375pb with an evolutionary distance of 5–8 SNPs between these three isolates (Figure S2, Table S1).
Hybrid assemblies’ analysis also revealed that blaKPC−3 and blaKPC−92 were encoding on a Tn4410 copy (~ 10 Kb) located on an IncFIB/IncFII multi-replicon plasmid (~ 110–115 Kb) along with blaTEM−1 A and a truncated blaOXA−9 (W112X) (Figure S3). This plasmid shared a 95%-98% of coverage and 99.7–99.9% of identity with the pKpQIL-307 (116,499 bp; KY271403) of the ST307-Kp clone described by Villa et al.5. On the other hand, blaCTX−M−15 was found flanked by IS26, on a larger IncFIB plasmid (~ 175–239 Kb) with other antimicrobial resistance genes [aac(3)-IIa, aph(3’’)-Ib, aph(6)-Id, qnrB1, sul2, blaOXA−1 and blaTEM−1B] (Figure S4). This pCTX-M-15 plasmid shared a 77%-97% of coverage and 99.9% of identity with the previously described pKPN3-307_typeA detected in the ST307 clone (227,989 pb; KY271404)5.
Discussion
In this study, we simultaneously detected KPC-Kp isolates belonging to the ST307 clone and resistant to ceftazidime-avibactam in infected and/or colonized patients and in environmental samples from the ICU of our hospital during the early period of COVID-19 pandemic. The HiRC ST307 has been largely associated with nosocomial dissemination and outbreaks in hospitals and long-term care centers, showing a high capacity to acquire different antimicrobial genes, including KPC5,6,11. In our hospital, ST307 increased dramatically in 2018 and since then it has been mainly associated with KPC-3 between 2019 and 2020, but also with the OXA-48 enzyme in 2021 and 2020 11.
Hospital surfaces and sinks in patient rooms, surgical wards and ICUs seems to play a critical role in the transmission of multi-drug resistant (MDR) organisms15,16,17. A study performed in our hospital demonstrated the persistence and evolution of the CTX-M-15-ST307-Kp clone, associated or not with KPC and OXA-48 production, in the patient care environment of our UCI from 2019 to 2023 18. The presence of other MDR/ extremely-drug resistant (XDR) K. pneumoniae HiRCs (ST405, ST11) have also been described in the ICUs environment18,19. Long-term survival of K. pneumoniae in ICU environmental reservoirs could facilitate the horizontal gene transfer and further amplification of genes conferring resistance to first line antibiotics such as those encoding ESBLs and/or carbapenemases20,21. Our genomic analysis confirmed identical resistance genes in both clinical and environmental KPC-Kp isolates. Moreover, the sequences of the blaKPC−3/blaKPC−92- and blaCTXM−15-encoding plasmids described in this study were almost identical among them and closely related to those previously described by Villa et al.5.
A close genetic relationship was also observed by the cgMLST analysis (≤ 10 alleles) between all clinical and KPC-ST307-Kp sink isolates, highlighting the critical role that the hospital water system might play in the persistence and dissemination of this clone within our hospital. However, although the phylogenetic analysis of the KPC-92-ST307-Kp isolates supported possible transmission between patients and the environment, the directionality of spread is difficult to establish. In this sense, insufficient infection control measures and/or inappropriate patient handling during the COVID-19 pandemic could have contributed to a high cross-transmission of these isolates between both patients and sinks.
Recent research has emphasized the importance of effective cleaning protocols (rigorous hand hygiene and disinfection and regular monitoring of high-risk surfaces) and the need to reinforce strategies for reservoir identification to reduce the risk of MDR infection22,23,24. However, in 2020, the COVID-19 pandemic further exacerbated the challenges in managing MDR infections, particularly in ICU patients25. A previous study in our hospital showed that critically ill patients with COVID-19 were at an increased risk of infection with CPE and were associated with a higher mortality rate. KPC-Kp was the MDR bacteria most frequently isolated in these patients, reflecting the epidemiological situation in our hospital between 2019 and 2020 [11,13]. In the present study, up to seven patients colonized or infected with ceftazidime-avibactam resistant KPC-ST307-Kp, had COVID-19 during their stay in the ICU.
The marked increase in ceftazidime-avibactam use in 2020 (1.41-fold compared to 2019) may have contributed to the selection and emergence of resistant KPC-Kp isolates in the ICU. Note that ceftazidime-avibactam was the only novel antimicrobial available during the COVID-19 pandemic in our institution for treating CPE infections. In this context, ceftazidime-avibactam was the most frequently used to prevent and treat superinfections, primarily caused by the KPC-ST307-Kp clone, in critically ill patients with COVID-19 admitted to our hospital (33% of cases in monotherapy and in 25% of cases in combination with other antimicrobials)13. Our results support that ceftazidime-avibactam resistance likely emerged through in vivo evolution primarily driven by the selective pressure of ceftazidime-avibactam therapy, rather than by a horizontal gene transfer event such as plasmid transmission. This interpretation is further reinforced by the mutational analysis of isolates harboring KPC-92 and by our previous studies with other KPC variants9,26.
The development of resistance in patients receiving ceftazidime-avibactam treatment underscores the complexity of managing infections caused by KPC-Kp and has forced the need for therapeutic alternatives as other novel combinations. In addition, the collateral sensitivity to carbapenems observed in ceftazidime-avibactam-resistant KPC-Kp strains has also raised the possibility of reconsidering carbapenems as a therapeutic strategy27,28. Nevertheless, high-level of carbapenem resistance and co-resistance to other novel antimicrobials has also been reported associated with KPC-Kp mutants or increased blaKPC copy number usually in combination with porin loss14,29,30,31,32,33. In fact, we detected resistance to cefiderocol and cefepime-taniborbactam in patients and sink isolates that combined mutated KPCs with defects in membrane proteins. Despite increased MICs, aztreonam-avibactam, meropenem-vaborbactam, and imipenem-relebactam showed retained susceptibility, supporting their potential role as effective treatment options. Our results suggest that the high use of ceftazidime-avibactam during this period could have selected isolates already accumulating different resistance mechanisms leading to decreased susceptibility or even cross-resistance to last-line antibiotics in clinical isolates. However, we cannot elucidate which was the effect of the high use of ceftazidime-avibactam in patients may have had on the bacteria that are part of the hospital environment.
In addition, at the end of 2020 we also detected an outbreak of KPC-62-ST307-Kp resistant to both ceftazidime-avibactam and cefiderocol in four ICU patients that were not treated with these antimicrobials14. The overburdening of health resources during the COVID-19 pandemic and the ability of K. pneumoniae to survive in the patient care environment for prolonged periods may have contributed to the outbreak34,35.
In conclusion, our study highlights the high transmissibility of KPC-ST307-K. pneumoniae in the ICU of our Hospital during the COVID-19 pandemic, with the emergence of resistance to ceftazidime-avibactam and a major role of the ICU sinks as environmental reservoir(s). Our findings show that to understand the epidemiology and resistance dynamics of K. pneumoniae HiRCs in healthcare settings, and particularly within the ICUs, is critical to analyze with both microbiological and genomic studies the role of the environment aligned with continuous surveillance of both patients and environmental reservoirs to manage the threat posed by KPC-producing K. pneumoniae resistant to novel antimicrobials.
Materials and methods
Collection of samples and study design
The study included all carbapenemase-producing K. pneumoniae isolates detected in clinical or surveillance samples from patients admitted at the ICU of Hospital Universitario Ramón y Cajal during 2020. These samples were collected as part of the routine of the clinical Microbiology Department. Rectal swabs were directly seeded on ChromID-ESBL and ChromID CARBA SMART biplate selective chromogenic agar plates (bioMérieux, Marcy-l’Etoile, France) and incubated at 37 °C for 24/48 h. Clinical isolates were recovered and cultured following established protocols. Patient’s data (demographic data, length of stay, antimicrobial therapy, microbiological samples, and date of sampling) were collected from the electronic medical records. This study was conducted according to the guidelines of the Declaration of Helsinki and was approved by the Hospital Universitario Ramón y Cajal Ethics Committee (reference 293/19). All experiments were performed in accordance with relevant guidelines and regulations. Patient consent was waived because it was not required by the Ethics Committee.
To perform a comparative analysis, a subset of 350 environmental samples from the patient care environment (sink, drain, siphon) collected between July and September 2020 in the same ICU as a part of the MicroCarbaFlux project (Ref. CC23140547) was included in the study15. Samples were collected with standard sterile swabs and aspiration probes, plated onto BD CHROMagar Orientation Medium, BD CHROMagar ESBL-Biplate, and mSuperCARBA (Becton Dickinson, Franklin Lakes, USA) and incubated at 37 °C for 24/48 h. Colonies of different morphotypes (size, color, and shape) were subcultured onto BD CHROMagar Orientation Medium and incubated for 24 h at 37 °C.
Bacterial identification was performed by MALDI-TOF-MS (Bruker-Daltonics, DE) in isolates from both patient and environmental samples.
Infection control measures and intervention strategies
Following the regional guidelines, infection control measures, including contact isolation and standard precautions, are implemented after each case of CPE detection36. Our facility’s protocol includes using a 4% chlorhexidine soap for patients’ daily hygiene, emphasizing hand hygiene of healthcare workers and visitors with an alcohol-based hand rub solution, using dedicated or disposable patient-care equipment and decontamination of the patients’ environment twice daily, focusing on frequently touched surfaces and equipment in the immediate vicinity of the patient. If equipment must be used with another patient, it is cleaned and disinfected according to its risk level (non-critical or semi-critical) with a disinfectant containing benzalkonium chloride and didecyldimethylammonium or with a 2% peracetic acid solution. For daily surface cleaning and disinfection, a combined detergent–chlorine solution is used. The standard ICU infection prevention protocol for decontamination regimens was also applied in all patients requiring orotracheal intubation as previously described14.
Antimicrobial susceptibility testing
Standard broth microdilution (Sensititre-EUMDROXF) and disk diffusion method were used to antimicrobial susceptibility testing in clinical and environmental isolates, respectively15. Ceftazidime-avibactam resistant KPC-Kp isolates recovered from environmental samples were also tested by microdilution (Sensititre-EUMDROXF). Susceptibility testing results were interpreted using EUCAST-2025 criteria (https://www.eucast.org/clinical_breakpoints).
Carbapenemase production
K. pneumoniae isolates collected from patients were phenotypically confirmed as KPC producers using the KPC/MBL/OXA-48 Confirm kit (Rosco Diagnostica, Taastrup, DE), the β-CARBA test (Bio-Rad, California, US), the O.K.N.V.I. RESIST-5 (CORIS BioConcept, Gembloux, BE), and the NG-Test CARBA 5 (Hardy Diagnostics, California, USA) immunochromatography tests. In isolates recovered from environmental samples, ESBL and carbapenemase production was screened by double-disk synergy test (DDST). KPC production was confirmed by multiplex PCR (blaVIM−1, blaOXA−48, blaKPC, blaNDM, and blaGES) and Sanger sequencing as previously described37.
WGS and bioinformatics analysis
All KPC-Kp resistant to ceftazidime-avibactam from both patient and environmental samples were selected for the subsequent WGS analysis. KPC-3-Kp isolates recovered from patient samples before treatment with ceftazidime-avibactam were also included. Genomic DNA extraction was performed using the QIAamp DNA Mini Kit (Qiagen, GmbH, Hilden, Germany). Short-read sequencing was performed in all KPC-Kp isolates using Illumina NovaSeq 6000 and HiSeq4000 platforms (OGC, Oxford, UK). KPC-92-Kp and KPC-3-Kp isolates from patients 4 and 6 and the environmental KPC-92-Kp were also analysed using Oxford-Nanopore® (MinION) technology. From these strains, complete hybrid assemblies were obtained from the combination of short- and long-reads using Unicycler, Flye and Pilon tools38.
Quality control and sequence processing was carried out as previously39. Genome assemblies were annotated by Prokka40. MLST was performed in silico. Acquired antimicrobial and disinfectant resistance genes were analysed (ResFinder, CGE database, http://genepi.food.dtu.dk/resfinder). Chromosomal point mutations were also studied using PointFinder41. Mutations in OmpK35 and OmpK36 porin proteins were confirmed using PCR and sanger sequencing. Mutations in genes previously associated with cefiderocol resistance in K. pneumoniae [siderophores (fiu), TonB-dependent energy transduction complex (tonB, exbB, exbD), PBP (ftsI-pbp3, mdrA-pbp2), enterobactin or ferrochrome receptors (fhuA, fepA), efflux transporter (sugE, chrA, yicM-nepI), other (baeS/R, envZ-ompR, ompA)] were also analyzed using Blast tool. A core genome MLST (cgMLST) analysis was also performed using the pipeline chewBBACA and the ST307-K. pneumoniae NCTN00000000 as the reference genome42. A minimum spanning tree was constructed and visualized using PHYLOViZ43. Variant calling analysis was performed with Snippy, and phylogenetic trees were obtained and constructed using iqtree2 and iTOL (https://itol.embl.de/) tools, respectively44,45. Mobile genetic elements and blaKPC genetic context were characterized in the hybrid assemblies using PlasmidFinder, pMLST, Blast webtool and Proksee46,47.
Assemblies were deposited at DDBJ/ENA/GenBank under the project numbers: PRJNA741867, PRJNA985214, PRJNA747261 and PRJNA1205681.
Data availability
Sequence data were deposited at DDBJ/ENA/GenBank under the project numbers: PRJNA741867, PRJNA985214, PRJNA747261 and PRJNA1205681.
References
Rojas, L. J. et al. An analysis of the epidemic of KPC-producing Klebsiella pneumoniae: convergence of two evolutionary mechanisms creates the perfect storm. J. Infect. Dis. 217, 82–92 (2017).
Pulzova, L., Navratilova, L. & Comor, L. Alterations in outer membrane permeability favor Drug-Resistant phenotype of Klebsiella pneumoniae. Microb. Drug Resist. 23, 413–420 (2017).
Mathers, A. J., Peirano, G. & Pitout, J. D. D. The role of epidemic resistance plasmids and international high- risk clones in the spread of multidrug-resistant Enterobacteriaceae. Clin. Microbiol. Rev. 28, 565–591 (2015).
Woodford, N., Turton, J. F. & Livermore, D. M. Multiresistant Gram-negative bacteria: The role of high-risk clones in the dissemination of antibiotic resistance. FEMS Microbiology Reviews 35 736–755 Preprint at (2011). https://doi.org/10.1111/j.1574-6976.2011.00268.x
Villa, L. et al. Diversity, virulence, and antimicrobial resistance of the KPC-producing Klebsiella pneumoniae ST307 clone. Microb. Genom. 3, e000110 (2017).
Peirano, G., Chen, L., Kreiswirth, B. N. & Pitout, J. D. D. Emerging Antimicrobial-resistant High-risk Klebsiella pneumoniae clones ST307 and ST147. Antimicrob. Agent Chemother. 64, 1–14 (2020).
van Duin, D. & Bonomo, R. A. Ceftazidime/avibactam and ceftolozane/tazobactam: Second-generation β-Lactam/β-Lactamase inhibitor combinations. Clin. Infect. Dis. 63, 234–241 (2016).
Shields, R. K., Chen, L., Cheng, S., Chavda, K. D. & Press, E. G. Emergence of ceftazidime-avibactam resistance Fue to plasmid-borne blaKPC-3 mutations during treatment of carbapenem-resistant Klebsiella pneumoniae infections. Antimicrob. Agents Chemother. 61, 1–11 (2016).
Hernández-García, M. et al. Impact of ceftazidime-avibactam treatment in the emergence of novel KPC variants in the ST307-Klebsiella pneumoniae high-risk clone and consequences for their routine detection. J Clin. Microbiol 60, (2022).
Ding, L. et al. Klebsiella pneumoniae carbapenemase variants: the new threat to global public health. Clinical Microbiol. Rev. 36 Preprint at (2023). https://doi.org/10.1128/cmr.00008-23
Nieto-Torres, M et al. Characterisation of carbapenemase-producing Klebsiella pneumoniae isolates in a Hospital in Madrid (Spain) before and during COVID-19 pandemic. 33tr ECCMID congress P1487.
Ruiz-Garbajosa, P. & Cantón, R. Covid-19: impact on prescribing and antimicrobial resistance. Revista Esp. De Quimioterapia. 34, 63–68 (2021).
Pintado, V. et al. Carbapenemase-producing enterobacterales infections in COVID-19 patients. Infect. Dis. 54, 36–45 (2022).
Castillo-Polo, J. A. et al. Outbreak by KPC-62-producing ST307 Klebsiella pneumoniae isolates resistant to ceftazidime/avibactam and cefiderocol in a University Hospital in Madrid, Spain. J. Antimicrob. Chemother. 78, 1259–1264 (2023).
Aracil-Gisbert, S. et al. The ICU environment contributes to the endemicity of the Serratia marcescens complex in the hospital setting. mBio 15, (2024).
Chia, P. Y. et al. The role of hospital environment in transmissions of multidrug-resistant gram-negative organisms. Antimicrobial Resistance and Infection Control 9, (2020).
Buchan, B. W. et al. The relevance of sink proximity to toilets on the detection of Klebsiella pneumoniae carbapenemase inside sink drains. Am. J. Infect. Control. 47, 98–100 (2019).
Córdova-Espinoza, M. G. et al. Isolation and identification of multidrug-resistant Klebsiella pneumoniae clones from the hospital environment. Pathogens 12, (2023).
Qiao, F. et al. Handwashing sink contamination and carbapenem-resistant Klebsiella infection in the intensive care unit: A prospective multicenter study. Clin. Infect. Dis. 71, S379–S385 (2020).
Natalia, G. P. et al. Larga persistencia post-brotes de Klebsiella pneumoniae ST307 y sus plásmidos multirresistentes en una Unidad de Cuidados Intensivos. Congreso SEIMC 0162.
Warnes, S. L., Highmore, C. J. & Keevil, C. W. Horizontal transfer of antibiotic resistance genes on abiotic touch surfaces: Implications for public health. mBio 3, (2012).
Dancer, S. J. Controlling hospital-acquired infection: focus on the role of the environment and new technologies for decontamination. Clin. Microbiol. Rev. 27, 665–690 (2014).
Woźniak, A. et al. How to defeat multidrug-resistant bacteria in intensive care units. A lesson from the COVID-19 pandemic. Prevention, reservoirs, and implications for clinical practice. Int. J. Med. Sci. 21, 530–539 (2024).
Ziegler, M. J. et al. Stopping hospital infections with environmental services (SHINE): A Cluster-randomized trial of intensive monitoring methods for terminal room cleaning on rates of Multidrug-resistant organisms in the intensive care unit. Clin. Infect. Dis. 75, 1217–1223 (2022).
Cantón, R., Gijón, D. & Ruiz-Garbajosa, P. Antimicrobial resistance in ICUs: An update in the light of the COVID-19 pandemic. Current Opinion in Critical Care 26, 433–441 (2020).
Castillo-Polo, J. A. et al. Evolution of ceftazidime-avibactam and cefiderocol resistance in ST131-H30R1- Escherichia coli isolates with KPC-3 mutants and application of FTIR biotyping. Microbiol Spectr 12, (2024).
Cano, Á. et al. Use of carbapenems in the combined treatment of emerging ceftazidime/avibactam-resistant and carbapenem-susceptible KPC-producing Klebsiella pneumoniae infections: report of a case and review of the literature. J. Glob Antimicrob. Resist. 22, 9–12 (2020).
Pariona, J. G. M. et al. Efficacy of meropenem against ceftazidime–avibactam-resistant Klebsiella pneumoniae producing KPC-31, KPC-33, KPC-90, KPC-106 and KPC-114. J. Antimicrob. Chemother. https://doi.org/10.1093/jac/dkae389 (2024).
Blanco-Martín, T. et al. Mutant prevention concentrations, in vitro resistance evolution dynamics, and mechanisms of resistance to Imipenem and Imipenem/relebactam in carbapenem-susceptible Klebsiella pneumoniae isolates showing ceftazidime/avibactam resistance. Antimicrob. Agents Chemother. 68, e0112024 (2024).
Gaibani, P. et al. Increased blaKPC copy number and OmpK35 and OmpK36 porins disruption mediated resistance to imipenem/ relebactam and meropenem/vaborbactam in a KPC-producing Klebsiella pneumoniae clinical isolate. Antimicrob Agents Chemother 66, (2022).
Xiang, X. et al. Multiple mechanisms mediate aztreonam-avibactam resistance in Klebsiella pneumoniae: driven by KPC-2 and OmpK36 mutations. Int J. Antimicrob. Agents 65, (2025).
Bianco, G., Boattini, M., Comini, S., Gibellini, D. & Gaibani, P. In vivo emergence of ceftazidime/avibactam, cefiderocol and aztreonam/avibactam cross-resistance in a patient with KPC-producing Klebsiella pneumoniae infection after cefiderocol-based treatment. Int. J. Antimicrobial Agents 64 (2024).
Hernández-García, M. et al. Deciphering mechanisms affecting cefepime-taniborbactam in vitro activity in carbapenemase-producing enterobacterales and carbapenem-resistant Pseudomonas spp. Isolates recovered during a surveillance study in Spain. Eur. J. Clin. Microbiol. Infect. Dis. 43, 279–296 (2024).
Otter, J. A., Yezli, S., Salkeld, J. A. G. & French, G. L. Evidence that contaminated surfaces contribute to the transmission of hospital pathogens and an overview of strategies to address contaminated surfaces in hospital settings. Am. J. Infect. Control. 41, 6 (2013).
Suleyman, G., Alangaden, G. & Bardossy, A. C. The role of environmental contamination in the transmission of nosocomial pathogens and healthcare-associated infections. Current Infectious Dis. Rep. 20 (2018).
Plan De Prevención Y Control Frente a La Infección Por EPC En La Comunidad De Madrid. Versión 1. https://www.comunidad.madrid/servicios/salud/prevencion-control-infecciones-epc
Poirel, L., Walsh, T. R., Cuvillier, V. & Nordmann, P. Multiplex PCR for detection of acquired carbapenemase genes. Diagn. Microbiol. Infect. Dis. 70, 119–123 (2011).
Wick, R. R., Judd, L. M., Gorrie, C. L. & Holt, K. E. Unicycler: resolving bacterial genome assemblies from short and long sequencing reads. PLoS Comput. Biol. 13, 1–22 (2017).
Hernández-García, M. et al. In vitro activity of imipenem/relebactam against Pseudomonas aeruginosa isolates recovered from ICU patients in Spain and Portugal (SUPERIOR and STEP studies). J. Antimicrob. Chemother. 77, 3163–3172 (2022).
Seemann, T. & Prokka Rapid prokaryotic genome annotation. Bioinformatics 30, 2068–2069 (2014).
Zankari, E. et al. PointFinder: A novel web tool for WGS-based detection of antimicrobial resistance associated with chromosomal point mutations in bacterial pathogens. J. Antimicrob. Chemother. 72, 2764–2768 (2017).
Silva, M. et al. ChewBBACA: A complete suite for gene-by-gene schema creation and strain identification. Microb Genom 4, (2018).
Ribeiro-Gonçalves, B., Francisco, A. P., Vaz, C., Ramirez, M. & Carriço, J. A. PHYLOViZ online: web-based tool for visualization, phylogenetic inference, analysis and sharing of minimum spanning trees. Nucleic Acids Res. 44, W246–W251 (2016).
Nguyen, L. T., Schmidt, H. A., Von Haeseler, A. & Minh, B. Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 32, 268–274 (2015).
Letunic, I. & Bork, P. Interactive tree of life v2: online annotation and display of phylogenetic trees made easy. Nucleic Acids Res 39, (2011).
Carattoli, A. et al. In silico detection and typing of plasmids using plasmidfinder and plasmid multilocus sequence typing. Antimicrob. Agents Chemother. 58, 3895–3903 (2014).
Grant, J. R. et al. Proksee: In-depth characterization and visualization of bacterial genomes. Nucleic Acids Res. 51, W484–W492 (2023).
Acknowledgements
This study was supported by Plan Nacional de I+D+i 2013–2016 and Instituto de Salud Carlos III, Subdirección General de Redes y Centros de Investigación Cooperativa, Ministerio de Economía, Industria y Competitividad, co-financed by the European Development Regional Fund ‘A way to achieve Europe’ (ERDF), Operative program Intelligent Growth 2014–2020, IRYCIS-HURyC grants for young investigators (2021/0442) to M.H.G.; research grants PI22/01283 to P.R.G., PI21/02027 to T.C. and PI24/01449 to M.H.G.; CIBER de Enfermedades Infecciosas (CIBERINFEC) (CB21/13/00084) to R.C., Instituto de Salud Carlos III, Madrid, Spain; and Fundación Francisco Soria Melguizo (CC23140547) to T.C. M.H.G. and N.G.P. are supported by a CIBERINFEC postdoctoral (CB21/13/00084) and a predoctoral (CC23140547) contract.
Author information
Authors and Affiliations
Contributions
All authors have made substantial contributions to this work. M.H.G. has been involved in the design of the study, acquisition, analysis and interpretation of data, as well as in the writing and revising of the original manuscript. M.N.T., N.G.P. and JA.C.P. have contributed to the acquisition, data analysis and revising the manuscript. M.M. and M.P.A. have contributed to the whole genome sequencing methodology. J.S.F, C.S.C. and C.D.A. have contributed to the clinical data acquisition and revising the manuscript. T.C., P.R.G. and R.C. are responsible for the conception, design and supervision of the study, as well as revising the final version of the article. All authors have reviewed and given their approval for the publication of the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hernández-García, M., Nieto-Torres, M., Guerra-Pinto, N. et al. ICU environment as a reservoir of KPC-ST307-Klebsiella pneumoniae high-risk clone resistant to ceftazidime-avibactam. Sci Rep 15, 29486 (2025). https://doi.org/10.1038/s41598-025-14987-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-14987-w