Abstract
Ethanol is being used in spark ignition engines in Ethiopia for powering vehicles. The target of this research is to use the higher percentage of gasoline-ethanol blend E30 with a simple fuel system modification on a 4-cylinder 4-stroke engine on a vehicle and test its performance. The modified engine was tested for its performance with gasoline ethanol blend by varying its operating conditions and the data were recorded on exhaust emission, wheel torque, brake torque, bp, bsfc and thermal efficiency. The emission test designated that when the engine uses blends with large diameter main-jet with E30 the CO percentage was found to be lower than the gasoline. The maximum CO percent recorded on E30 was 2.8%, while the gasoline has 5.4% of CO. E30 has also recorded with maximum brake torque of 56 Nm at 1700 rpm, brake power of 16 kW at 3600 rpm and minimum fuel consumption of 576 gm/kWhr at 2800 rpm. When it was tested with gasoline its maximum brake torque was 56.5 Nm at 1500 rpm. Maximum brake power was 13.5 kW at 3300 rpm and minimum fuel consumption was 324 gm/kWhr 2800 rpm which is 25.2% lower than that of the gasoline-ethanol blend (E30). Therefore, the test result indicated reduced emission with a slight increase in bsfc for gasoline-ethanol blend (E30) fuel.
Similar content being viewed by others
Introduction
NOx Usage of ethanol which is renewable and sustainable fuel both in transportation and agriculture helps in reduction of the consumption of fossil fuels which are nonrenewable. When whole life cycle of ethanol production and combustion is considered, it does not emit net greenhouse gasses which cause global warming. Also the ethanol molecules contain oxygen, the addition of which to gasoline helps in complete combustion leading to reduction in emissions.
Possibility of using ethanol with purity of 80 to 95% in IC engines helps in reduction of cost involved in dehydration process which needs sophisticated systems. Simple distillation process is sufficient. Even, modification of engine is more cost effective than dehydration of ethanol. Octane number also increases with addition of ethanol to gasoline, leading to better combustion. Also, it helps in saving petroleum gasoline fuel1. This research shows the way, how alternative fuels can be used in gasoline engines and attracts the attention of the manufacturers and car owners towards the usage of ethanol blends in their cars with slight modification of fuel system. The reasons for blending ethanol into gasoline have varied over time. Ethanol can also be used as a gasoline extender. The system helps in reducing the greenhouse gas emissions. The dependency on importation of oil reduces. Reduces the requirement of adding high-octane inducing agents. Usage of ethanol, which is a locally grown fuel, revitalizes the economy of the country.
There are many sugar factories in Ethiopia which are working since long tome like Fincha, Metahara and Wonji. Fincha sugar factory produces 8 million liters of ethanol annually by using the byproduct of the sugar production process. Metahara Sugar Factory also started to produce ethanol with a target of producing 10 million liters. Another sugar factory, Tendaho Sugar Factory, which is under construction, It is also aimed at producing a much higher quantity of ethanol after its completion2. Due to its higher antiknock characteristics, it is possible to use ethanol in engines with high CR, improving thermal efficiency. In addition, ethanol’s high self-ignition temperature and high latent heat of vaporization help in increasing the thermal efficiency of the engine3.
Ethanol has inbuilt oxygen and its latent heat of vaporization making it a good fuel for IC Engines. Also, it possesses other characteristic which helps in appropriate storage and distribution4. Ethanol is less harmful compared to methanol5. Ethanol is slight odor, volatile, and colorless liquid is the properties of ethanol and burns with blue flame without smoke6,7. A 40% by volume, solution of ethanol-water can burn, when open flame is applied above 26 °C8. Complete combustion forms water vapor and carbon dioxide9. Fractional distillation of petroleum is used to produce gasoline which is a mixture of many hydrogen carbon compounds and with some additives.
Due to additives and the presence of highly volatile hydrocarbon compounds, gasoline is more volatile than other petroleum fuels. Butane’s boiling point is − 0.5 °C. By varying the quantity of butane, the volatility of the gasoline can be controlled. The volatility of the fuel can be measured by the ASTM distillation test or the Reid vapor pressure test. The volatility requirement depends on the weather conditions. During hot weather, gasoline having higher molecular weight compounds with lower volatility is required. Starting the vehicle with low-volatility fuels during cold weather is difficult. High volatility causes combustion failure due to vapor lock in hot weather10,11. Spark ignition engines are designed to burn gasoline. But in some cases, engine damage can occur due to sudden auto-ignition of the unburned mixture releasing the heat at a higher rate12. Gasoline has about 35 35 MJ/L. The energy content of the gasoline fuel varies ± 4% according to the seasons13. The variation range of specific gravity or relative gasoline density is between 0.71 and 0.77 kg/l, with higher densities and more aromatics14. These days, mostly available ethanol fuel is E-85, which is not straight ethanol. E-85 means 85% ethanol and 15% gasoline by volume. The blend is prepared usually by mixing denatured ethanol of 95% purity and ULR gasoline in required proportions15. With the emergence of electronic fuel injection, the problem of vapor lock is totally solved in modern vehicles. In this system, there exists a return line in the fuel rail. Vapor lock does not occur due to continuous recirculation of the fuel without getting hot16,17.
Gasoline fuel
Petrol, often known as gasoline, is a clear liquid made from petroleum that is mostly used as fuel in internal combustion engines. It is mostly made up of organic compounds that are improved with various additions and obtained through fractional distillation of petroleum. Ethanol is another alternative fuel included in some gasoline. In contrast to natural gas or liquefied petroleum gas, its material state is liquid under typical ambient conditions18. Because of its base ingredients as well as its additions, gasoline is more volatile than diesel oil and kerosene. Blending with butane, which boils at -0.5 °C, is a common method of controlling volatility. The Reid vapor pressure test is used to measure gasoline volatility. The ambient temperature affects the desired volatility. Higher molecular weight gasoline components with lesser volatility are utilized in hotter climates. Too little volatility causes automobiles to not start in cold weather. Excessive volatility in hot temperatures.
What is known as “vapor lock” is a situation in which the engine runs out of fuel because the liquid fuel has turned into a gas in the fuel lines, making the fuel pump useless. The purpose of spark ignition engines is to burn fuel in a regulated manner. However, the unburned mixture may occasionally auto-ignite, causing a sudden release of heat and perhaps damaging the engine. This phenomena is commonly known as end-gas knock or engine banging. Increasing the gasoline’s resistance to auto ignition, which is indicated by its octane rating, is one method of lowering knock in spark ignition engines.
When gasoline burns, a hydrocarbon is converted to carbon dioxide and water, which provides energy. The following reaction occurs when octane burns:
About 35 MJ/L (46.6 MJ/kg, or 9.7 kWh/L, 13 kWh/kg, 132 MJ/US gal, or 36.6 kWh/US gal) are found in gasoline. According to the US EPA, real energy content might vary by up to 4% depending on the producer and season due to variations in gasoline blends. Depending on the quality of the crude and the grade of gasoline, a barrel of crude oil (about 46% by volume) can yield an average of 74 L of gasoline (19.5 US gal, 16.3 imp gal).
The range of gasoline’s specific gravity, also known as its relative density, is 0.71 to 0.77 kg/l (719.7 kg/m2; 0.026 lb/in2; 6.073 lb/US gal; 7.29 lb/imp gal); higher densities correspond to a larger volume of aromatics19. Since gasoline floats on water, it is usually not possible to put out a gasoline fire with water unless it is applied in a thin mist20.
Nowadays, a blend known as E-85 is the most widely accessible ethanol fuel; it is not pure ethanol. The E in the blend stands for ethanol by volume, and the 85 for 85% ethanol and 15% gasoline. In order to achieve the E-85 blend proportions, denatured ethanol, which is an E-95 to -98 substance, is typically mixed with ULR gasoline.
Blends raise two issues: water-induced phase separation and excessively high RV vapor lock behavior. Modern electronic fuel injection eliminated vapor lock concerns for vehicle use. These have a return line and a fuel rail. Because the fuel is always circulating, it never gets hot and doesn’t vapor lock21. Ethanol-gasoline blends have been the subject of much research due to the growing interest in alternative fuels, especially in relation to lowering greenhouse gas emissions and enhancing fuel economy. Higher ethanol percentages have been demonstrated in earlier research to improve engine efficiency and lower emissions, such as when comparing E10 and E85. There is still a lack of insight into the performance traits of intermediate blends such as E30, especially in terms of their thermal efficiency and combustion stability across different tuning conditions. By examining the hydrogen storage capabilities of gasoline-ethanol mixtures, particularly in automobiles with spark ignition (SI) engines, this effort seeks to close this gap. Although the advantages of high ethanol blends demonstrated by earlier research, the effects of E30 on emissions and fuel consumption have not adequately examined. This study adds to a more sophisticated knowledge of how mid-level ethanol blends might combine performance and environmental advantages by offering a thorough examination of E3022.
The objective of this paper is to evaluate the performance of E30 fuel blends work in gasoline engines, with an emphasis on how they affect thermal efficiency and braking power output. The goal of the study is to ascertain whether making small adjustments to the carburettor may make it easier to utilize E30 and improve engine performance in comparison to ordinary gasoline.
This study is unusual since it examines the possible advantages of E30 in current gasoline engines, especially with regard to increased efficiency and lower emissions. This study helps by showing that employing greater ethanol mixtures with very minor adjustments is feasible.
Experimental scheme and methodology
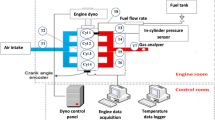
Figure 1 shows the experimental set up schematic diagram. Experiments are conducted on a vehicle with 4-cyl, 4-stroke, 8- valve, and naturally aspirated SI engine. The experiment involves the following tasks:
-
Modification of carburettor main metering jet (enlarging the jet diameter) and adjusting the float bowl to make the mixture rich to operate on higher percentage of gasoline-ethanol blend.
-
Adjusting ignition timing (timing advance),
-
Gathering data like power, tractive effort, fuel consumption, brake thermal efficiency, bp, engine torque and bsfc produced by engine running on standard gasoline under different engine speeds.
-
Gathering data like power, tractive effort, fuel consumption, brake thermal efficiency, bp, engine torque and bsfc produced by engine with ethanol-gasoline blend before and after modification, under different engine speeds.
-
Gathering data of the emission produced by the test engine running on standard gasoline under different engine speeds.
-
Gathering data of the emission produced by the engine running with ethanol- gasoline blend before and after modification, under different engine speeds.
-
The data thus obtained was tabulated and further analysed to obtain performance curves.
-
Then finally, conclusions were drawn by comparing the results obtained with pure gasoline and when it was run with ethanol-gasoline blend23.
Specification
The vehicle specifications are given in Table 1. Engine specifications are given in Table 2. Essential properties of gasoline, ethanol, and E30 are given in Table 3.
Test equipment
The experiment includes different equipment such as chassis dynamometer, tachometer, exhaust gas analyzer, and test light to adjust ignition timing, digital balance for measuring the fuel consumption. The blending of gasoline and ethanol is performed Automotive Work Shop. 30% ethanol by volume and 70% gasoline are mixed to form E30. Enlarging the main metering jet a gasoline engine runs with an A/F ratio of about 14.7/1. The jets in the carburettor are sized to provide this ratio. Whereas the air/fuel ratio of ethanol is about 9/1, to make the mixture rich when the engine is operated with a higher percentage of ethanol-gasoline blends (E30), the carburettor main metering jet must be enlarged. The whole size of the existing metering jets is determined by actually referring in a shop, manual that its manufacture number size is 125, then taking the jet of its manufacture number size is 14 which hasa larger diameter than the existing jet, existing small size 1.25 mm and enlarge jet size 1.4 mm. The engine was run with gasoline. The mass flow rate of gasoline was measured by measuring the time for the consumption of 10 gms of gasoline. A stopwatch was used to measure the time for consumption of 10 gms of gasoline at a wide-open throttle position and different speeds. The maximum load refers to the engine running at full throttle, necessitating the highest power output. This is usually indicated by the engine operating at or near its maximum RPM for a given speed. To determine the maximum load, the vehicle driven at a constant speed of 30 km/hr. The driver needs to fully press down on the accelerator pedal to guarantee that the engine gets the maximum amount of fuel and air mixture.
To maintain load during the experiments, utilizing a chassis dynamometer can replicate road conditions and provide consistent resistance. The driver should keep an eye on the tachometer to confirm that the speed stays constant while the engine is at full throttle. If there are fluctuations in speed, further adjustments will be needed to the throttle position to ensure the engine remains at maximum load. For different tests, speed will be adjusted as 30 km/hr, while still keeping the throttle fully engaged. Each speed setting should be held for a specified duration to enable precise measurements of fuel consumption and emissions. When the vehicle is being driven at 30 km/hr, 40 Km/hr the operator keeps the engine running with maximum load by fully depressing the accelerator pedal, while another person records the time for consumption of the indicated amount of gasoline. Similarly, the consumption rate of the ethanol-gasoline blend was also measured. A stopwatch was used to capture the time required to consume 10gm of the ethanol-gasoline blend (E30) at a wide-open throttle position and the speed under consideration. Based on the values obtained the specific fuel consumption can be calculated at each speed (for both pure gasoline and ethanol-gasoline blend). Emissions are compared by running the engine with standard gasoline and ethanol-gasoline blend before and after modification. This is measured by using a CO emission analyzer at 500 rpm intervals starting from 1000 rpm.
First, the exhaust emission is measured by running the engine with standard gasoline fuel and taking the result, next the exhaust emission of the blend is measured by running the engine with ethanol-gasoline blend (E30).
Adjusting float bowl
Ethanol is heavier and denser than gasoline. The float will ride higher with a certain amount of ethanol than with the same amount of gasoline. Since fuel flow is halted when the float reaches a specific point, the ethanol-gasoline level will be lower than if the float bowl were filled with pure gasoline. Restoring the fuel level to normal is therefore essential. Otherwise, the engine might cut off during turns due to a shortage of gasoline. This may be changed by simply bending the connector of the float bowl. The exact amount of linkage bending is found via trial and error. A number of parameters were tested in order to get the optimal float height.
Adjusting the ignition timing
The fuel system is done when it mixes the right amount of gasoline and air and sends the mixture to each cylinder.
The ignition system regulates the actual firing and useable power production. Its job is to ignite the fuel/air combination at a specific moment in time with the piston’s movement. Pre-ignition pinging will happen if the mixture ignites too soon, which could harm the engine. Fuel will be wasted and power would be lost if the mixture ignites too late.
A gasoline engine is set to start either immediately before or right before the piston reaches the peak of its stroke. Gasoline burns and ignites so quickly that it resembles an explosion rather than a burning. The test engine ignition timing is advanced 8 before TDC.
Ethanol burns far more evenly. It takes longer to fully evaporate and burn, and it is more difficult to ignite. It is required to “advance” (cause to fire earlier) the engine’s timing in order to accommodate this additional time when the engine is running with a greater proportion of ethanol-gasoline blend (E30). After releasing the distributor and turning it in the opposite direction of the rotor’s spin, ignition timing is used to achieve this light and test light, the E30’s ignition timing is set 10 min ahead of TDC.
The equivalence ratio defined as the comparison of the actual fuel-to-air ratio to the stoichiometric fuel-to-air ratio. It is essential for evaluating combustion efficiency and emissions.
Measurement and Control Instrumentation: Lambda Sensors, Lambda values were determined using wideband oxygen sensors that can offer real-time feedback on the air-fuel mixture. Data Acquisition Systems, These systems collected lambda values alongside engine parameters during experiments.
Lambda trends in results
Ethanol blends: E30 configurations generally operated at slightly richer lambda values (Φ < 1) than gasoline to enhance performance and reduce knocking.
Performance observation, Improvements in torque and power were observed as lambda values decreased (richer mixtures), especially with the larger main jet configuration.
Measurement of tractive effort (force) and power
Driving a car on a chassis dynamometer at various test speeds allows one to assess the tractive effort (force) wheel power. Starting at 25 km/h, the test speed is chosen at intervals of 5 km/h. This is the lowest speed at which the Sun Road-a-matic XII chassis dynamometer may be tested.
Quantification of fuel usage
Both when the engine runs on gasoline and when it runs on E30, the fuel usage is measured.
The test engine is powered by regular gasoline.
By timing how long it took to consume 10 g of gasoline, the mass flow rate f was measured. The time needed to consume 10 g of liquid gasoline at wide open throttle position and the speed under consideration were recorded using a stop watch.
The operator fully depresses the accelerator pedal to maintain the engine running at maximum load when driving a test car at 30 km/h. Another person keeps track of how long it takes to utilize the allotted quantity of fuel in the meanwhile.
The test engine is powered by an E30 ethanol-gasoline combination.
By timing how long it took to drink 10 g of ethanol-gasoline blend, the mass flow rate of the blend was measured. The time needed to consume 10 g of ethanol-gasoline blend (E30) at wide open throttle position and the speed under consideration were recorded using a stop watch.
The precise fuel usage at each speed (for both pure gasoline and ethanol-gasoline blend) can be computed using the numbers acquired.
Measurement of exhaust emission
The main objective of emission test is to compare engine emission result by running the engine with standard gasoline and running with ethanol- gasoline blend before and after modification. This is measured by using CO emission analyzer at 500 rpm intervals starting from 1000 rpm.
First the exhaust emission is measured by running the engine with standard gasoline fuel and takes the result, next the exhaust emission of the blend is measured by running the engine with ethanol-gasoline blend (E30).
Ethanol’s charge-cooling effect
Ethanol displays a notable charge-cooling effect because it has a higher latent heat of vaporization compared to gasoline. This feature allows ethanol to absorb more heat during the vaporization process, resulting in a cooler intake charge. Such a cooling effect can significantly influence engine performance, knocking resistance, and combustion timing. In terms of knock resistance, the charge-cooling effect helps reduce the temperature of the incoming air-fuel mixture, decreasing the likelihood of pre-ignition and knocking. Moreover, ethanol’s higher octane rating also enhances its resistance to knocking. When it comes to combustion stability, a cooler intake charge can lead to a more reliable combustion process, permitting the use of higher compression ratios and more aggressive ignition timings without the risk of knocking. In analyzing this, the following aspects of ethanol’s charge-cooling effect examined,
-
Performance with Various Jet Sizes: The effects of the charge-cooling effect may explain why E30 configurations, particularly with the larger main jet, attained better thermal efficiency, as the cooler intake mixture can enhance combustion efficiency.
-
Tuning Considerations: The analysis shows that optimizing the engine to harness ethanol’s characteristics, including the charge-cooling effect, can improve performance and efficiency. This demonstrated by the differences in BSFC noted across the different fuel mixtures and jet configurations.
Uncertainty and error analysis
The measurements were performed using a chassis dynamometer to gauge torque with an accuracy of ± 1 Nm and an uncertainty of 2%. A digital tachometer was utilized to record engine speed, providing an accuracy of ± 10 rpm and an uncertainty of 1.5%. Fuel consumption was measured with a digital scale, which had an accuracy of ± 0.1 g and an uncertainty of 1%. Additionally, a gas analyzer was employed to evaluate CO emissions, offering an accuracy of ± 0.05% and an uncertainty of 1.5%. The combined uncertainty u for each measured quantity was calculated using the root-sum-square (RSS)method.The combined uncertainty u for each measured quantity was calculated using the root-sum-square (RSS)method.
Error bars were included in Figs. 14, 15, 16, 17, 18, 19, 20, 21 and 22 to reflect these uncertainties.
Results and discussion
Carbon monoxide (CO) emission
CO which is lighter than air is a colourless, odourless, and tasteless gas. CO is dangerous to animals and human beings if exposed to this gas in higher concentrations. Petroleum fuels with no oxygen in their molecular structure produce CO due to an incomplete combustion process.
The engine rpm is measured by using a tachometer and the % of CO versus engine speed graph is shown in Fig. 2. From Fig. 2, it is shown that gasoline CO emission is higher at lower engine speeds. At 1000 rpm CO emission is 4.4%. At lower engine speed the air-fuel mixture becomes rich. That means the amount of oxygen in the air-fuel mixture is less, so there is not enough oxygen for full combustion and conversion to CO2. At 1500 rpm the CO emission slightly drops to 3.6%. It shows that at 1500 rpm there is good charge mixing and better combustion which is why the CO emission becomes reduced at this engine speed.
As engine speed increases to about 2000 rpm and above the CO emission becomes increased to a higher percentage as the curve in Fig. 2 shows. This is because as engine rpm increases with full load acceleration under a wide-open throttle valve the carburettor acceleration and power system starts supplying additional fuel to the incoming air this causes the mixture to become richer and the amount of oxygen becomes lower than the fuel supplied by the system. Therefore, as engine speed increases there is not enough oxygen to convert all carbon to CO2 and because of this, the CO emission becomes increases. In another case as engine speed increases, the duration for intake valve opening becomes decreases which means the intake valve closes before enough oxygen enter the cylinder; hence, there is not enough oxygen to convert all carbon to CO2. So the amount of CO emission gets increased.
At lower engine speed as in Fig. 2, the curve shows CO emission is high. The CO emission is 2.1%at 1000 rpm and slightly decreases to 1.6% at around 1500 rpm (for larger size jet).When engine speed increases, the CO emission is slightly increased to 2.8% at about 3000 rpm. This is because as engine speed increases as it is tried to explain for gasoline CO emission under higher engine speed, with full load acceleration under wide-open throttle valve the carburettor acceleration and power system starts supplying additional fuel to the incoming air this causes the mixture to become richer and the amount of oxygen becomes lower than the fuel supplied by the system. So even if ethanol is an oxygenated fuel, as engine speed increases there is not enough oxygen to convert all carbon to CO2, and because of this the E30 CO emission becomes increases slightly.
Even if CO emission is considered in both when the engine is running with gasoline and E30 (70% gasoline and 30% ethanol) as shown in Fig. 2, there is a higher difference in the CO emission. The E30, CO emission is much lower when compared to the amount of CO when the engine runs with pure gasoline fuel at each engine speed. This is because ethanol fuels an oxygenated fuel; it contains oxygen in its molecular structure. Since E30 contains 30% ethanol, it causes the combustion complete as much as possible.
Brake torque (Tb) and brake power (Pb)
The resistance offered to the Engine by the pole/Fly wheel against turn or measure of constrain required to stop a pivoting shaft/fly of range “r’ is named as Brake Torque additionally assigned as Load the units typically utilized for Brake Torque or Load are Nm. We require a measure of Force “N” Newton’s to pivot the pole, Pulley or Fly wheel of Radius “m”. The term Brake shows the estimation technique for Torque utilizing Brake framework. The Brake Torque increments with speed up to a most extreme esteem named as Max Brake Torque then it gets lower as at higher speeds as it gets to be distinctly troublesome for Engine to consume a full Charge of air.
Brake power: - The measure of mechanical work “w” in time “t” accessible at the yield shaft of Diesel Engine is called Brake Power. Ordinarily Given as kW different units is Horse power and kilo Watt hours. Brake Power relies on upon Brake torque and Engine Speed (rpm). Brake is lower at lower speeds where as it gets on higher at speed increments. It gets bring down after the temperate range at rapid of 6000 rpm where the torque diminishes significantly due lesser admission of air/fuel blend.
Engine dynamometers and rolling road or chassis dynamometers are the two varieties of dynamometers. For this project, a Sun Road-a matic-XII chassis dynamometer was utilized. Both the power production and the tractive effort given to the drive wheels can be measured by it. If the drive train gear ratios and transmission losses are known, the power available at the engine crankshaft, also known as brake power, can be computed.
Tractive effort is the force available between there are wheel tyres and the road. It is also tested by a chassis dynamometer. The tractive effort obtained from the chassis dynamometer test for both standard gasoline and ethanol-gasoline blend is presented in Table 4.
When running on gasoline the maximum Pb is 13.5 kW at 3300 rpm. A maximum Pb of 16 kW is obtained at 3600 rpm when running on E30 (with larger size main metering jet) and12.5Kw at 4800 rpm is obtained when running on E30 with smaller main metering jet. At lower engine speed the friction power is relatively low and hence the brake power increases with speed. As engine speed increases, the friction power at continuously greater rate and Therefore Pb reaches a peak and starts reducing. The reason for the decreasing of brake power at higher engine speed is less complete filling of the cylinder23,24.
Brake specific fuel consumption (BSFC)
Brake specific fuel consumption (BSFC), which measures the amount of gasoline used per unit of braking power produced, is an essential metric for evaluating the efficiency of diesel engines. Due to its direct correlation with fuel efficiency, this component is essential to diesel engine design. Given the current energy management concerns, fuel economy is essential for enhancing engine performance and sustainability. Lower BSFC values indicate greater engine efficiency, which measures how well an engine transforms fuel into useable power. Whereas BSFC explicitly indicates the fuel consumption per unit of braking power, specific fuel consumption is the measurement of fuel utilized over a given period. Manufacturers can increase the overall efficiency of diesel engines and help with energy management and environmental impact reduction by concentrating on lowering BSFC. To create diesel engines that are more inexpensive and efficient, it is essential to comprehend and optimize BSFC25.
Brake torque (Tb)
Brake Torque (Tb) is the torque available at the Flywheel‘. After wheel torque at the wheel is known the brake torque. For torque at the tire surface transmission efficiency is 0.9 at the top gear and 0.8 at other gears can be assumed. The test vehicle uses manual gear box type K40. It is a 4 speed transmission. While gasoline achieves 57 Nm at about 1550 rpm, the maximum engine brake torque for E30 fuel is 57.5 Nm at 1750 rpm. Figures 3 and 4 show that the efficiency decreases as heat dissipation affects performance, highlighting the relationship between engine speed, torque output, and thermal management. Brake torque is lower at lower engine speeds due to increased heat loss, which occurs because more heat is rejected to the engine coolant at lower speeds, resulting in a longer duration for heat conduction to engine components.
Additionally, at lower engine speeds, the engine rejects a large amount of heat to the coolant, which lowers the amount of heat that can be used for work. As a result, the brake torque output is reduced. A more efficient combustion process causes the brake torque to increase initially as engine speed increases, but it subsequently decreases at higher speeds. As the amount of heat rejected to the coolant diminishes, the braking torque increases at moderate speeds. As engine speed increases, more heat can stay in the engine for work since there is less time for heat to condense to the coolant. At extremely high speeds, however, other elements like friction and pumping losses may cause the torque to decrease. In order to maximize performance and efficiency under a variety of operating conditions, this connection emphasizes how crucial it is to balance engine speed and temperature control.
There are a number of reasons why brake torque decreases with increasing engine speed. First, the intake valve is open for a much shorter period of time at higher speeds. This causes incomplete combustion and a subsequent decrease in brake torque because the intake valve closes before the cylinder is completely filled with the air-fuel mixture. Additionally, the exhaust valve opens earlier in the cycle as engine speed rises. More heat is lost as a result of this early opening since more heat can escape with the exhaust fumes. At faster speeds, braking torque decreases due to a combination of a shorter air-fuel intake time and a greater amount of heat loss through the exhaust. The significance of improving valve timing and heat management to improve engine performance under a range of operating circumstances is underscored by these dynamics. Enhancing engine production and efficiency requires an understanding of these relationships.
Figure 5 shows that when running on gasoline the maximum brake power is 13.5 kW at 3300 rpm. Figure 6 reveals maximum brake power of 16 kW is obtained at 3600 rpm when running on E30 (with larger size main metering jet) and 12.5 kW at 4800 rpm is obtained when running on E30 with smaller main metering jet. The braking power increases with speed because the friction power is comparatively low at lower engine speeds. Pb hits a high and begins to decrease as engine speed rises because friction power grows steadily. The ratio of the engine’s fuel consumption in grams per hour (gm/hr) to its brake power in kW is known as brake specific fuel consumption. For calculating the amount of ethanol gasoline blend (E30), the amount of time it took to consume 10 g of E30 was recorded. By dividing 10 g by the amount of time needed to consume the ethanol fuel, one can determine the mass flow per unit time. At lower engine speed as Figs. 7 and 8 shows, the brake specific fuel consumption is relatively high. This is because at lower engine speed fuel is intentionally made rich. Hence there is not complete combustion and unburned fuel exits with the exhaust gas.
Brake specific fuel consumption of both gasoline and gasoline-ethanol blend E30 are decreasing as engine speed Therefore we conclude that brake specific fuel consumption rate is higher at low speed increases a little for medium speeds and increases more at high speeds. This is because at low and high speeds a rich mixture is required. Increases to some extent, and then increasing at high speed, In Figs. 7 and 8 the minimum fuel consumption for gasoline is 324gm/kWh at 2800 rpm and for E30 (with larger main jet) is 576gm/kWh at 2800 rpm and the minimum brake specific fuel consumption ofE30 with small size jet is 500gm/kWhr at 3000 rpm. At higher engine speeds the consumption rises, because the frictional loses of the engine rapidly and so the energy of combustion is again Bing wasted. There as on that bsfc is lowest at middle rpm, because the engine stand to develop best cylinder filling at middle speed, the engine‘s breathings at highest efficiency at these speeds.
The time it took to consume 10 g of fuel was recorded in order to measure gasoline consumption. By dividing 10 g by the amount of time needed to consume it, one can determine the mass flow rate of gasoline consumed per unit of time. The time it took to eat 10 g of the ethanol-gasoline blend (E30) was recorded in order to measure its consumption. By dividing 10 g by the amount of time needed to consume it, one can determine the mass flow rate of ethanol-gasoline consumed. E30 bsfc (for smaller and larger main jet) at lower engine speed, the bsfc is relatively high. This is because at lower engine speed fuel is intentionally made rich. Hence there is no complete combustion and unburned fuel exits with the exhaust gas. Bsfc of both gasoline and gasoline-ethanol blend E30 are decreasing as engine speed increases to some extent, and then increases at high speed.
Brake thermal efficiency
Brake thermal efficiency shows how efficiently fuel is used in the engine. The lower calorific value of both E30 and gasoline fuel is taken from the literature, 42.5 MJ/kg for gasoline and 41.47 MJ/kg for E30. The brake thermal efficiency of gasoline and E30 are given the maximum brake thermal efficiency for small size jet before modification, the maximum brake thermal efficiency is16% at 3800 rpm and the minimum thermal efficiency is 11% at 5600 rpm (Fig. 9). When the engine run son gasoline is 27% at 3200 rpm and the minimum brake thermal efficiency is 14% at 5600 rpm (Fig. 10). The maximum brake thermal efficiency, when the engine runs onE30 (after modification) is 18% at 3600 rpm and the minimum brake thermal efficiency is 10% at 5600 rpm. It is that at lower speeds the brake thermal efficiency is low because of thermal loss. At lower engine speed, there is sufficient time for the heat to dissipate to engine parts. This causes the brake thermal efficiency to decrease at lower engine speed. The brake thermal efficiency is also decreasing at maximum engine speed this is because; at maximum engine speed the exhaust valve opens and closes rapidly, this causes thermal loss. Hence brake thermal efficiency is reduced at higher engine speed. The maximum brake thermal efficiency, when the engine runs on gasoline is 27% at 3200 rpm and the minimum brake thermal efficiencyis14%at 5600 rpm. For small size jet before modification the maximum brake thermal efficiency is 16% at 3800 rpm and the minimum thermal efficiency is 11% at 5600 rpm. The maximum brake thermal efficiency, when engine runs on E30 (after modification) is18% at 3600 rpm.
Minimum brake thermal efficiency is 10% at 5600 rpm. From Figs. 9 and 10 it is shown that at lower speeds the brake thermal efficiency is low because of thermal loss. At lower engine speed the longer hot air spends in the cylinder, the more time it has to heat the engine parts. This causes the brake thermal efficiency to decrease at lower engine speed. The brake thermal efficiency is also decreasing at maximum engine speed this is because at maximum engine speed the exhaust valve open and close rapidly, this causes thermal loss. Hence brake thermal efficiency reduced at higher engine speed.
Figures 14, 15, 16, 17, 18, 19, 20, 21 and 22 present a comprehensive comparison of engine performance metrics across gasoline and E30 fuel blends (with both small and large main jets), each plotted against engine speed (RPM) and accompanied by error bars to represent experimental uncertainty: Fig. 14 shows CO emissions with ± 0.2% uncertainty; Figs. 15 and 16 compare brake torque (± 1.5 Nm) for gasoline and E30 blends; Figs. 17 and 18 display brake power (± 0.5 kW); Figs. 19 and 20 illustrate brake-specific fuel consumption (± 15 g/kWh); and Figs. 21 and 22 depict brake thermal efficiency (± 1%), highlighting measurement precision across all conditions.
Conclusion
This study is to investigate experimentally the fuel system of a gasoline engine to use higher blends of Ethanol gasoline (E30) while deriving the benefits of ethanol as a fuel and evaluating its performance and comparing with the engine running on pure gasoline. Based on the data obtained from the experiment the following conclusions can be summarized from the above analysis are drawn for different characteristics. As it can be seen in Fig. 2, depending on emission characteristics gasoline-ethanol blend E30 is much better than gasoline at every engine speed. Hence better in reducing the environmental pollution. Depending on brake torque as shown in Figs. 11 and 13, E30 is as good as gasoline. Hence it is possible to use E30 in place of gasoline fuel with only a simple modification.
As indicated in Figs. 11 and 12, depending on brake power performance characteristics, E30 generates higher brake power than gasoline after a simple modification performed on the carburettor. Consequently, it is possible to use E30 on gasoline engines with only minor modifications. Depending on bsfc as indicated in Figs. 11 and 12, gasoline has lower bsfc than E30. Even if the bsfc of gasoline is lower than E30, it is better to use E30 since ethanol is homegrown and simple to produce. In Figs. 12 and 13, E30 have higher brake thermal efficiency at higher engine speed than gasoline, and in Table 5. Efficiency Analysis E30 equipped with the Larger Main Jet (0.007743 MJ/kWh) demonstrates the lowest energy basis BSFC, suggesting it is the most effective in converting fuel energy into power. E30 with the Smaller Main Jet (0.008267 MJ/kWh) shows slightly reduced efficiency but still outperforms gasoline. Gasoline (0.008438 MJ/kWh) exhibits the highest energy basis BSFC, indicating it is less efficient compared to both variants of E30. hence it is better to use E30 on an engine designed for gasoline.
According to this study, E30 fuel blends have the potential to significantly boost gasoline engines’ braking power output, especially when combined with minor carburetor changes. In addition to boosting braking power, E30 exhibits superior thermal efficiency at higher engine speeds as compared to regular gasoline. These findings suggest that engine designs might be changed to accommodate larger ethanol mixes, allowing for higher compression ratios and improved combustion efficiency. Manufacturers may alter current gasoline engines to run on E30, which might lead to improved efficiency and reduced pollution. From a policy perspective, promoting E30 as a sustainable alternative to gasoline aligns with goals for energy independence and the environment. Encouragement of E30 use can reduce reliance on fossil fuels and benefit local farmers. Future research should examine the effects of varying ethanol concentrations on engine performance as well as the long-term effects of E30 on emissions and engine longevity. The focus on certain engine configurations in the current study is one of its drawbacks; this highlights the need for more thorough research including a variety of engine types and real-world circumstances.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Khan, K. H. & Mia, S. Experimental Investigation on Use of Ethanol-Gasoline Blend as Alternate Fuel on SI Engine Performance (SSRN, 2024).
Bashir, M. S. & Khan, M. A. Performance and emission characteristics of ethanol-gasoline blends in a spark ignition engine. Energy Rep. (2023).
Almeida, F. M. & Silva, A. R. The influence of ethanol blending on the performance of SI engine: A review renewable energy (2022).
Zhang, Y. & Wang, J. Experimental study on the combustion characteristics of ethanol-gasoline blends in SI engine. Fuel (2021).
Kumar, A. & Singh, R. Performance evaluation of ethanol-gasoline blends in spark engine. J. Clean. Prod. (2020).
Mishra, S. & Gupta, A. Impact of ethanol blending on the performance and emissions of SI engine: An experimental study. Energy Convers. Manag. (2023).
Mishra, S. & Gupta, A. Impact of ethanol blending on the performance and emissions of SI engines: An experimental study. Energy Convers. Manag. (2023).
Patel, R. & Desai, S. Experimental investigation of ethanol-gasoline blends in SI engine: Performance and emission analysis. Appl. Energy (2020).
Reddy, K. S. & Kumar, P. The effect of ethanol on the performance of spark ignition engines: A comprehensive review. Renew. Sustain. Energy Rev. (2021).
Sahu, A. & Mohanty, S. Experimental analysis of ethanol-gasoline blends in si engine: performance and emission characteristics. Fuel Process. Technol. (2020).
Choudhary, A. & Sharma, R. Performance and emission characteristics of ethanol-gasoline blends in spark ignition engines. J. Energy Resour. Technol. (2023).
Sing, S. & Kumar, A. Experimental investigation of ethanol-gasoline blends in Si engines: A review of recent advanced. Energy Rep. (2022).
Patil, S. & Jadhav, S. The role of ethanol in enhancing the performance of spark ignition engines: An experimental study. J. Fuel Chem. Technol. (2021).
Verma, A. & Gupta, R. Performance evaluation of ethanol-gasoline blends in a spark ignition engines: An experimental approach. Energy Sources (2020).
Khan, M. A. & Bashir, M. S. Ethanol-gasoline blends: A sustainable alternative for sparking ignition engines. Sustain. Energy Technol. Assess. (2023).
Rao, P. S. & Reddy, K. R. Experimental investigation of the effects of ethanol blending on SI engine performance. Int. J. Automot. Eng. (2022).
Nayak, S. B. & Mohanty, S. Performance and emission characteristics of ethanol-gasoline blends in spark ignition engines: An experimental study. Energy Rep. (2021).
Kumar, R. & Singh, P. The impact of ethanol on the performance of spark ignition engines: A review. Renew. Energy (2020).
Global Partners, L. P. E85 Ethanol/Gasoline Blend - Safety Data Sheet (Retrieved from Global Partners, 2023).
Johnson, M. Fire safety in fuel handling. Saf. Sci. Rev. 39(1), 45–58 (2022).
Smith, L. The density and composition of gasoline. Fuel Sci. J. 12(3), 78–85 (2021).
Badawy, T. et al. Performance and emissions evaluation of a multi-cylinder research engine fueled with ethanol, methanol, gasoline Euro-6, E85, and iso-stoichiometric ternary GEM mixtures operated at lean conditions. Fuel 363, 130962. https://doi.org/10.1016/j.fuel.2024.130962 (2024).
Sahu, P. & Mohanty, A. Experimental Investigation of ethanol-gasoline Blends in SI Engines: Performance and Emission Analysis. J. Clean. Prod. (2023).
Choudhary, R. & Sharma, A. The effects of ethanol bending on SI engine performance: An experimental study. Fuel (2022).
Patel, A. & Desai, R. Experimental study on the performance of ethanol-gasoline blends in spark ignition engines. Energy Convers. Manag. (2021).
Acknowledgements
The authors would like to express their heartfelt gratitude to Hawassa University for their invaluable support in conducting this research work.
Author information
Authors and Affiliations
Contributions
D.T. Conceptualization, Methodology, Software, Validation, Formal Analysis, Investigation, Data Curation, Writing - Original Draft.R.B.N. Conceptualization, Methodology, Validation, Writing-Reviewing and Editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tafesse, D., Nallamothu, R.B. Experimental investigation on impact of fuel jet size on performance of gasoline-ethanol blend in a SI engine vehicle. Sci Rep 15, 30092 (2025). https://doi.org/10.1038/s41598-025-15717-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-15717-y