Abstract
Inappropriate prescribing of medicines could result in adverse drug reactions and interactions thereby leading to morbidity and mortality. Potential drug-drug interactions are preventable drug-related problems with the risks of hospitalization, adverse effects, and treatment failure. This study aimed to assess medication utilization patterns, and potential drug-drug interactions and its determinants among outpatient prescriptions. A cross-sectional survey was conducted. The study included dispensed outpatient prescriptions in six community chain pharmacies in Asmara, Eritrea, between June 16 and July 16, 2023. A stratified random sampling was utilized and drug interactions were screened using Lexi-comp drug interaction checker. Logistic regression was used to assess the determinants of clinically significant potential drug-drug interactions using IBM SPSS (version-26.0). A total of 417 outpatient prescriptions were included in the study. Moreover, a total of 830 medicines were prescribed from the 417 dispensed outpatient prescriptions. The most commonly prescribed medicines were omeprazole (8.4%) and amoxicillin (8.1%). Out of 267 prescriptions containing two or more medicines, the prevalence of clinically significant pDDIs was 29.2% (95%CI: 23.7, 34.7). The most common medicines involved in clinically significant pDDIs were diclofenac (22.6%) and ciprofloxacin (10.9%). Adult population (AOR = 4.23, 95%CI: 1.49, 12.01), older adult population (AOR = 7.09, 95%CI: 2.24, 22.44), number of drugs prescribed (AOR = 2.27, 95%CI: 1.53, 3.37), and tertiary level health facility (AOR = 9.13, 95%CI: 1.17, 71.42) were significant determinants of clinically significant pDDIs. Clinically significant pDDIs were detected in considerable number of outpatient prescriptions requiring strict monitoring from healthcare professionals, policy makers, and program managers.
Similar content being viewed by others
Introduction
Medicines play an essential role in healthcare delivery and disease prevention. The availability and affordability of good quality medicines, along with their rational use, are needed for effective healthcare. However, inappropriate prescribing of medicines is a global burden that could result in drug resistance, adverse drug reactions and interactions thereby leading to morbidity and mortality1. Drug-drug interactions (DDIs) are defined as two or more drugs interacting to a magnitude that could change the effectiveness of the drugs2. Drug interactions can occur in several different ways basically, at pharmacokinetic, pharmacodynamics, and pharmaceutical interactions with pharmacokinetic interactions being the most common one3,4.
Drug interactions are a significant cause of adverse drug reactions (ADRs)5. DDIs may cause clinically significant life-threatening conditions resulting in increased duration of hospital stay and extra health-related expenses associated with it2,5. DDIs in an outpatient setting account for approximately two in five ADRs6. Moreover, they are responsible for 17% of all avoidable adverse drug events among hospitalized patients 5 and 44% of all drug-related deaths7.
Multiple use of drug treatments may further put patients at a greater risk of DDIs with serious consequences for health2. However, at times polypharmacy is inevitable, the potential benefits are weighed against the risk of clinically significant DDIs, considering other alternative choices8. Numerous studies have reported the prevalence of potential DDIs ranging from 5–91.6%2,8,9,10. Factors associated with the occurrence of potential DDIs were age, sex, healthcare setting, polypharmacy, presence of comorbidity, and disease state9,11.
Eritrea, like many other developing countries, faces numerous challenges related to the surveillance of DDIs. The absence of DDI screening tools, a relatively high number of prescriptions dispensed in community chain pharmacies compared to private community pharmacy, and a lack of documentation of medication histories are the main issues hindering the prevention of DDIs. Given these challenges, it is crucial to conduct a study on potential DDIs in outpatient pharmacy settings. A previous study in Eritrea revealed a significant number of potential drug-drug interactions involving non-steroidal anti-inflammatory drugs among the older population12. However, there is a lack of data on the prevalence of clinically significant drug-drug interactions and its determinants, and medication utilization pattern in the general population of Eritrea. Understanding medication utilization patterns can help healthcare professionals be aware of commonly prescribed medications, make informed decisions about patient care, ensure appropriate prescribing, and monitor for potential drug-drug interactions. Therefore, the study aimed to assess medication utilization patterns and potential drug-drug interactions and its determinants among outpatient prescriptions in six community chain pharmacies in Asmara, Eritrea.
Materials and methods
Study design and setting
A cross-sectional study was conducted in six community chain pharmacies in Asmara, Eritrea. A total of thirteen community chain pharmacies, owned and directed by the Ministry of Health, are available in Eritrea. They are distributed across five of the six regions in the country. Moreover, six of the thirteen community chain pharmacies are located in Asmara (the capital city of Eritrea). Two of the six community chain pharmacies are located inside Orotta National Referral and Teaching Hospital and Halibet National Referral Hospital. The community chain pharmacies are authorized to sell over-the-counter medicines and dispense prescription-only medicines upon receiving a legal prescription. They implement quality assurance measures to ensure safe and effective medication dispensing, monitor adverse drug reactions, and maintain high standards of patient care. Due to the availability and affordability of quality medicines, the majority of patients who cannot get their medicines in the outpatient department (OPD) pharmacies of healthcare facilities prefer to take their medicines from the community chain pharmacies. Data were collected between June 16 and July 16, 2023.
Target population
Outpatient prescriptions, regardless of age and sex, dispensed in the study sites during the study period formed the study population.
Sample size determination
The sample size was computed by considering the finite population correction factor: n ≥ NZ2pq/ [pqZ2 + (N-1)d2]. The total sample size (n) was calculated using the following assumptions: the expected proportion of outpatient prescriptions with drug interactions (p) and those prescriptions without drug interactions (q) were taken as 0.5, Z statistic for 95% level of confidence (Z = 1.96), estimated population size (N) of 21,280, margin of error (d) of 0.05 and 10% non-response rate. Considering the above assumptions, to have an optimum sample, the least required number was 415.
Sampling design and allocation
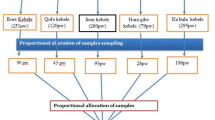
To achieve representative samples from each community chain pharmacy, stratified random sampling was utilized. The six community pharmacies were considered as separate strata, and samples were systematically selected from each one. The calculated sample size was allocated proportionally among the community pharmacies, ensuring that each location’s contribution reflected its dispensing volume. Ultimately, a total of 417 prescriptions were collected for the analysis.
Data collection tool and DDI screening
A structured data collection tool was used to collect data from outpatient prescriptions. The data collection tool was prepared upon a review of questionnaires from similar studies8,12,13. It comprised of three sections. The first section inquired about the demographic characteristics of patients, level of health facility, and prescriber qualification. The second section was designed to capture information on medicine prescription patterns. The World Health Organization’s (WHO) Anatomic Therapeutic Chemical (ATC) classification was used to group the prescribed medicines. Finally, the third section was intended to capture the names of the prescribed medicines for the detection of drug-drug interactions using the Lexi-comp® drug interaction checker. Lexi-comp® drug interaction checker was shown to have the most accurate and reliable drug interaction checker in detecting potential DDIs among other databases14.
Outpatient prescriptions which contain two or more medicines were screened for potential DDIs. Lexi-comp® drug interaction checker provides information on risk rating, severity, and clinical management of drug interactions15. Based on the risk rating of interactions, DDIs are categorized as A, B, C, D, and X. Risk category ‘A’ indicates that no evidence of pharmacodynamic or pharmacokinetic drug interactions, while risk category ‘B’ denotes the existence of little to no evidence of drug interactions15. Moreover, no action is needed for categories A and B and therefore of no clinical attention. Therapy monitoring and dosage adjustments of one or more medicines are required for risk category ‘C’ as there is evidence of drug interactions. However, the benefits usually outweigh the risks. In risk category ‘D’, therapy modification that includes, the selection of alternative agents, aggressive monitoring, and empiric dosage changes, is to be considered to realize the benefits and/or minimize toxicity. The risks associated with drug interactions in risk category ‘X’ usually outweigh the benefits, therefore they are contraindicated15. Risk categories C, D, and X were considered clinically significant potential drug-drug interactions (pDDIs) in this study.
Furthermore, based on severity, drug interactions are categorized as minor, moderate, and major15. Major interaction is highly clinically significant and can lead to life-threatening conditions, whereas moderate interaction is of moderate significance resulting in deterioration of the patient’s condition unless therapy monitoring is exercised. Minor interaction is minimal in clinical relevance which did not require alteration in therapy.
Variables
Clinically significant potential drug-drug interactions were considered as a dependent variable and the independent variables were age and sex of outpatients, number of drugs prescribed, level of health facility, and prescriber qualification.
Quality assurance
Prior to the main survey, the data collection tool underwent evaluation by a panel of five experts in medicine, public health, pharmacy, epidemiology, and pharmacoepidemiology to assess face and content validity. Based on their feedback, modifications were made to refine the tool, enhancing question clarity and relevance. Following these changes, a pre-test was conducted using 30 outpatient prescriptions randomly selected from a community chain pharmacy to evaluate the tool’s applicability and the Lexi-comp interaction checker. The pre-test revealed ambiguities, leading to further adjustments in phrasing and structure. After analyzing pre-test results, additional revisions were implemented to improve usability. Finally, experienced data collectors were selected to ensure reliability in data gathering. This systematic process ensured that the data collection tool was valid and reliable for assessing medication utilization patterns and potential drug-drug interactions. Each stage aimed to enhance the overall quality of the data collected. The study was reported in line with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement16.
Statistical analysis
The collected data were double-entered, and cleaned and analyzed using the Census and Survey Processing system (version 7.3)17 and Statistical Package for Social Sciences (version 26.0)18 respectively. Descriptive statistics of the quantitative variables: age, number of drugs prescribed, and potential DDIs were computed using mean (Standard Deviation (SD)) or median (Interquartile range (IQR)) as appropriate. On the other hand, categorical variables were summarized using frequency and percentage. Associates of clinically significant drug-drug interaction were determined primarily using bivariate logistic regression. Furthermore, factors that were significant at bivariate were retained for multivariable logistic regression and adjusted odds ratios (AORs) were computed thereafter. Tables were used to present the findings. The odds ratio with a 95% confidence interval was reported in all logistic regression analyses. P < 0.05 was considered significant in all analyses.
Ethics approval and consent to participate
Ethical clearance was obtained from the Ministry of Health Research Ethics and Protocol Review Committee (Ref. number: 020/01/2023). Besides, approval to conduct the study was obtained from the heads of the community chain pharmacies after presenting the research study proposal and demonstrating the ethical approval. In this study, data were collected retrospectively, thus the need for consent to participate was deemed unnecessary by the MOH Research Ethics and Protocol Review Committee (Ref. number: 020/01/2023). Information acquired from the prescriptions was kept confidential and used only for the purpose of the survey. Name of patients and prescribers were de-identified, and only collected information is reported. All data collection procedures were carried out in accordance with the Declaration of Helsinki.
Results
Socio-demographic characteristics of the study participants
A total of 417 outpatient prescriptions were included in the study. The majority of the outpatients were adults (55.8%) with a median age of 35 (IQR: 17, 58) years. Moreover, more than half of the outpatients were females (59.5%). Most of the encounters were ordered by general practitioners (48.7%). The majority of the encounters were from a tertiary-level health facility (63.5%) [Table 1].
Drug utilization pattern
A total of 830 medicines were prescribed from the 417 dispensed outpatient prescriptions. The mean number of drugs per prescription was 1.99 (SD: 0.98). Most of the prescriptions contained a single medication (36.0%). Moreover, 2.6% of the outpatients received five or more medicines (95%CI: 0.9, 3.9) [Table 2].
At the fourth level Anatomic Therapeutic Chemical (ATC) classification, the most commonly prescribed medication classes were proton pump inhibitors (8.4%), penicillins with extended-spectrum (8.1%), acetic acid derivatives and related substances (6.4%), and fluoroquinolones (6.3%) [Table 3]. Moreover, the most commonly prescribed individual medicines were omeprazole (8.4%), amoxicillin (8.1%), ciprofloxacin (6.3%), and paracetamol (5.9%).
Evaluation of clinically significant potential drug-drug interactions
A total of 541 drug pairs were screened for potential drug-drug interactions (pDDIs), of which 161 drug pairs had potential interactions of varying significance. Out of 267 prescriptions containing two or more medicines, the prevalence of clinically significant pDDIs was 29.2% (95%CI: 23.7, 34.7). Furthermore, a total of 133 clinically significant pDDI pairs were detected with an average of 0.5 per prescription (Median = 0, IQR = 0, 1).
Out of 161 drug pairs with potential drug interactions, more than half of them (63.4%) were risk category ‘C’ and 62.7% of them were of moderate severity [Table 4].
About 32% of the 830 medications prescribed (n = 266) were involved in clinically significant pDDIs. The most common medicines involved in clinically significant pDDIs were diclofenac (22.6%), ciprofloxacin (10.9%), enalapril (10.9%), acetylsalicylic acid (7.7%), and prednisolone (7.7%). The most common interacting pair that resulted in clinically significant pDDIs was between furosemide and enalapril (n = 9). Table 5 displays the most common clinically significant pDDIs along with their risk category, severity, clinical implication, and patient management approaches.
Determinants of clinically significant potential drug interactions
Binary logistic regression revealed that adults (COR = 5.29, 95% CI: 1.99, 14.05), older adults (COR = 10.31, 95% CI: 3.55, 29.95), number of drugs prescribed (COR = 2.60, 95% CI: 1.81, 3.75), and tertiary level of health facility (COR = 18.28, 95% CI: 2.44, 136.91) were significant associates of clinically significant pDDIs [Table 6].
Multivariable logistic regression indicated that adults had four times greater odds to clinically significant pDDIs than pediatrics (AOR = 4.23, 95% CI: 1.49, 12.01). Moreover, older adults had seven times greater odds to clinically significant pDDIs than pediatrics (AOR = 7.09, 95%CI: 2.24, 22.44). As the number of drugs prescribed increased by one unit, the odds of clinically significant pDDIs increased by 2.27 units (AOR = 2.27, 95% CI: 1.53, 3.37). Prescriptions from a tertiary-level health facility had nine times greater odds to clinically significant pDDIs than prescriptions ordered from a primary health facility (AOR = 9.13, 95% CI: 1.17, 71.42) [Table 6].
Discussion
In this study, almost one-third of the outpatient prescriptions containing two or more medicines (29.2%) had at least one clinically significant pDDI. This finding is in line with studies conducted in Qatar (31.9%)7 and Ethiopia (32.6%)9. It was however lower than studies conducted in India (91.2%)19, Croatia (84%)20, Saudi Arabia (90.6%)21, Iraq (57.6%)22, Iran (41.6%)11, and Sri Lanka (52.5%)13. Moreover, it was higher than studies conducted in Greece (13.7%)8, Egypt (18%)2, and Pakistan (22.3%)10. This inconsistency in results could be due to differences in study design, study setting, study population, drug prescription pattern, and drug interaction checker software used across the studies.
The most commonly prescribed medicines were amoxicillin and omeprazole. This finding is in contrast to a study conducted in Sri Lanka where they reported atorvastatin and metformin as the most prescribed medicines13. Differences in disease trends and prescribing practices could contribute to the variation in these findings.
More than half of the potential drug interactions were risk category ‘C’ and moderate in severity. This finding is comparable to similar studies conducted elsewhere2,6,7,11,21,22,23. This mandates healthcare professionals to strictly monitor such interactions while taking patient factors and the clinical significance of drug interactions into consideration and provide counseling to patients on the effect of drug interaction and when to consult medical care.
In this study, the most medicines involved in clinically significant pDDIs were diclofenac, ciprofloxacin, and enalapril. This finding is similar to studies conducted in Jordan6 and Egypt2 where they reported cardiovascular system drugs and diclofenac as the most medicines involved in pDDIs, respectively. Ciprofloxacin is often prescribed with prednisolone, raising concerns about potential side effects like tendonitis and tendon rupture, especially in older adults or those with pre-existing health conditions24. Moreover, combining ciprofloxacin with ibuprofen may increase the risk of seizures, requiring careful monitoring when these drugs are used together25. Enalapril often interacts with spironolactone and furosemide, which can lead to complications such as hyperkalemia26 and hypotension27 respectively. Furthermore, diclofenac is commonly prescribed with itself and prednisolone, increasing the risk of gastrointestinal problems28,29,30. To mitigate risks of adverse drug outcomes, healthcare professionals should be vigilant in monitoring prescriptions containing these medicines.
The most common interacting pair was furosemide-enalapril and enalapril-spironolactone. This result is in contrast with studies conducted in Saudi Arabia (atorvastatin-omeprazole)21, Pakistan (ciprofloxacin-diclofenac)10, Iraq (non-steroidal anti-inflammatory drugs-aspirin)22, Ethiopia (enalapril-hydrochlorothiazide)23 and Croatia (Indapamide-perindopril)20. The possible explanation for the variation in results may be due to the difference in prescribing habits, disease patterns, essential medicine lists, study population, study setting, and drug interaction checker software. The implication is that the interacting pairs of furosemide-enalapril and enalapril-spironolactone would result in increased hypotensive and hyperkalemic effects, respectively. When at times concomitant use of these medicines is inevitable, prescribers should closely monitor for any signs of adverse effects resulting from the interactions while properly managing the comorbid conditions of patients.
The determinants of clinically significant pDDIs in this study were age, number of drugs prescribed, and level of health facility. Adults had four times greater odds to clinically significant pDDIs than pediatrics. Moreover, the older adult population had seven times greater odds to clinically significant pDDIs than pediatrics. This could be because an increased number of medications were prescribed in adults (aged 18–64 years) and older adults (aged 65 and above years). Moreover, the fact that the older adults are encountered with multiple chronic illnesses, often requiring multiple medications could contribute to the occurrence of clinically significant pDDIs. Thus, healthcare professionals should be cautious when prescribing medicines in these age groups.
The occurrence of clinically significant pDDIs increased by 2.27 units with an increase in the number of drugs prescribed. This finding is similar to studies conducted in Saudi Arabia21, Qatar7, India19 and Ethiopia9. Prescribers should therefore carefully assess the risk-benefit ratio while prescribing multiple medications.
Prescriptions from a tertiary-level health facility had nine times greater odds to clinically significant pDDIs than prescriptions ordered from a primary health facility. This could be because prescribers in tertiary-level health facilities are authorized to prescribe a majority of medications in the Eritrean National List of Medicine (ENLM) version-7 (2019) which could further contribute to an increased number of prescribed medications and thereby drug interactions of clinical significance.
Outpatient prescriptions should be carefully screened and monitored for potential drug interactions while taking the medical and medication histories and age of the patient into consideration. Inappropriate prescribing and lack of prescription monitoring could result in hospitalization and adverse drug reactions. The significance of this study and the results it comes up with are probably of particular interest to countries with similar socio-economic status as Eritrea. To minimize complications associated with drug interactions, educational programs targeting healthcare professionals and patients, introduction of electronic drug interaction checker software in health facilities, and regular updating of standard treatment guidelines and formularies are highly recommended.
Limitations of the study
All the drug-drug interactions documented in this study are theoretical, their clinical effect in the real ground might be under- or over-estimated. Moreover, a retrospective prescription-based study could not allow information related to the comorbid condition of outpatients which can impact the occurrence of potential drug interactions. Finally, the results from this study were not generalizable to the whole country. Thus, the authors recommended further broader research with larger sample size and longer duration to get a comprehensive picture of the burden of drug interactions on outpatients.
Conclusion
In this study, clinically significant pDDIs were detected in a considerable number of outpatient prescriptions. Age, number of drugs prescribed, and level of health facility were determinants of clinically significant pDDIs. To mitigate risks associated with drug interactions, educational sessions targeting healthcare professionals and patients, introduction of electronic drug interaction screening tools, and adherence to good prescribing and dispensing practices are highly recommended.
Data availability
All relevant data are within the manuscript. The complete dataset used for this study can be obtained from the corresponding author upon request.
Abbreviations
- ADR:
-
Adverse drug reaction
- AOR:
-
Adjusted odds ratio
- ATC:
-
Anatomical Therapeutic Chemical classification
- CI:
-
Confidence interval
- COR:
-
Crude odds ratio
- ENLM:
-
Eritrean National List of Medicine
- IQR:
-
Interquartile range
- M:
-
Mean
- Md:
-
Median
- MOH:
-
Ministry of Health
- pDDI:
-
Potential drug–drug interactions
- Ref.:
-
Reference
- SD:
-
Standard deviation
- SPSS:
-
Statistical Package for Social Sciences
- WHO:
-
World Health Organization
References
Desalegn, A. A. Assessment of drug use pattern using WHO prescribing indicators at Hawassa University Teaching and Referral Hospital, South Ethiopia: a cross-sectional study. BMC Health Serv. Res. 13, 1–6 (2013).
Abdelkawy, K. et al. Prevalence of drug–drug interactions in primary care prescriptions in Egypt: A cross-sectional retrospective study. Pharmacy 11, 106 (2023).
Prueksaritanont, T. Gibson drug-drug interaction studies: regulatory guidance and an industry perspective. AAPS J. 15, 629–645 (2013).
4 Palleria, C. et al. Pharmacokinetic drug-drug interaction and their implication in clinical management. J. Res. Med. Sciences: Official J. Isfahan Univ. Med. Sci. 18, 601 (2013).
Kalash, A., Abdelrahman, A., Al-Zakwani, I. & Al Suleimani, Y. Potentially harmful drug–drug interactions and their associated factors among hospitalized cardiac patients: A cross-sectional study. Drugs-Real World Outcomes. 10, 371–381 (2023).
Nusair, M. B. et al. The prevalence and severity of potential drug-drug interactions among adult polypharmacy patients at outpatient clinics in Jordan. Saudi Pharm. J. 28, 155–160 (2020).
Abbas, A. et al. Determination of potential drug–drug interactions in prescription orders dispensed in a community pharmacy setting using Micromedex® and Lexicomp®: a retrospective observational study. Int. J. Clin. Pharm. 44(2), 348–356 (2022).
Anna Chatsisvili, I. S. et al Potential drug–drug interactions in prescriptions dispensed in community pharmacies in Greece. Pharm. World Sci. 32, 187–193. https://doi.org/10.1007/s11096-010-9365-1 (2010).
Admassie, E., Melese, T., Mequanent, W., Hailu, W. & Srikanth, B. A. Extent of poly-pharmacy, occurrence and associated factors of drug-drug interaction and potential adverse drug reactions in Gondar Teaching Referral Hospital, North West Ethiopia. J. Adv. Pharm. Tech. Res. 4, 183–189 (2013).
Ismail, M. et al. Potential drug-drug interactions in Outpatient Department of a Tertiary Care Hospital in Pakistan: a cross-sectional study. BMC Health Serv. Res. 18, 1–7 (2018).
Dirin, M. M., Mousavi, S., Afshari, A. R., Tabrizian, K. & Ashrafi, M. H. Potential drug-drug interactions in prescriptions dispensed in community and hospital pharmacies in East of Iran. J. Res. Pharm. Pract. 3, 104–107 (2014).
Abdu, N. et al. Non-steroidal anti-inflammatory drugs (NSAIDs): usage and co-prescription with other potentially interacting drugs in elderly: A cross-sectional study. PloS One. 15, e0238868 (2020).
Rathish, D. et al. Drug utilization, prescription errors and potential drug-drug interactions: an experience in rural Sri Lanka. BMC Pharmacol. Toxicol. 17, 1–9 (2016).
Kheshti, R., Aalipour, M. & Namazi, S. A comparison of five common drug–drug interaction software programs regarding accuracy and comprehensiveness. J. Res. Pharm. Pract. 5, 257–263 (2016).
Lexicomp. Lexicomp Drug Interactions (Wolters Kluwer Health, Inc. UpToDate, 2018).
Von Elm, E. et al. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Int. J. Surg. 12, 1495–1499 (2014).
U.S. Census Bureau, ICF International, and Serpro S.A. Released Census and Survey Processing System for Windows, Version 7.3.1. (2019).
IBM Corp. Released 2019. IBM SPSS Statistics for Windows, Version 26.0. (IBM Corp., 2019).
Shetty, V. et al. Evaluation of potential drug-drug interactions with medications prescribed to geriatric patients in a Tertiary Care Hospital. J. Aging Res. 2018 (2018).
Samardžić, I., Marinović, I., Kuča, N. & Vrca, V. B. Potential clinically significant drug-drug interactions in prescribed pharmacotherapy in an outpatient setting. Die Pharm. Int. J. Pharm. Sci. 76, 390–395 (2021).
Aljadani, R. & Aseeri, M. Prevalence of drug–drug interactions in geriatric patients at an Ambulatory Care Pharmacy in a Tertiary Care Teaching Hospital. BMC Res. Notes. 11, 1–7 (2018).
Kasim, H. F. Prevalence of potential drug interactions among outpatients’ prescriptions of community pharmacies in Nineveh Governorate, Iraq. J. Pharm. Pharmacognosy Res. 11, 281–290 (2023).
Gobezie, M. Y., Bitew, H. B., Tuha, A. & Hailu, H. G. Assessment of potential drug–drug interactions and their predictors in Chronic Outpatient Department of Dessie Referral Hospital, Dessie, Northeast Ethiopia. Drug Healthc. Patient Saf. 29–35 (2021).
Van der Linden, P. D. et al. Increased risk of Achilles tendon rupture with quinolone antibacterial use, especially in elderly patients taking oral corticosteroids. Arch. Intern. Med. 163, 1801–1807 (2003).
Kim, J., Ohtani, H., Tsujimoto, M. & Sawada, Y. Quantitative comparison of the convulsive activity of combinations of twelve fluoroquinolones with five nonsteroidal antiinflammatory agents. Drug Metab. Pharmacokinet. 24, 167–174 (2009).
Abbas, S., Ihle, P., Harder, S. & Schubert, I. Risk of hyperkalemia and combined use of spironolactone and long-term ACE inhibitor/angiotensin receptor blocker therapy in heart failure using real‐life data: a population‐and insurance‐based cohort. Pharmacoepidemiol. Drug Saf. 24, 406–413 (2015).
Good, J. M., Brady, A., Noormohamed, F. H., Oakley, C. M. & Cleland, J. Effect of intense angiotensin II suppression on the diuretic response to Furosemide during chronic ACE inhibition. Circulation 90, 220–224 (1994).
Sohail, R. et al. Effects of non-steroidal anti-inflammatory drugs (NSAIDs) and gastroprotective NSAIDs on the gastrointestinal tract: A narrative review. Cureus 15 (2023).
Piper, J. M., Ray, W. A., Daugherty, J. R. & Griffin, M. R. Corticosteroid use and peptic ulcer disease: role of nonsteroidal anti-inflammatory drugs. Ann. Intern. Med. 114, 735–740 (1991).
Tai, F. W. D. & McAlindon, M. E. Non-steroidal anti-inflammatory drugs and the gastrointestinal tract. Clin. Med. 21, 131–134 (2021).
Acknowledgements
The authors sincerely acknowledge the heads of the community chain pharmacies for their cooperation and contribution during data collection process.
Author information
Authors and Affiliations
Contributions
NA: Conceptualization; Methodology; Data curation; Formal analysis; Investigation; Resources; Validation; Visualization; Project administration; Supervision; Writing – original draft; Writing – review and editing. TT: Methodology; Investigation; Validation; Writing – review and editing. NJ: Methodology; Investigation; Validation; Writing – review and editing. SI: Methodology; Data curation; Formal analysis; Investigation; Resources; Validation; Supervision; Writing – original draft; Writing – review and editing. EHT: Methodology; Data curation; Formal analysis; Validation; Visualization; Supervision; Writing – review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Ethical clearance was obtained from the Ministry of Health Research Ethics and Protocol Review Committee (Ref. number: 020/01/2023). Besides, approval to conduct the study was obtained from the heads of the community chain pharmacies after presenting the research study proposal and demonstrating the ethical approval. In this study, data were collected retrospectively, thus the need for consent to participate was deemed unnecessary by the MOH Research Ethics and Protocol Review Committee (Ref. number: 020/01/2023). Information acquired from the prescriptions was kept confidential and used only for the purpose of the survey. Name of patients and prescribers were de-identified, and only collected information is reported. All data collection procedures were carried out in accordance with the Declaration of Helsinki.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Abdu, N., Tewelde, T., Jabir, N. et al. Evaluation of potential drug–drug interactions and its determinants among outpatient prescriptions in six community chain pharmacies in Asmara, Eritrea: a cross-sectional study. Sci Rep 15, 33403 (2025). https://doi.org/10.1038/s41598-025-18739-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-18739-8