Abstract
Maintaining genetic fidelity in in vitro-regenerated plants derived from chimeric tissues presents a significant challenge during tissue culture experiments. Sansevieria trifasciata cv. Laurentii exhibits striking periclinal chimeric tissues along its leaves, which are compromised when propagating plants through leaf cuttings. In addition to establishing in vitro production of S. trifasciata cv. Laurentii, the regeneration efficiency of light exposed single-node rhizome explants was also evaluated. Among various types of rhizome explants, the single-node rhizome explant produced the highest mean number of shoots per explant (2.5) on MS medium supplemented with 3 mg L− 1 BA and 0.1 mg L− 1 NAA, which was approximately five times higher than that of other explants. Notably, all regenerated plants originating from nodal and terminal meristems were morphologically similar to their corresponding mother plants and retained their chimeric characteristics. In contrast, shoots derived from explants containing both nodal segments and terminal meristems (MN explants) exhibited some degree of morphological variation. Specifically, higher concentrations of BA (6 mg L− 1) were found to decrease the phenotypic stability of regenerated shoots by 50%. De novo rhizomes derived from light exposed single-node rhizome (NC explants) yielded the highest mean number of shoots (6 shoot per explant) on MS medium supplemented with 4 mg L− 1 NAA and either 0.5 or 0.35 mg L− 1 BA. Shoots from de novo rhizomes closely resembled the mother plants, while callus-derived shoots showed only 65% similarity. The presence of light-influenced elevated sugar levels in NC explants supports the hypothesis that the enhanced regeneration capacity in light-exposed single-node rhizome tissues is likely attributed to the upregulation of cytokinin synthesis-related genes and light-induced carbohydrate allocation, which alters hormonal homeostasis and triggers the mitotic activities necessary for de novo rhizome regeneration and shoot multiplication.
Similar content being viewed by others
Introduction
Ornamental plants have been integral to modern life, providing economic value and enhancing well-being through aesthetic appeal, especially in urban environments. Sansevieria, native to Africa and Asia, has become a popular indoor plant prized for its variegated leaves. Beyond aesthetics, some Sansevieria species have offered benefits like fiber production and medicinal applications1. The genus has comprised over 50 species, with S. trifasciata favored as a houseplant for its striking, upright foliage and resilience in low-light conditions. It has also been recognized for air-purifying qualities2. Many desirable cultivars have originated from S. trifasciata cv. Laurentii. Sansevieria can be propagated via seeds, leaf cuttings, and rhizomes3, but seed propagation has been limited by slow growth, seed scarcity, and genetic variability. S. trifasciata cv. Laurentii has exhibited a periclinal chimera with a yellow-pigmented outer epidermal layer (L.II) surrounding non-mutated inner tissues (L.III)4. Crucially, true-to-type propagation of this chimera via leaf cuttings has proven unsuccessful5. Division has remained the most reliable method for maintaining the chimeric nature of this cultivar, but it has been labor-intensive and has yielded low outputs. Micropropagation could have offered a solution for mass propagation, but explant selection, regeneration methods, and plant growth regulators (PGRs) have significantly impacted plant uniformity6,7, making explant origin critical. While several studies have detailed Sansevieria micropropagation, including shoot regeneration from leaf explants of non-chimeric S. trifasciata using 2,4-dichlorophenoxyacetic acid (2,4-D) and kinetin8,9 or 6-Benzylaminopurine (BA) and naphthaleneacetic acid (NAA)10, and indirect organogenesis from S. trifasciata cv. Laurentii and Hahnii leaf explants11, none have reported success in producing chimeric S. trifasciata.
Since the propagation of this cultivar through leaf cuttings has been unsuccessful, innovative approaches have been needed. This study has explored the potential of light exposed single-node rhizome explants as a novel strategy to propagate true-to-type chimeric S. trifasciata cv. Laurentii, aiming to overcome the challenges of producing non-chimeric plants and to enhance regeneration efficiency. The main innovation of our study was the use of rhizome explants, particularly those exposed to light, which contain chlorophyll. This approach was not previously reported in tissue culture studies for this species, especially using rhizomes under artificial light conditions for chlorophyll production. Our method demonstrated a significantly higher propagation efficiency and a much greater likelihood of producing plants that resemble the original than traditional propagation through leaf cuttings, which has been extensively reported. This novel approach offers a promising alternative with superior outcomes for true-to-type plant production.
Methods
In the current study, Sansevieria trifasciata cv. Laurentii was utilized. The parent pots of the Sansevieria plants were purchased from a commercial greenhouse. Prior to transfer, mother plants were confirmed free of microbial contamination. All of the plants used in the experiment were of uniform size and consistent in appearance.
Primary mother plant establishment, surface sterilization and direct organogenesis
To minimize contamination and potential interference from soil-borne pathogens, rhizomes from purchased mother plants were initially established in sterile perlite (5 mm) within standard 25-cm diameter plastic pots, instead of soil or other organic substrates. Before planting, the root and rhizome sections were briefly immersed in a 10% Benomyl fungicide. The perlite beds were maintained under controlled conditions in a greenhouse. The average temperature was maintained at 25 °C ± 4. Plants were fertilized monthly with a water-soluble commercial fertilizer (N-P-K ratio, 20-20-20) at a concentration of 2 g/L. The greenhouse was supplemented with artificial lighting to provide a light intensity of 100 µmol m− 2 s− 1. Humidity was maintained at approximately 70%. Several months after establishment of mother plants, the rhizome segments were then cut and transferred to a laminar airflow hood, where they were treated with 70% ethanol for 2 min and 0.2% mercuric chloride for 20 min12,13. Following this, the segments were thoroughly rinsed at least three times with sterilized distilled water. Ultimately, three types of explants (Fig. 1) were prepared from the sterilized rhizomes, designated as M (explant containing terminal meristem), N (explant containing nodal segment), and MN (explant containing both nodal segment and terminal meristem). All explants were cultured on Murashige and Skoog (MS) basal medium supplemented with kinetin (0.3, 1, 3, and 6 mg L− 1), TDZ (1, 2, and 4 mg L− 1), or a combination of BA (0, 1, 3, and 6 mg L− 1) with NAA (0, 0.1, and 0.3 mg L− 1). These hormonal treatments were selected based on a preliminary evaluation from a wider range of hormonal treatments. Some treatments did not produce any response from the explants, and therefore, those concentrations are not included in the results. For each treatment, 3 replicates (Plastic containers) were used and 4 explants were cultured in each container.
Four types of explants, obtained from the sterilized rhizomes of Sansevieria trifasciata cv. Laurentii, are designated as M (explant containing terminal meristem), N (explant containing nodal segment), MN (explant containing both nodal segment and meristem section), and NC (nodal segment explant containing chlorophyll).
Indirect regeneration
In this phase of the experiment, the mother plants of Sansevieria were carefully removed from their pots (perlite) and replanted in transparent, light-permeable containers without disturbing the root system. The root system and rhizomes of the mother plants were exposed to an indirect spectrum of sunlight (30–50 µmol m⁻² s⁻¹) for two months to promote chlorophyll synthesis and enhance regenerative potential. Samples were maintained under a 16:8 light-dark photoperiod. The light exposure aimed to activate photosynthetic processes and increase chlorophyll content, potentially improving explant development. These light exposed rhizomes were then used to obtain explants. As a result, two types of explants (Fig. 1) were prepared for indirect regeneration, designated as N (nodal segment) and NC (nodal segment containing chlorophyll). The aforementioned explants were then cultured on Murashige and Skoog (MS) basal medium supplemented with 4 mg L− 1 NAA, in combination with 0.15, 0.25, 0.35, and 0.5 mg L− 1 of BA, TDZ, or kinetin. The NAA concentration of 4 mg L− 1 was selected based on the preliminary response of explants to this PGR concentration. Each treatment consisted of 3 replicates (plastic containers), with 4 explants cultured per container.
Adaptation
All the regenerated shoots (over 5 cm in length) were carefully removed from the culture medium after thorough washing and separation from the agar. The shoots were then immersed in a 1000 mg L− 1 IBA solution for 5 s to encourage rooting. After this treatment, they were planted in a substrate consisting Coco-peat and perlite (1:1 in volume). To promote rooting and establishment, all samples were maintained under a mist system for 60 days. The average temperature was maintained at 25 °C ± 4. Plants were fertilized monthly with a water-soluble commercial fertilizer (N-P-K ratio, 20-20-20) at a concentration of 2 g/L. The greenhouse was supplemented with artificial lighting to provide a light intensity of 100 µmol m− 2 s− 1. Humidity was maintained at approximately 85%. During the hardening stage (2 months), the intensity of the mist gradually decreased. The adaptation process went smoothly, with all plantlets developing healthy root systems and adapting within a short period.
Data analysis
The experiment was conducted using a completely randomized design. Comparative data analysis between the two treatments (N and NC explant) was performed using an independent samples T-test with SPSS software version (V.30). For each treatment, 3 replicates (plastic containers) were used, with 4 explants cultured per container. Cultures were initiated simultaneously, and data collection commenced 12 weeks after explant establishment at a single time point.
The following parameters were measured: mean number and length of shoots and roots per explant, shoot height, mean number of leaves per explant, regenerated shoot diameter, and morphological similarity between regenerated shoots and their corresponding mother plant. The criteria used for assessing morphological similarity included the presence of two yellow stripes on the margins of newly developing leaves, which was one of the morphological traits used to evaluate the similarity of regenerates to the mother plant. Number and length of de novo rhizomes were recorded in experiments aimed at rhizogenesis.
For shoot fresh weight measurement, shoots were removed from culture media under an air flow cabinet and weighed on an analytical balance with a precision of ± 0.0001 g.
Total sugar content was measured in N and NC explants. Total sugar was quantified using the method described by McCready et al.14, which involves the use of anthrone reagent and spectrophotometric determination at 620 nm. Sugar measurements were performed on three independent biological replicates per treatment.
Results
Direct shoot multiplication from nodal explant
Shoot multiplication from single-node rhizome explants (N) has proven recalcitrant, with shoot induction observed in only a subset of the tested hormonal treatments. The majority of explants cultured on MS media supplemented with various PGRs have exhibited limited or no response. Significant proliferation has been observed only with the following treatments: 3 mg L-1 BA combined with 0.1 mg L-1 NAA, 3 mg L-1 kinetin, 1 mg L-1 TDZ, and 3 mg L-1 BA alone (Table 1). All regenerated shoots retain the chimeric characteristics of their respective mother plants. The highest mean number of shoots per explant (2.5) has been achieved with 3 mg L-1 BA in combination with 0.1 mg L-1 NAA (The data were obtained from evaluating 12 explants), a value that has been significantly higher than that observed for all other treatments (p ≥ 0.01; Table 1). Shoots in this treatment have originated from both nodal and internodal regions. Critically, all proliferated shoots have maintained consistent morphological characteristics, exhibiting no observable variation (Fig. 2). Furthermore, explants cultured on MS media supplemented with 3 mg L-1 BA exhibited a tendency toward decreased root length (0.06 cm). The highest mean number of leaves per regenerated shoot and total fresh weigth was observed in MS media supplemented with 3 mg L-1 BA and 0.1 mg L-1 NAA. This value was significantly different from that of other treatments (p ≥ 0.01; Table 1). No statistically significant differences (p ≤ 0.05) were observed among the various treatments with respect to the number or length of adventitious roots generated (Fig. 2; Table 1). This data was obtained from the evaluation of 12 explants per treatment.
Representative true-to-type regenerated shoots from nodal explants (N) of Sansevieria trifasciata cv. Laurentii on MS medium supplemented with different concentrations of plant growth regulators (PGRs): 1 mg L-1 TDZ (a), 3 mg L-1 6-BA along with 0.1 mg L-1 NAA (b and c), 1 mg L-1 TDZ (d), 3 mg L-1 kinetin (e), and 3 mg L-1 6-BA (f and g).
Direct shoot multiplication from apical meristem explant
The objective of this experiment was to assess the response of apical meristem explants (M) derived from rhizomes (Fig. 1) when cultured on MS media supplemented with six different concentrations of plant growth regulators (PGRs). While M explants cultured on PGR-free MS media exhibited basal growth, the rate was demonstrably slow, and growth eventually ceased. Specifically, initial signs of activity (production of new leaves) were not observed in the control explants until six months. In contrast, M explants exposed to PGRs exhibited a significantly reduced time to initial activity, averaging 45 days (Table 2). Due to the insufficient data collected from control explants within the 12-week observation period, further characterization of control explants was not performed.
In treatments where growth was successfully induced, all resulting plantlets exhibited morphological similarity to the corresponding mother plant (p ≤ 0.05). All explants produced a single shoot (Table 2; Fig. 3). M explants cultured on media containing 2 and 4 mg L− 1 TDZ produced single shoots with greater diameter and length compared to other treatments. However, these higher concentrations of TDZ were associated with a significant reduction in root number and length. TDZ, in general, appeared to positively influence the number of leaves produced (p ≥ 0.05). In contrast, the higher root length (2.9 cm) was observed in media supplemented with 1 mg L− 1 BA (Table 2), which was significantly longer than that of other treatments (p ≥ 0.05).
Direct shoot multiplication from MN explant
Interestingly, some of the regenerated shoots derived from MN explants exhibited morphological differences from the chimeric mother plant. The lowest percentage of variation among MN-derived regenerated shoots was observed in MS media supplemented with 1 mg L-1 of BA, where more than 80% of the regenerated shoots showed phenotypic resemblance to the original plant. This concentration also resulted in the highest shoot length and total fresh weight per explant. In contrast, explants cultured in PGR-free MS media showed a very low growth rate, with most remaining unchanged or exhibiting minimal growth even after six months; consequently, they were excluded from the final analysis (Table 3).
While MN explants treated with higher concentrations of BA produced an increased number of shoots (p ≤ 0.05, Fig. 4), the elevated concentrations also led to a significant decrease in the morphological similarity of the regenerated shoots to their corresponding mother plant, particularly in media supplemented with 6 mg L-1 of BA, where similarity dropped to approximately 50% (Table 3). As anticipated, the mean length of de novo shoots declined with higher concentrations of BA. In addition, the number and length of regenerated roots decreased at lower concentrations of BA (0.1 mg L-1), while the total fresh weight was significantly higher at this concentration. Morphological similarity was assessed based on specific traits, including the presence of two yellow stripes along the margins of newly developing leaves, which served as a key criterion for evaluating the similarity of regenerates to the mother plant.
Comparison of regeneration ability of simple nodal explant with light exposed single-node rhizome (NC)
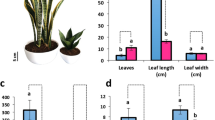
In this section, we evaluate the regeneration capacity of single-node explants using a specific type of rhizome explant grown under two different light conditions (refer to the Materials and Methods section for details on light intensity and duration). The light exposed single-node rhizome tissue (NC) demonstrated a remarkable ability to generate de novo rhizomes (p ≥ 0.01), producing seven times more than the simple single-node rhizomes (N). Although no significant differences were observed in rhizome length and diameter, variations were noted in the number of de novo rhizomes, regenerated shoots, and roots between these two types of explants (Table 4; Fig. 5). Furthermore, the fresh weight of regenerated shoots from NC explants was three times greater than that of N explants (p ≥ 0.01). This data was obtained from the evaluation of 12 explants per treatment.
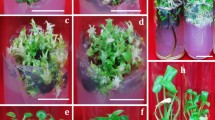
Representative shoots and de novo regenerated rhizomes from light-exposed rhizome explants (NC) of Sansevieria trifasciata cv. Laurentii on MS medium. a: True-to-type generated shoot (4 mg L-1 NAA + 0.5 mg L-1 TDZ). b: Production of chimeric shoot on rhizome-originated callus (4 mg L-1 NAA + 0.15 mg L-1 BA). c: regeneration of green shoots.
Interestingly, the shoots originating from de novo rhizomes exhibited morphological similarities to their corresponding mother plants, while the shoots derived from rhizomes-originated callus showed only 60% similarity to their respective mother plants (p ≥ 0.01, Table 4). This data was obtained from the evaluation of 24 explants per treatment.
Utilizing NC explants in MS media supplemented with various PGRs
In this section, we used NC explants in MS media supplemented with various PGRs. Only a few treatments successfully produced NC-originated de novo rhizomes. One effective medium was MS media supplemented with 4 mg L-1 NAA (Table 5). The length and number of de novo rhizomes did not show statistically significant differences among the treatments presented in Table 5 (p ≤ 0.05). Most combinations of PGRs yielded 4 to 7 de novo rhizomes, except for the MS medium supplemented with 4 mg L-1 NAA in combination with 0.15 mg L-1 BA, which only induced callus formation. Research findings indicated that the de novo rhizome-originated shoots maintained their phenotypic resemblance and were morphologically similar to their corresponding mother plants, while the callus-originated shoots from NC explants displayed 65% similarity to their respective plants. In some instances, de novo rhizomes and callus were produced concurrently on NC explants (Fig. 5). The MS medium supplemented with 4 mg L-1 NAA and 0.5 mg L-1 BA maximized both the number of de novo rhizomes (7) and the total fresh weight (8.3 g) of the regenerated shoots on NC explants (p ≥ 0.01, the data were obtained from evaluating 12 explants per treatment). Since the NC explants were exposed to artificial light, one possible reason for their higher regeneration rate is the increased sugar content resulting from photosynthesis within these tissues. Therefore, sugar levels were measured in both the NC and simple single-node rhizomes. The results indicated that the NC explants exposed to light had significantly higher sugar content, which may have contributed to their enhanced regenerative capacity (Fig. 6). The highest mean number of shoots (6.3) was observed in the MS medium supplemented with 4 mg L-1 NAA and 0.35 mg L-1 BA. All samples treated with IBA rooted and acclimatized well, with 95% of the plantlets ultimately establishing and growing vigorously (Fig. 7). The treatment of 4 mg L-1 NAA in combination with various concentrations of kinetin (Supplementary 1) resulted in significantly lower regeneration rates and fewer de novo rhizomes compared to the treatment of 4 mg L-1 NAA combined with BA and TDZ.
Discussion
The Sansevieria plant is popular for its elongated, fleshy, and visually striking leaves, as well as its resilience to environmental stress and drought, making it well-suited as a houseplant. The presence of heterogeneous periclinal tissues in S. trifasciata cv. Laurentii and the difficulties in propagating it through leaf cuttings3, highlight the need for effective solutions to enhance the production efficiency of various chimeric Sansevieria cultivars. Currently, the most reliable method for propagating periclinal cultivars of Sansevieria is through plant division, which is an inefficient process3. A key objective of this research is to explore rhizome-mediated shoot induction in the chimeric Laurentii cultivar and to enhance its regeneration efficiency. Chimeras, or plants with heterogeneous tissues, contain cells of distinct genetic makeups. S. trifasciata cv. Laurentii exhibits a periclinal chimera with color changes due to mutations in the L.II layer of meristem4,15. The micropropagation of this cultivar through leaf explants does not yield true-to-type plants3,5,9,16,17. Therefore, alternative asexual propagation methods are needed to maintain maternal characteristics and improve production efficiency.
This study demonstrates higher regeneration efficiency in Sansevieria when nodal explants (N explants) from rhizomes are cultured on MS medium supplemented with 3 mg L-1 BA and 0.1 mg L-1 NAA. This treatment yielded about five times more plantlets and leaves than other treatments, and a significantly greater number of roots. Because rhizome explants did not respond in hormone-free medium, the control was omitted in line with prior findings5,9.
The plantlets produced through direct regeneration, whether derived from the node or internode (N explant), were identical to the corresponding mother plant. It is likely that all plantlets regenerating directly from the rhizome originated from the cortex (Supplementary 2) in the L.II layer, which explained their consistent resemblance to the mother plant in direct regeneration4. Traditional snake plant propagation by underground rhizome division consistently produced periclinal plants. Literature indicated that plants derived from the epidermal tissue of thornless blackberry (via rooted cuttings, budding, grafting, divisions, or tip layering) maintained the thornless genotype. Conversely, shoots arising from root cuttings tended to produce a thorny genotype because the primary root cutting originated from the subepidermal tissues of the stem (L.II and L.III); thus, all new shoots on root cuttings carried the L.II and L.III layers18,19. The low number of plantlets produced from M explants has been attributed to apical dominance, as Kudoyarova et al.20 suggested, where auxin hormones produced in terminal meristems have inhibited the development of additional plantlets. The presence of the apical shoot in M explants has likely resulted in high internal auxin concentrations, inhibiting further plantlet development while promoting root production, even in the presence of high cytokinin concentrations. Plantlets directly derived from MN explants have exhibited approximately 60% phenotypic similarity to the maternal plant. The observed phenotypic variation has likely resulted from the inclusion of leaf-derived tissue within the explants, a phenomenon consistent with Sarmast et al.5, who demonstrated that in vitro regeneration of shoots from leaf explants of the variegated Laurentii cultivar yielded exclusively green regenerates, suggesting potential somaclonal variation or epigenetic modification in newly generated plantlets. The increased number of roots generated on MN explants cultured with 1 mg L-1 BA may be due to the lower cytokinin concentration, which has likely enhanced the intra-tissue ratio of auxins to cytokinins—a phenomenon also observed by Kudoyarova et al.20. Elevated concentrations of BA in MS media have shown an inverse correlation with the phenotypic resemblance of regenerated shoots; specifically, phenotypic similarity to the maternal explant has decreased to approximately 50% when the BA concentration has been increased to 6 mg L-1. This finding aligns with Majumder7 and Ferreira et al.21, who have noted that higher concentrations of plant growth regulators, especially cytokinins, can result in somaclonal variation.
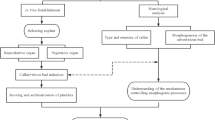
Optimal rhizogenesis has been observed, with an average of 7.3 rhizomes per explant, in light exposed NC explants cultured on medium supplemented with 4 mg L-1 NAA in combination with 0.35 mg L-1 BA. Research results have indicated that NC explants have generated over five times more new de novo rhizomes compared to the N explants. Additionally, the fresh weight, growth rate, number of roots, and number of plantlets produced from the NC explants have been significantly higher than those from the N explants. Figure 8 has illustrated the optimized protocol for various types of rhizome explants used in shoot regeneration of Sansevieria trifasciata cv. Laurentii.
This substantial increase has underscored the potential of in vitro propagation to significantly enhance Sansevieria production. Notably, all the plantlets derived from the de novo regenerated rhizomes have closely resembled the mother plant. Based on these investigations, all de novo rhizomes have been produced directly from the primary rhizome explants. A salient innovation of this study has been the utilization of single-node, light exposed explants, which have significantly enhanced the efficiency of clonal propagation. This difference in results could be attributed to the presence of light in the production of plant growth regulators and certain enzymes, since the plastid is the primary source of hormone production22. Irradiation has likely mediated the catabolism of diverse auxin species within the rhizome tissue, concurrent with the promotion of cytokinin accumulation in the same region23. Given the modulation of mitotic activity in meristematic tissues by carbon source24, we have quantified total sugar content in N and NC explants. Rhizomes grown on transparent, light-permeable containers have shown a 1.5-fold increase in total sugar content (light exposed single-node rhizome tissue) compared to simple nodal rhizome explants grown in normal containers (Fig. 6).
Light intensity, photoperiod, and light quality have been identified as critical physical factors that influence plant growth and development, including processes such as morphogenesis, phototropism, photosynthesis, and overall plant metabolism25. It has been stated that sugar is necessary to trigger bud outgrowth of in vitro-cultivated single nodes of R. hybrid26. Moreover, sugar has the ability to alter hormonal homeostasis through the induction of IPT3 expression and the repression of cytokinin oxidase in Arabidopsis27, and has up-regulated CK synthesis-related genes in R. hybrid26. Therefore, the enhanced regenerative capacity observed in light exposed single-node rhizome tissue has likely been attributable to the upregulation of genes involved in cytokinin biosynthesis and light-mediated carbon partitioning, leading to altered hormonal homeostasis that promotes mitotic activity for de novo rhizogenesis and shoot proliferation. One of the main limitations of micropropagation in variegated Sansevieria cultivars is the risk of microbial contamination of the rhizome explants. Additionally, there is a possibility of producing plants that do not resemble the original cultivar due to the use of inappropriate explants or excessively high hormone concentrations, which could increase the cost of commercial production. However, these limitations may vary depending on the specific cultivation conditions and cultivar used, and with optimized protocols, these challenges can potentially be mitigated. Nonetheless, scaling up this protocol for large-scale commercial applications may present further challenges related to explant variability, contamination control, and cost efficiency. Variability in explant size and age may have led to inconsistent results, though this can have been minimized through standardized selection. Additionally, minor fluctuations in environmental conditions during in vitro cultivation could have influenced plant development, impacting reproducibility. Addressing these factors with stricter sterilization protocols, uniform explant preparation, and controlled growth conditions could have improved the reliability of future studies. Our study introduced a novel propagation protocol based on light exposed rhizome explants, especially those exposed to light, a method not referenced in prior literature for tissue culture or propagation of Sansevieria. The regeneration efficiency achieved was substantially higher, increasing the likelihood of obtaining true-to-type plants. Compared to conventional leaf cutting techniques, which have been widely documented, our approach resulted in a much higher plantlet yield and better morphological fidelity. This indicates that using light-exposed rhizomes offers significant advantages—both in efficiency and plant quality—and provides a unique, previously unreported pathway for mass propagation through tissue culture.
In conclusion, indirect regeneration from light exposed rhizome explants offers a compelling strategy to significantly enhance the regeneration efficiency of true-to-type Sansevieria plants. This significant enhancement in propagation efficiency highlights the value of utilizing rhizomes for regeneration, offering a substantial advantage in plantlet production compared to traditional methods. This approach has the potential to enhance mass production of high-quality, true-to-type plants with much higher efficiency.
Data availability
All data generated or analyzed during this study are included in this manuscript and are available from the corresponding author on reasonable request.
References
Thu, Z. M. et al. Structures and bioactivities of steroidal saponins isolated from the genera Dracaena and Sansevieria. Molecules 26 (7), 1916. https://doi.org/10.3390/molecules26071916 (2021).
Yusnita Pungkastiani, W. & Hapsoro, D. In vitro organogenesis of two Sansevieria cultivars on different concentration of benzyladenine (BA). Agrivita 2011, 147–153. https://doi.org/10.17503/agrivita.v33i2.57 (2011).
Arnold, M. A. Sansevieria trifasciata. In Landscape plants for Texas and environments, Third Edition (Stipes Publishing, 2004).
Zhou, S., Ma, K., Mower, J. P., Liu, Y. & Zhou, R. Leaf variegation caused by plastome structural variation: an example from Dianella Tasmanica. Hortic. Res. 11, 526. https://doi.org/10.1093/hr/uhae009 (2024).
Sarmast, M. K., Dolati, M., Abbasabad, M., Seyfi, E. & Alizadeh, M. Appraisal of leaf cutting, soil mixture and leaf explants on production of Sansevieria trifasciata under ex/in vitro condition. Flower. Ornam. Plants. 7 (2), 261–276. https://doi.org/10.61186/flowerjournal.7.2.261 (2023).
Sarmast, M. K., Salehi, H. & Khosh-Khui, M. Seismomorphogenesis: a novel approach to acclimatization of tissue culture regenerated plants. 3 Biotech. 4, 599–604. https://doi.org/10.1007/s13205-013-0191-8 (2014).
Majumder, S., Igamberdiev, A. U. & Debnath, S. C. Somaclonal variation and clonal fidelity in commercial micropropagation: challenges and perspectives. Agronomy 15 (6), 1489. https://doi.org/10.3390/agronomy15061489 (2025).
Blazich, F. A. & Novitzky, R. T. In vitro propagation of sansevieria trifasciata. HortScience 19 (1), 122–123. https://doi.org/10.21273/HORTSCI.19.1.122 (1984).
Sarmast, M. K., Salehi, M. & Salehi, H. The potential of different parts of Sansevieria trifasciata L. leaf for meristemoids production. Aust. J. Basic Appl. Sci. 3, 2506–2509 (2009).
Wahyuningsih, T. Pengaruh Beberapa konsentrasi benziladenin (BA) Atau Kinetin Pada Pembentukan tunas adventif Sansevieria trifasciata lorentii in vitro. Skripsi Fakultas Pertanian, Universitas Lampung 26 (2006).
García-Hernández, E. et al. Indirect organogenesis for high frequency shoot regeneration of two cultivars of Sansevieria trifasciata Prain differing in fiber production. Sci. Rep. 12, 8507. https://doi.org/10.1038/s41598-022-12640-4 (2022).
Kazemzadeh Bahnamirei, M., Sarmast, M. K., Padasht Dahkaei, M. N. & Alizadeh, M. Evaluation of mother-plant growing beds, explant type, and different disinfection treatments in control of Sansevieria rhizome contamination under in vitro conditions. J. Plant. Physiol. Breed. 14 (2), 23–38 (2024).
Yang, B. M., Huang, Y. L., Xu, W. J. & Bao, L. X. Explant selection and cluster buds induction in vitro of saffron (Crocus sativus L). Agric. Sci. Technol. Commun. 2, 106–108 (2015).
McCready, R. M., Guggolz, J., Silviera, V. & Owens, H. S. Determination of starch and amylase in vegetables. Anal. Chem. 22, 1156–1158 (1950).
Satina, S., Blakeslee, A. F. & Avery, A. G. Demonstrations of the three germ layers in the shoot apex of Datura by means of induced polyploidy in periclinal chimeras. Am. J. Bot. 27, 895–905. https://doi.org/10.2307/2436558 (1940).
Shahzad, A., Ahmasd, N., Rather, M. A. & HusainMK, Anis, M. Improved shoot regeneration system through leaf derived callus and nodule culture of Sansevieria cylindrical. Biol. Plant. 53, 745–749 (2009).
Catalano, C. et al. Somatic embryogenesis and flow cytometric assessment of nuclear genetic stability for Sansevieria spp.: an approach for in vitro regeneration of ornamental plants. Horticulturae 9, 138. https://doi.org/10.3390/horticulturae9020138 (2023).
McPheeters, K. & Skirvin, R. M. Histogenic layer manipulation in chimeral ‘Thornless evergreen’ trailing blackberry. Euphytica 32, 351–360. https://doi.org/10.1007/BF00021443 (1983).
McPheeters, K. & Skirvin, R. M. Everthornless’ blackberry. HORTSCIENCE 35 (4), 778–779. https://doi.org/10.21273/HORTSCI.35.4.778 (2000).
Kudoyarova, G. R. et al. Accumulation of cytokinins in roots and their export to the shoots of durum wheat plants treated with the protonophore carbonyl cyanide m-chlorophenyl hydrazone (CCCP). J. Exp. Bot. 65, 2287–2294. https://doi.org/10.1093/jxb/eru113 (2014).
Ferreira, M. S. et al. The role of Somaclonal variation in plant genetic improvement: a systematic review. Agronomy 13 (3), 730. https://doi.org/10.3390/agronomy13030730 (2023).
Zhang, Y., Berman, A. & Shani, E. Plant hormone transport and localization: signaling molecules on the move. Ann. Rev. Plant. Biol. 74, 453–479 (2023).
Suzuki, R. M. et al. Dark -induced hormone changes coincide with the resumption of light -inhibited shoot growth in Catasetum fimbriatum (Orchidaceae). J. Plant Physiol. 167, 375–381. https://doi.org/10.1016/j.jplph.2009.10.002 (2010).
Skylar, A., Sung, F., Hong, F. X., Chory, J. & Wu, X. L. Metabolic sugar signal promotes Arabidopsis meristematic proliferation via G2. Develop Biology. 351, 82–89 (2011).
Meghwal, P. R. H. C., Sharma, A. M. & Goswami, Srivastava, K. N. Effect of stock plant etiolation on in vitro phenol exudation during culture establishment of guava (Psidium Guajava L). Indian J. Hort. 58, 328–331 (2001).
Henry, C. et al. Regulation of RhSUC2, a sucrose transporter, is correlated with the light control of bud burst in Rosa Sp. Plant. Cell. Environ. 34 (10), 1776–1789 (2011).
Moree, B. et al. Role of the Arabidopsis glucose sensor HXK1 in Nutrient, Light, and hormonal signaling. Science 300, 332–336. https://doi.org/10.1126/science.1080585 (2003).
Author information
Authors and Affiliations
Contributions
M.K.B. Performed the experiments and prepared the data. M.K.S: Conceived the research and wrote the original draft. M.A: Provide resources, review and revised the manuscript. M.N.P.D: Providing ideas and guidance to the work, revised the manuscript. All authors read and approved the final version of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bahnamirei, M.K., Sarmast, M.K., Alizadeh, M. et al. Innovative in vitro shoot regeneration in variegated snake plant (Sansevieria trifasciata cv. Laurentii) through utilization of light exposed single-node rhizome tissue. Sci Rep 15, 39505 (2025). https://doi.org/10.1038/s41598-025-23247-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-23247-w