Abstract
Liraglutide has been shown to alleviate heart failure in patients with type 2 diabetes. High-intensity interval training (HIIT) has also been proven to improve cardiac function in diabetes. The present study explored the effects and underlying mechanisms of liraglutide and HIIT combination therapy in alleviating diabetic cardiomyopathy (DCM). A high-fat diet and low-dose streptozotocin (STZ) were utilized to induce the DCM model. Eight weeks of liraglutide injection and HIIT were used to treat DCM. Subsequently, cardiac function, serum metabolic biomarkers, serum glucagon-like peptide-1 (GLP-1), histology examination, cardiac alpha-myosin heavy chain (α-MHC), and β-MHC messenger RNA (mRNA) expression, forkhead box protein O1 (FOXO1) and muscle-specific RING finger protein 1 (MURF1) mRNA expression and colocalization, and expression of GLP-1 and GLP-1 receptor (GLP-1R) proteins were detected after the intervention. Results showed that DCM rats developed hyperglycemia with eccentric hypertrophy, fibrosis, and reduced systolic and diastolic function. All interventions significantly reversed the development of heart failure by alleviating the disruption of contractile apparatus, reversed the adult α-MHC transformed to fetal β-MHC, and reduced FOXO1 and MURF1 mRNA expression. Combination therapy had a better effect in alleviating cardiac fibrosis, reducing cardiovascular risk biomarkers, controlling eccentric hypertrophy, and improving systolic function. Combination therapy significantly reduced FOXO1 and MURF1 colocalization and improved the GLP-1R sensitivity in diabetic hearts. Overall, these findings demonstrate that combination therapy can reverse cardiac failure in diabetic rats by controlling the degradation of contractile apparatus by downregulating the cardiac atrophy gene expression and interrupting their colocalization, as well as upregulating GLP-1 signaling.
Similar content being viewed by others
Introduction
Diabetic cardiomyopathy (DCM) is primarily caused by diabetes and is independent of coronary artery disease and hypertension. Heart failure is characterized by acute or chronic loss of cardiomyocytes due to ischemia injury, inflammation, lip-toxicity, and glucose-toxicity1. Heart failure with preserved ejection fraction (HFpEF) is a complex clinical syndrome characterized by clinical symptoms of heart failure, left ventricular ejection fraction (LVEF) ≥ 50%, increased natriuretic peptides, and structural heart disease (left ventricular hypertrophy or enlargement) or diastolic dysfunction2. Numerous studies have revealed that diabetes and heart failure have a bidirectional epidemiological association; diabetes directly damages the myocardium and causes heart failure without obvious clinical symptoms, which is characterized by HFpEF3. The progress of diabetes is often accompanied by abnormalities of structure changes, including enlarged left ventricular volume, cardiac hypertrophy, and fibrosis, which are accompanied by the degradation of contractile proteins4. Therefore, choosing appropriate treatment that considers both glycemic control and preservation of cardiac contractile protein is particularly important.
Glucagon-like peptide-1 (GLP-1) receptor agonists (GLP-1RAs) are a cornerstone for the treatment of type 2 diabetes and exhibit favorable effects on cardiovascular outcomes5. Liraglutide, one of the GLP-1RAs, has been widely recognized to reduce the incidence of cardiovascular mortality and preserve cardiac function in diabetic patients6. The cardiac protection function is independent of the anti-hyperglycemic effect of liraglutide7. Our previous study demonstrated that liraglutide significantly improved cardiac function by alleviating cardiac ectopic lipid deposition8. However, the effects of liraglutide on DCM from the myocardial cytoskeleton maintenance perspective remain poorly understood.
The American Heart Association supported a cause-and-effect relationship between exercise and reduced cardiovascular disease events, and exercise-based cardiac rehabilitation has been strongly recommended9. High-intensity interval training (HIIT) consists of high-intensity and active low-intensity exercises, which are used for heart rehabilitation for individuals with or without cardiovascular disease10. Vigorous exercise appears to be more effective than moderate-intensity exercise in reducing cardiovascular events, as it may induce profitable cardiac remodeling for increased cardiomyocyte volume and diameter in physically active people11. HIIT may also accelerate coronary artery calcification and myocardial fibrosis and release cardiac biomarkers and atrial fibrillation even in athletes12. Therefore, HIIT increases the risk of sudden death and myocardial infarction13. Compared with pharmacological treatment, exercise intervention has conflicting effects on cardiac function, with the results being affected by exercise intensity, duration, and mode and the targeted population14. Therefore, exploring strategies to mitigate risks during and after exercise is essential.
Both liraglutide and HIIT have a complicated interaction with cardiac function. However, the mechanisms by which HIIT and liraglutide combination affects cardiac function remain elusive. Our recent study found that exercise intervention had positive effects in alleviating DCM symptoms in rats15. The current study evaluated the effect of 8 weeks of liraglutide treatment plus HIIT on cardiac function in DCM rats and the underlying mechanisms in terms of cardiac remodeling. We hypothesized that liraglutide combined with HIIT intervention is better than HIIT intervention alone in improving cardiac function in DCM rats.
Materials and methods
Experimental design
The study protocols were approved by the Chinese Council on Animal Research and Guidelines of Animal Care and the Animal Ethical and Welfare Committee of Tianjin University of Sport and strictly followed ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines.
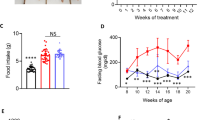
DCM was induced by a high-fat diet and low-dose streptozotocin (STZ). 90 Male Wistar rats (250–280 g, 8 weeks old) were individually housed at 20–24 °C in a 12:12-h light-dark cycle with free access to food and water. 10 rats were randomly selected to form the non-diabetic control group (CON), and 80 rats were used for the induction of the rat model of diabetes. Rats were subjected to a high-fat diet (57.5% fat, 27% carbohydrate, and 15.5% protein) combined with an intraperitoneal injection of 35 mg/kg STZ diluted with a precooled citrate buffer solution (Sigma, St. Louis, USA) for 4 weeks to induce the diabetes model. Rats in the CON group were injected with citrate buffer alone and fed with a normal diet (5% fat, 53% carbohydrate, and 23% protein). Fasting blood glucose (FBG) levels above 11.1 mmol/L for 2 consecutive readings indicated diabetes. 55 rats that tested positive for diabetes were subjected to an echocardiogram examination to assess heart function, with 40 rats reaching the level of standard DCM. DCM rats were randomly divided into four groups based on their blood glucose level: the DCM group (DCM, n = 10), DCM with liraglutide treatment group (Lira, n = 10), DCM with the HIIT intervention group (HIIT, n = 10), and DCM with liraglutide and HIIT intervention (Lira + HIIT, n = 10). Rats in the Lira and Lira + HIIT groups were treated with subcutaneous injections of liraglutide (200 µg/kg/day) for 8 weeks, whereas other groups received the same volume of normal saline. Rats in the HIIT and Lira + HIIT groups were subjected to an 8-week HIIT intervention, with those in other groups remaining secondary until sacrifice (Fig. 1).
Exercise intervention protocol
The rat exercise intervention was performed with a motorized rodent treadmill (ZH-PT, Zheng Hua Biologic Apparatus Facilities, Anhui, China). It consisted of a 10-min warm-up period (50–60% maximal oxygen consumption (VO2 max)), four interval training periods (7-min periods at 85–95% VO2 max interspersed with a 3-min interval at 50–60% VO2 max), followed by a 10-min cool-down period. This protocol was performed once a day, 5 days a week for 8 weeks16. The exercise intensity was indirectly calculated by converting the oxygen uptake and treadmill exercise intensity as previously described17 (Fig. 2).
Echocardiogram examination
Cardiac function was evaluated using the 2D-guided M-mode images coupled with a high-frequency, high-resolution digital imaging platform with linear array technology and color Doppler mode (Vevo ® 2100 Imaging System, FUJIFILM Visual Sonics, Inc., Toronto, Canada). Three consecutive heartbeats were measured, and the average of these measurements was calculated. Parameters such as left ventricular posterior wall (LVPW), interventricular septum (IVS), left ventricular ejection fraction (LVEF), fractional shortening (FS) and cardiac output (CO) were determined using the Vevo® 2100 software. In addition, the Pulse Wave Doppler mode records the peak velocity of early LV filling (E-wave) and the peak velocity of late LV filling (A-wave), allowing the calculation of the E/A ratio and isovolumic relaxation time (IVRT).
Blood sampling and biochemical analysis
Rats were anesthetized with a pelltobarbitalum (35 mg/kg) intraperitoneal injection and sacrificed. Serum samples were collected and prepared for FBG analysis through the enzymatic and colorimetric methods (Nanjing Jiancheng Bioengineering institute, Nanjing, China). Cardiac troponin T (cTnT), B-type natriuretic peptide (BNP) and GLP-1 levels were quantified using enzyme-linked immunosorbent assay (ELISA) kits (Shanghai MLBIO Biotechnology Company, Shanghai, China).
Tissue sampling and histology
Body weight was measured using a top pan balance. Hearts were harvested from sacrificed rats, drained of blood, and weighed on a fine balance. Hearts were then sectioned (4 μm) for hematoxylin and eosin (HE) and Masson trichrome staining. Heart weight/body weight (HW/BW) ratio and myocardial cell cross-sectional area (CSA) were calculated. Approximately 1.0 × 1.0 × 1.0 mm thick tissue sample from the middle part of the left ventricular was fixed with 2.5% glutaraldehyde for 24 h and then osmic acid for 1 h. Tissue sections were dehydrated by ethanol and acetone and embedded in epoxy resin and then examined under a transmission electron microscope. The remaining sections were stored in a freezer at − 80 °C for further assessment.
Immunohistochemistry
Tissue sections were stained according to the manufacturer’s instructions and incubated at 4 °C overnight with primary antibodies: forkhead box protein O1 (FOXO1) (Sericebio, Wuhan, China) and muscle-specific RING finger protein 1 (MURF1) (ABclonal, Wuhan, China) diluted with phosphate-buffered saline (PBS) to 1:200. After rinsing with PBS, sections were incubated with a horseradish peroxidase-conjugated secondary antibody (Sericebio, Wuhan, China) at room temperature for 50 min in the dark. The DNA fluorochrome 4’,6-diamidino-2-phenylindole (DAPI) was used to stain the nuclei for 10 min in the dark and examined under Fluorescent Microscopy (Olympus IX71, Tokyo, Japan). The nucleus appears blue when labeled with DAPI, FOXO1 appears red when labeled with cyanine 3 (CY3), and MURF1 green when labeled with fluorescein isothiocyanate (FITC). Processing and analysis were consistent between images and were subjected to relative fluorescence intensity analysis (calibrated with a nucleus) and colocalization analysis (Pearson’s correlation coefficient and Mander’s overlap coefficients).
Quantitative real-time PCR (qRT-PCR)
Total RNA was extracted from heart tissue sections using TRIzol reagent (Invitrogen, USA) and reverse transcribed into complementary DNA (cDNA) using a reverse transcription kit (Thermo, USA). qRT-PCR was performed using SYBR Green kits (Roche, Switzerland). The β-tubulin reaction product served as the internal control. The relative transcript levels were normalized to those of β-tubulin using the 2−△△Ct method. Primer sequences used for PCR amplification are shown in Table 1.
Western blotting
Isolated heart tissues were lysed, and protein concentrations were calculated using a BCA protein assay kit. Proteins (24 µg protein per lane) were separated with 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to a polyvinylidene difluoride (PVDF) membrane. The membrane was blocked with a protein-free blocking buffer (BlockPRO™, Taiwan, China) for 30 min. Protein expression of GLP-1 (Abcam, Cambridge, UK) and GLP-1R (Santa Cruz Biotechnology, CA, USA) were calculated as the gray-scale ratio of protein and β-tubulin (Santa Cruz Biotechnology, USA).
Statistical analysis
Data were presented as mean ± standard deviation, with a significant difference at p < 0.05. Statistical analysis was performed using SPSS 22.0 software (IBM, New York, USA). Data were transformed using the natural logarithm to obtain a normal distribution before analysis. One-way between-groups analysis of variance (ANOVA) with a Bonferroni post hoc test was used to evaluate differences between groups after the intervention.
Results
Establishment of a rat model of DCM
Rodent models have been used to study DCM. A high-fat diet with low-dose STZ is a reliable model for studying type 2 diabetes mellitus, and STZ-induced cardiac dysfunction provided a viable model for DCM18. As shown in Table 2, FBG was significantly increased in DCM rats compared with the CON group (p < 0.01), which indicated the induction of diabetes models. DCM rats exhibited a markedly reduced E/A ratio (the ratio of peak early (E-wave) and late (A-wave) left ventricular filling velocity) and prolonged isovolumic relaxation time (IVRT) (p < 0.05) and slightly reduced LVEF and fractional shortening (FS) (p < 0.05), with normalized cardiac output (CO) (p > 0.05), indicating that DCM rats suffered impaired diastolic function, and the DCM model was successfully established.
Cardiac function parameters
Subsequently, cardiac hemodynamics and cardiac contractility were assessed to further explore whether cardiac eccentric hypertrophy and cardiac fibrosis can lead to cardiac contraction dysfunction (Fig. 3). Compared with the CON group, DCM rats showed impaired diastolic function (elevated E/A ratio, p < 0.01), reduced systolic functions (reduced LVEF and FS, p < 0.01), and cardiac cavity enlargement (reduced left ventricular posterior wall (LVPW) and interventricular septum (IVS), p < 0.01). After 8 weeks of HIIT and combination therapy, the E/A ratio significantly reduced with increased LVEF, FS, and IVS (p < 0.01). Liraglutide treatment reduced the E/A ratio and improved LVEF (p < 0.01), with insignificant evaluation of FS (p > 0.05). IVRT levels were not significantly different among all groups (p > 0.05).
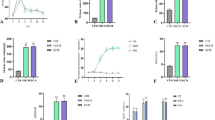
Effects of Liragutide and/or HIIT on cardiac diastolic and systolic function. Data were expressed as the mean ± SD. (A,B) E/A ratio > 1.5 and prolonged IVRT means impaired diastolic dysfunction. (C,D) Decreased LVEF and FS means impaired systolic dysfunction. (E,F) Reduced LVPW and IVS means cardiac eccentric hypertrophy. **p < 0.01 compared with the CON group, ##p < 0.01 compared with the DCM group, &p < 0.01 compared with the Lira + HIIT group. E/A ratio the ratio of peak velocity of early LV filling (E-wave), and the peak velocity of late LV filling (A-wave), IVRT isovolumic relaxation time, LVEF left ventricular ejection fraction, FS fractional shortening, LVPW left ventricular posterior wall, IVS interventricular septum.
Cardiac eccentric hypertrophy
Cardiac morphology was assessed to determine cardiac structural changes. The HW/BW ratio was calculated to measure the extent of cardiac hypertrophy, whereas HW/BW and CSA combination was used to identify eccentric hypertrophy. DCM hearts exhibited an increased HW/BW ratio accompanied by elongation of cardiomyocyte and enlarged ventricular cavity (p < 0.01), rather than increased CSA (p > 0.05), compared with the control group (Fig. 4). Only combination therapy significantly reduced the HW/BW ratio after 8 weeks of treatment (p < 0.05), with only HIIT intervention increasing the CSA compared with other groups (p < 0.05). However, liraglutide treatment showed no statistical differences in controlling cardiac eccentric hypertrophy (p > 0.05). Collectively, these findings suggest that combination therapy has a better effect than single intervention in controlling cardiac hypertrophy.
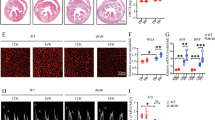
Liraglutide and/or HIIT alleviated eccentric cardiac hypertrophy. (A) Myocardial HE staining of different groups of rats (×50 magnification). Hearts in the CON group had normal structure; DCM rats exhibited loose structure, large ventricular cavity; hearts after Liraglutide and/or HIIT intervention, indicating closely arranged myocardial cells, similar to the normal heart. (B) HW/BW ratio of heart weight/body weight. (C) CSA cross sectional area. **p < 0.01 compared with the CON group; #p < 0.05 compared with the DCM group; ##p < 0.01 compared with the DCM group, ψp < 0.01 compared with the HIIT group.
Cardiac remodeling
The sarcomere is the basic unit of muscle contraction and an important node of mechanical force transmission. Therefore, we sought to determine whether the change in cardiac function could be the result of defects in the structure of the cardiomyocyte contractile apparatus. Transmission electron microscopy revealed that cardiomyocytes displayed a mild disruption in the M-line and the Z-disc of the sarcomere in the DCM group, which exhibited myofilament permutation disorder, lipid accumulation, and disruption of the M-line. However, liraglutide and HIIT treatment alleviated myofilament permutation and unformal mitochondria structures (Fig. 5A).
Cardiac fibrosis is one of the hallmarks of heart failure, accompanied by collagen accumulation. As shown in Fig. 5B, marked interstitial fibrosis was observed in DCM rats. The 8-week liraglutide and/or HIIT intervention significantly reduced collagen depositions and alleviated myocardial fibrosis, demonstrated by a reduced fibrosis area percentage (p < 0.05). Liraglutide had a better effect than HIIT intervention in reducing fibrosis area percentage (p < 0.05).
Liraglutide and/or HIIT alleviated cardiac remodeling in DCM rats. (A) Upper A panel: Electron microscope images showing the myocardial ultrastructure in different groups of rats (×20,000 magnification), arrows indicate M-line and Z-disc. Lower A panel: Tissue sections were stained with Masson staining (×200 magnification). (B) Quantification of cardiac fibrosis. Data were expressed as the mean ± SD. **p < 0.01 compared with the CON group; ##p < 0.01 compared with the DCM group; Ɨp < 0.05 compared with the Lira group.
Cardiac atrophy
The sarcomere is mainly composed of thick and thin muscle filaments. Heart failure exhibits a transition between two isoforms of the alpha-myosin heavy chain (α-MHC) and β-MHC19. As fetal genes, re-expression of α-MHC and β-MHC messenger RNA (mRNA) is also related to cardiac hypertrophy. Our data revealed a declining trend in α-MHC mRNA levels (p > 0.05), and β-MHC mRNA increased three-fold in DCM rats (p < 0.01) (Fig. 6A, B). Liraglutide and/or HIIT intervention significantly reversed abnormal expression of α-MHC and β-MHC mRNAs (p < 0.01). Notably, the HIIT intervention and combination therapy dramatically increased α-MHC mRNA expression compared with the control group (p < 0.01).
The effects of HIIT on cardiac remodeling. (A) α-MHC mRNA relative expression. (B) β-MHC mRNA relative expression. (C) MURF1 mRNA relative expression. (D) FOXO1 mRNA relative expression. Data were expressed as the mean ± SD. **p < 0.01 as compared with CON group; ##p < 0.01 compared with the DCM group. α-MHC Myosin heavy chain-α, β-MHC Myosin heavy chain-β, MURF1 Muscle Ring Finger1, FOXO1 Forkhead Box Protein1.
Furthermore, the sarcomere structure and function were examined to test the contractility under eccentric hypertrophy in the aforementioned DCM situation. MURF1 is implicated in the regulation of cardiac myocyte size and contractility, which has been proven to be localized to the M-line and Z-disc of the sarcomere20. Therefore, we determine whether the MURF1 mRNA expression leads to the disruption of sarcomere function and subsequent heart failure. As shown in Fig. 6C, MURF1 mRNA levels were significantly increased in the DCM group compared with the control group but significantly reduced following liraglutide and/or HIIT intervention (p < 0.01). FOXO1 plays an important role in muscle atrophy in mammals. It acts as a transcription factor on the promoters for MURF1 to stimulate their expression21. As shown in Fig. 6D, DCM rats exhibited increased FOXO1 expression, which was reduced after liraglutide and/or HIIT treatment (p < 0.01).
Colocalization of FOXO1 and MURF1
An immune-fluorescence double-labeled image collected by laser confocal microscope was analyzed to further determine the relationship between FOXO1 and MURF1 (Fig. 7). Red, green, and yellow fluorescence signals depict positive expression of FOXO1, positive expression of MURF1, and colocalization of FOXO1 and MURF1, respectively. DCM rats showed significantly elevated FOXO1 (p < 0.01) and MURF1 relative intensity with more overlap (p < 0.01) between the two proteins than the CON group, indicating that FOXO1 colocalizes with MURF1, promoting myocardial cell atrophy. Although liraglutide and HITT intervention reduced FOXO1 (p < 0.01) and MURF1 (p > 0.05) relative intensity, respectively, the colocalization relationship between them still existed. Combination therapy had a better effect in reducing both FOXO1 and MURF1 relative intensity as well as colocalization ratio compared with the DCM group (p < 0.01).
Combination therapy prevented the colocalization of FOXO1 and MURF1 in diabetic hearts. (A) Immunofluorescence staining of DAPI, FOXO1 and MURF1 (×200 magnification) with co-localization scatter plot within groups. (B) The relative FOXO1 and MURF1 fluorescence intensity normalized to the DAPI fluorescence. (C) The co-localization parameters within groups. Analysis of fluorescence intensity histogram showed that pixels were localized along the y = x line, which indicated an overlap in scatter gram. Pearson’s correlation coefficient and Mander’s colocalization coefficients were utilized to indicate colocation relationship. **p < 0.01 compared with the CON group; ##p < 0.01 compared with the DCM group. Ɨp < 0.01 compared with the HIIT group, ψp < 0.05 compared with the Lira + HIIT group.
Serum biomarkers
Serum myocardial injury biomarkers have been widely utilized for accurate and rapid diagnosis of cardiac disease. Serum cardiac troponin T (cTnT) and brain natriuretic peptide (BNP) levels in DCM rats were to evaluate the safety of HIIT intervention (Fig. 8). As an effective serum biomarker of heart failure, serum cTnT levels increased remarkably in the DCM group compared with the CON group but were significantly decreased after liraglutide and/or HIIT intervention (p < 0.05). As an indicator of myocardial hypertrophy, BNP levels of BNP were elevated in DCM rats (p < 0.01) but significantly reduced after 8 weeks of treatment with Lira and combination therapy (p < 0.01). Although BNP levels showed a decreasing trend in the HIIT group, results did not reach statistical significance.
GLP-1, a gastrointestinal hormone primarily secreted by L cells in the intestine in response to food intake, has a protective effect on DCM. Serum GLP-1 levels decreased remarkably in the DCM group compared with the CON group. After the 8-week HIIT intervention, serum GLP-1 levels increased significantly following liraglutide and combination therapy (p < 0.01). Serum GLP-1 levels were significantly lower in the HIIT group than in the Lira group (p < 0.05) and had an insignificant decline compared with the DCM group (p > 0.05).
The concentration of serum biomarkers of myocardial injury levels in the indicated groups. (A) Serum BNP levels. (B) Serum cTNT levels. (C) Serum GLP-1 level. Data are expressed as the mean ± SD. **p < 0.01 compared with CON group; #p < 0.05 compared with the DCM group; ##p < 0.01 compared with the DCM group. BNP brain natriuretic peptide, cTNT cardiac troponin.
GLP-1 signaling
Intriguingly, GLP-1 exhibits beneficial pleiotropic effects on diabetic heart by binding to a specific receptor (GLP-1R). Thus, cardiac GLP-1 and GLP-1R protein expressions were assessed to determine whether liraglutide and HIIT have different mechanisms (Fig. 9). DCM rats had impaired GLP-1 secretion and function, demonstrated by reduced cardiac GLP-1 and GLP-1R protein expression (p < 0.01).
Cardiac GLP-1 relative expressions were improved to a normal level compared with the DCM group after 8 weeks of intervention (p < 0.05). A similar pattern was observed in Lar and/or HIIT groups (p > 0.05). There was a significant improvement in cardiac GLP-1R protein expression among treatment groups compared with the DCM group (p < 0.01). Meanwhile, GLP-1R protein expression was significantly increased in HIIT and combination therapy groups compared with the Lira group (p < 0.05).
Cardiac GLP-1 and GLP-1R proteins in the indicated groups. Data are expressed as the mean ± SD. *p < 0.05 as compared with the CON group; **p < 0.01 compared with the CON group; #p < 0.05 compared with the DCM group; ##p < 0.01 compared with the DCM group; Ɨp < 0.05 compared with the Lira group; ƗƗp < 0.01 compared with the Lira group.
Discussion
The present study investigated the effect of 8 weeks of liraglutide plus HIIT in DCM rats. It was found that HFpEF hearts in STZ-induced type 2 diabetes mellitus models exhibited significant biochemical (higher serum FBG, cTnT, and BNP levels and lower serum GLP-1 levels), structural (left ventricular enlargement, wall thinning, disruption of the M-line and Z disc), and functional (reduced EF and FS) changes, as well as cardiac fibrosis and conversion of α-MHC and β-MHC mRNA expression. Based on the established rat model, it was found that combination therapy was superior to single intervention in improving cardiac function, alleviating cardiac eccentric hypertrophy and fibrosis by increasing the cardiac GLP-1R sensitivity, and inhibiting FOXO1 and MURF1 colocalization.
Our results showed that DCM model exhibits key features of HFpEF22. Cardiac concentric hypertrophy is a compensatory condition, characterized by increased heart mass in the primary period of chronic heart failure, which is attributed to a corresponding increase in CSA and wall thickness23. Echocardiogram results revealed a decreased E/A ratio, prolonged IVRT, and normalized CO, confirming that the heart was in a pathological hypertrophy state at the early phase of DCM. The heart initiated a hypertrophic response and increased myocardial mass to adapt to mechanical and hemodynamic stimulation, thus causing pathological myocardial hypertrophy, which is an important stage in the development of heart failure. This resulted in a reduced number of myocardial cells, altered cardiac structure and cytoskeleton, collagen deposition, ventricular cavity enlargement, and an increased HW/BW ratio accompanied by cardiomyocyte elongation, rather than increased CSA, ultimately leading to a dilated heart at the end stage of DCM. After 8 weeks of intervention, HIIT and combination therapy controlled eccentric hypertrophy, thereby reversing pathological hypertrophy to physiological hypertrophy. While liraglutide showed no significant improvement in alleviating eccentric hypertrophy, it compacted myocardial fiber arrangement, suggesting that liraglutide may optimize myocardial structure in DCM rats and significantly reduce BNP and cTnT levels. However, HIIT had a limited effect in reducing BNP levels.
Changes in the cardiac structure affect cardiac function by remodeling the extracellular matrix of the heart. Research on myocardial infarction (MI) in rats has shown that extracardiac matrix fibrosis and collagen accumulation directly influence early diastolic ventricular aspiration; therefore, myocardial fibrosis caused by abnormal proliferation of myocardial collagen is one of the main pathological features of heart disease24. Cardiac fibrosis is injurious to the heart, a 2–3 times increase in myocardial collagen content can alter ventricular filling and increase diastolic stiffness, and a more than 4 times increase directly influences the contraction function. In the present study, Masson trichrome staining exhibited excessive collagen accumulation in cardiomyocytes, which was effectively reduced by liraglutide and/or HIIT intervention. A variant dose of liraglutide had a positive effect on the clearance of collagen deposition25,26. Our findings also showed that administration of 200 µg/kg of liraglutide decreased the deposition of collagen fibers in the heart of DCM rats. Castellar et al. reported that exercise training reduced collagen accumulation in the myocardium of diabetic rats27. Four-week treadmill running significantly reduces cardiac types I and III collagen fiber, suggesting that HIIT may significantly control myocardial fibrosis and protect a diabetic heart28. However, Franklin et al. argued that HIIT increased the potential for exercise-related acute cardiac events, partly resulting from fibrosis12. The current study found that although HIIT intervention alleviated collagen accumulation in DCM hearts, the collagen clearance effect was inferior to liraglutide treatment; combination therapy might reduce the incidence of cardiovascular events during high-intensity exercise.
Diastolic dysfunction is the most prominent feature of DCM, which eventually develops into systolic dysfunction due to prolonged damage29. Heart failure occurs due to the primary or secondary weakening of myocardial contractile function, which is related to the structure of the sarcomere. MURF1, also known as tripartite motif containing 63 (Trim63), is a key E3 ubiquitin ligase particularly expressed in cardiac and skeletal muscle, which localizes to the Z-desc and M-line of the sarcomere by binding to myomectomy of sarcomere thick myofilaments to regulate the stability of the myofibril M-band structure30. Our data showed that MURF1 mRNA expression was upregulated in DCM rats, thereby leading to disruption of sarcomere function and subsequent heart failure, which may be due to the re-expression of certain fetal genes31. During the remolding, fetal gene profiles are accompanied by the degradation of structural proteins and transcription factors32. Transformation of adult α-MHC to fetal β-MHC gene is a hallmark of heart disease, which is accompanied by cardiac dysfunction, as α-MHC has a higher ATPase activity than β subunits; therefore, β-MHC based myocardial contractility is weak, leading to slowed myocardial contraction and systolic dysfunction33. Rundell and colleagues found that STZ-induced diabetic rat models exhibited a higher expression level of β-MHC and a lower expression level of α-MHC, resulting in reduced myocardial fiber tension and decreased contraction activity34, consistent with the findings of the present study. We found that changes in myoglobin heavy chain subtypes affected normal excitation-contraction coupling and corresponded to echocardiography results in terms of reduced LVEF and FS. Moderate exercise improves cardiac contractile function by preventing a shift to β-MHC and reducing MURF1 expression in MI rats35. A previous study found that HIIT treatment lowered β-MHC expression, reduced myocardial fibrosis and apoptosis, and significantly increased systolic function in C57BL/6J mice with diabetic MI36, which aligns with our findings that HIIT treatment significantly suppressed MURF1 gene expression, inhibiting fetal gene expression. Although without direct evidence that liraglutide works on cardiac MURF1 expression, Wang et al. noted that GLP-1 analogs exendin changed myosin heavy chain type composition and enhance insulin sensitivity37. The present study found that liraglutide alone or in combination with HIIT reduced MURF1 and converted fetal gene expression, which may explain the correlation between eccentric hypertrophy and contraction dysfunction in DCM rats.
FOXO family can regulate energy metabolism, cell proliferation, apoptosis, and tumor suppression38. Our previous studies also revealed that FOXO1 overexpression in the diabetic heart participates in abnormal myocardial metabolism15. Several studies have indicated that FOXO1 acts on MURF1 promoters to stimulate its expression in the conditions of skeletal muscle atrophy39,40. The current study found that FOXO1 mRNA expression was increased in DCM rats; FOXO1 enters the nucleus and upregulates MURF1 expression, promoting ubiquitin-proteasome pathway-mediated cardiac atrophy. An 8-week treatment with liraglutide and/or HIIT significantly reduced FOXO1 and MURF1 mRNA expression and slowed cardiac muscle wasting. FOXO1 and MURF1 colocalization was assessed to further determine the relationship between atrophy protein and diabetes. Pearson’s correlation coefficient is proven to be an effective measure of the degree of overlap when the proportion of two fluorescence is similar. However, our study found that liraglutide treatment had a higher proportion of MURF1 fluorescence, whereas HIIT intervention had a higher proportion of FOXO1 fluorescence. The Mander’s overlap coefficient is a more intuitive measurement of colocalization as it may display the overlapping proportion between FOXO1 and MURF1. In our study, only combination therapy was effective in reducing colocalization of FOXO1 and MURF1 in both methods, which may partly explain the better effect of combination therapy than single intervention.
GLP-1RAs are an essential treatment in type 2 diabetes. The extra-pancreatic effect of GLP-1, especially in cardiac protection, has garnered considerable attention. It has been demonstrated that GLP-1 may improve the local and global heart function of patients with MI and left ventricular systolic dysfunction on successful implementation of direct angioplasty41. Diabetic patients had diminished GLP-1 effect, suggesting altered GLP-1 secretion and function is associated with pathophysiology42. The insulin secretion-promoting and anti-diabetic functions of GLP-1 are achieved through G-protein-coupled receptor GLP-1R, widely expressed in kidney, lung, heart, brain, and islet cells43. An in vitro study has shown that GLP-1R-dependent signaling may protect the heart from oxidative stress and apoptosis through exchange factor directly activated by cAMP (Epac)-signaling44. The present study found that the GLP-1 effect was severely diminished, as demonstrated by GLP-1 resistance in DCM hearts.
Liraglutide can increase GLP-1R expression in the myocardium of diabetic rats, reverse ventricular hypertrophy, and protect the diabetic heart after treadmill running45. Although the impact of exercise on GLP-1 remains elusive, Burmeister and coworkers found that GLP-1R−/− mice had hyperglycemia after treadmill running compared with the wild control46. Farzanegi et al. reported that 8 weeks of aerobic exercise significantly increased cardiac GLP-1 and GLP-1R expression in diabetic rats47. Kullman and colleagues noted that exercise improved GLP-1 resistance rather than increased GLP-1 secretion in nonalcoholic fatty liver disease48. A different study found that GLP-1 levels increased immediately after HIIT and continued to rise 2.6 h after HIIT, indicating that exercise may stimulate GLP-1 secretion by L cells in the small intestines and promote increased release of endogenous GLP-149, consistent with the results of our study, which indicated that the serum GLP-1 and cardiac GLP-1 protein expression were significantly elevated after 8 weeks of HIIT and combination therapy similar to liraglutide treatment results. However, cardiac GLP-1R expression was significantly higher in HIIT and combination therapy groups than in the liraglutide treatment group, implying that exercise may ameliorate GLP-1R resistance.
Conclusions
In summary, this study is the first to demonstrate the cardioprotective effect of liraglutide and HIIT combination, which alleviated eccentric cardiac hypertrophy and improved cardiac function, at least in part, by inhibiting cardiac atrophy. While liraglutide and HIIT have independent heart-protecting effects, their synergistic effect is superior. The combined therapy counteracts liraglutide or HIIT limitations, reduces the stress effect of exercise on the heart, limits FOXO1 and MURF1 colocalization, and improves myocardial GLP-1R sensitivity.
Data availability
Data is provided within the manuscript.
Abbreviations
- BNP:
-
B-type natriuretic peptide
- CO:
-
Cardiac output
- cTnT:
-
Cardiac troponin T
- CSA:
-
Cross-sectional area
- DAPI:
-
The DNA fluorochrome 4’,6-diamidino-2-phenylindole
- DCM:
-
Diabetic cardiomyopathy
- E/A ratio:
-
The ratio of peak velocity of early LV filling and the peak velocity of late LV filling
- FBG:
-
Fasting blood glucose
- FINs:
-
Serum insulin level
- FOXO1:
-
Forkhead box protein O1
- FS:
-
Fractional shortening
- GLP-1:
-
Glucagon like peptide-1
- GLP-1R:
-
Glucagon like peptide-1 receptor
- GLP-1RA:
-
Glucagon like peptide-1 receptor agonists
- HFpEF:
-
Heart failure with preserved ejection fraction
- HIIT:
-
High intensity interval training
- HW/BW:
-
Heart weight/body weight
- IVRT:
-
Isovolumic relaxation time
- IVS:
-
Interventricular septum
- LVEF:
-
Left ventricular ejection fraction
- LVPW:
-
Left ventricular posterior wall
- MHC:
-
Myosin heavy chain
- MURF1:
-
Muscle-specific RING-finger protein 1
- STZ:
-
Streptozotocin
References
Huang, J. P. et al. Exosomal microRNAs miR-30d-5p and miR-126a-5p are associated with heart failure with preserved ejection fraction in STZ-induced type 1 diabetic rats. Int. J. Mol. Sci. 23 (14), 7514. https://doi.org/10.3390/ijms23147514s (2022).
Lejeune, S. et al. Diabetic phenotype and prognosis of patients with heart failure and preserved ejection fraction in a real-life cohort. Cardiovasc. Diabetol. 20 (1), 48. https://doi.org/10.1186/s12933-021-01242-5 (2021).
Jia, G. et al. Diabetic cardiomyopathy: an update of mechanisms contributing to this clinical entity. Circ. Res. 122 (4), 624–638. https://doi.org/10.1161/CIRCRESAHA.117.311586 (2018).
Heinzel, F. R. et al. Myocardial hypertrophy and its role in heart failure with preserved ejection fraction. J. Appl. Physiol. 119 (10), 1233–1242. https://doi.org/10.1152/japplphysiol.00374.2015 (2015).
Yamada, S. et al. Effect of GLP-1 receptor agonist, liraglutide, on muscle in spontaneously diabetic torii fatty rats. Mol. Cell. Endocrinol. 539, 111472. https://doi.org/10.1016/j.mce.2021.111472 (2022).
Marso, S. P. et al. LEADER trial investigators. Effects of Liraglutide on cardiovascular outcomes in patients with diabetes with or without heart failure. J. Am. Coll. Cardiol. 75 (10), 1128–1141. https://doi.org/10.1016/j.jacc.2019.12.063 (2020).
Di, I. L. et al. Glucagon-like peptide-1 receptor agonist and the relation between metabolic effects and cardiovascular outcomes: insight into mechanisms of action. Eur. Heart J. 42 (Supplement_1). https://doi.org/10.1093/eurheartj/ehab724.2956 (2021).
Cai, H. et al. Beneficial effects and mechanisms of Liraglutide on improving cardiac ectopic lipid deposition and protect cardiac function in diabetic cardiomyopathy rats. Chin. Circ. J. 34 (10), 1–9 (2019).
Thompson, P. D. et al. Exercise and acute cardiovascular events placing the risks into perspective: a scientific statement from the American Heart Association Council on Nutrition, Physical Activity, and metabolism and the Council on Clinical Cardiology. Circulation 115 (17), 2358–2368. https://doi.org/10.1161/CIRCULATIONAHA.107.181485 (2007).
Mcgregor, G. et al. High-intensity interval training versus moderate-intensity steady-state training in Uk cardiac rehabilitation programmes (HIIT or MISS UK): study protocol for a multicenter randomized controlled trial and economic evaluation. BMJ Open 6 (11), e012843. https://doi.org/10.1136/bmjopen-2016-012843 (2016).
Li, Y. et al. Impact of healthy lifestyle factors on life expectancies in the US population. Circulation 138 (4), 345–355. https://doi.org/10.1161/CIRCULATIONAHA.117.032047 (2018).
Franklin, B. A. et al. Exercise-related acute cardiovascular events and potential deleterious adaptations following long-term exercise training: placing the risks into perspective-an update: a scientific statement from the American Heart Association. Circulation 141 (13), e705–e736. https://doi.org/10.1161/CIR.0000000000000749 (2020).
Squires, R. W. et al. Progression of exercise training in early outpatient cardiac rehabilitation: an official statement from the American Association of cardiovascular and pulmonary rehabilitation. J. Cardiopulm. Rehabil. Prev. 38, 139–146 (2018).
Aengevaeren, V. L. et al. Endurance exercise-induced changes in BNP concentrations in cardiovascular patients versus healthy controls. Int. J. Cardiol. 227, 430–435. https://doi.org/10.1016/j.ijca (2017).
Cai, H. et al. An attempt to reverse cardiac lipotoxicity by aerobic interval training in a high-fat diet- and streptozotocin-induced type 2 diabetes rat model. Diabetol. Metab. Syndr. 11, 43. https://doi.org/10.1186/s13098-019-0436-8 (2019).
Yue, X. et al. FSTL1 as a potential mediator of exercise induced cardioprotection in post-myocardial infarction rats. Sci. Rep. 6, 32424. https://doi.org/10.1038/srep32424 (2016).
Bedford, T. G. et al. Maximal oxygen consumption of rats and its changes with various experimental procedures. J. Appl. Physiol. 47 (6), 1278–1283. https://doi.org/10.1152/jappl.1979.47.6.1278 (1979).
Bugger, H. & Abel, E. D. Rodent models of diabetic cardiomyopathy. Dis. Model. Mech. 2 (9–10), 454–466. https://doi.org/10.1242/dmm.001941 (2009).
Narolska, N. A. et al. Myosin heavy chain composition and the economy of contraction in healthy and diseased human myocardium. J. Muscle Res. Cell. Motil. 26 (1), 39–48 (2005).
Centner, T. et al. Identification of muscle specific ring finger proteins as potential regulators of the titin kinase domain. J. Mol. Biol. 306 (4), 717–726. https://doi.org/10.1006/jmbi.2001.4448 (2001).
Chen, B. L. et al. Activation of AMPK inhibits cardiomyocyte hypertrophy by modulating of the FOXO1/MuRF1 signaling pathway in vitro. Acta Pharmacol. Sin. 31 (7), 798–804. https://doi.org/10.1038/aps.2010.73 (2010).
Sharp, T. E. et al. Reply: tolerating large preclinical models of HFpEF but without the intolerance? JACC Basic Transl. Sci. 6 (4), 397–399. https://doi.org/10.1016/j.jacbts.2021.03.004 (2021).
Grossman, W. et al. Wall stress and patterns of hypertrophy in the human left ventricular. J. Clin. Investig. 56 (1), 56–64 (1975).
Yu, Y. et al. Kinetic alterations of collagen and elastic fibres and their association with cardiac function in acute myocardial infarction. Mol. Med. Rep. 17 (3), 3519–3526. https://doi.org/10.3892/mmr.2017.8347 (2018).
Chen, P. et al. Liraglutide attenuates myocardial fibrosis via inhibition of AT1R-mediated ROS production in hypertensive mice. J. Cardiovasc. Pharmacol. Ther. 26 (2), 179–188. https://doi.org/10.1177/1074248420942007 (2021).
Huang, D. D. et al. Liraglutide improves myocardial fibrosis after myocardial infarction through inhibition of CTGF by activating cAMP in mice. Eur. Rev. Med. Pharmacol. 22 (14), 4648–4656 (2016).
Castellar, A. et al. Collagen and reticular fibers in left ventricular muscle in diabetic rats: physical exercise prevents its changes? Tissue Cel 43 (1), 24–28 (2011).
Silva, F. S. et al. Exercise training ameliorates matrix metalloproteinases 2 and 9 messenger RNA expression and mitigates adverse left ventricular remodeling in streptozotocin-induced diabetic rats. Cardiovasc. Pathol. 29, 37–44. https://doi.org/10.1016/j.carpath.2017.05.003 (2017).
Grigorescu, E. D. et al. Left ventricular diastolic dysfunction in type 2 diabetes-progress and perspectives. Diagnostics (Basel) 9 (3), 121. https://doi.org/10.3390/diagnostics9030121 (2019).
Lindqvist, J. et al. Removal of MuRF1 increases muscle mass in nemaline myopathy models, but does not provide functional benefits. Int. J. Mol. Sci. 23 (15), 8113. https://doi.org/10.3390/ijms23158113 (2022).
Kedar, V. et al. Muscle-specific RING finger 1 is a bona fide ubiquitin ligase that degrades cardiac troponin I. Proc. Natl. Acad. Sci. U.S.A. 101 (52), 18135–18140. https://doi.org/10.1073/pnas.0404341102 (2004).
Adams, V. et al. Myocardial expression of Murf-1 and MAFbx after induction of chronic heart failure: Effect on myocardial contractility. Cardiovasc. Res. 73 (1), 120–129. https://doi.org/10.1016/j.cardiores.2006.10.026 (2007).
Wan, W. et al. Exercise training induced myosin heavy chain isoform alteration in the infarcted heart. Appl. Physiol. Nutr. Metab. 39 (2), 226–232. https://doi.org/10.1139/apnm-2013-0268 (2004).
Rundell, V. et al. Depressed cardiac tension cost in experimental diabetes is due to altered myosin heavy chain isoform expression. Am. J. Physiol. Heart Circ. Physiol. 287 (1), H408–H413. https://doi.org/10.1152/ajpheart.00049.2004 (2004).
Adams, V. et al. Modulation of Murf-1 and MAFbx expression in the myocardium by physical exercise training. Eur. J. Cardiovasc. Prev. Rehabil. 15 (3), 293–299. https://doi.org/10.1097/HJR.0b013e3282f3ec43 (2008).
Li, L. et al. Aerobic interval training improves cardiac function recovery after ischemia in obese mice. Chin. Heart J. 29 (3), 281–285. https://doi.org/10.13191/j.chj.2017.0072 (2017).
Wang, L. et al. Divergent effects of GLP-1 analogs exendin-4 and exendin-9 on the expression of myosin heavy chain isoforms in C2C12 myotubes. Peptides 32 (6), 1313–1319. https://doi.org/10.1016/j.peptides.2011.03.018 (2011).
Behl, T. et al. Exploring the genetic conception of obesity via the dual role of FoxO. Int. J. Mol. Sci. 22 (6), 3179. https://doi.org/10.3390/ijms22063179 (2021).
Reed, S. A. et al. Inhibition of FoxO transcriptional activity prevents muscle fiber atrophy during cachexia and induces hypertrophy. FASEB J. 26 (3), 987–1000 (2012).
Liu, Y. et al. Aspartate inhibits LPS-induced MAFbx and MuRF1 expression in skeletal muscle in weaned pigs by regulating akt, AMPKα and FOXO1. Innate Immun. 23 (1), 34–43. https://doi.org/10.1177/1753425916673443 (2017).
Nguyen, T. D. et al. GLP-1 improves diastolic function and survival in heart failure with preserved ejection fraction. J. Cardiovasc. Transl. Res. 11 (3), 259–267. https://doi.org/10.1007/s12265-018-9795-z (2018).
Arturi, F. et al. Liraglutide improves cardiac function in patients with type 2 diabetes and chronic heart failure. Endocrine 57 (3), 464–473. https://doi.org/10.1007/s12020-016-1166-4 (2017).
Tahrani, A. A. et al. Once-weekly GLP-1R agonists: moving the goal posts. Lancet Diabetes Endocrinol. 6 (4), 260–261. https://doi.org/10.1016/S2213-8587(18)30049-4 (2018).
Mangmool, S. et al. Epac is required for GLP-1R-mediated inhibition of oxidative stress and apoptosis in cardiomyocytes. Mol. Endocrinol. 29 (4), 583–596 (2015).
Romaní-Pérez, M. et al. Activation of the GLP-1 receptor by liraglutide increases ACE2 expression, reversing right ventricle hypertrophy, and improving the production of SP-A and SP-B in the lungs of type 1 diabetes rats. Endocrinology 156 (10), 3559–3569. https://doi.org/10.1210/en.2014-1685 (2015).
Burmeister, M. A. et al. Regulation of glucose kinetics during exercise by the glucagon-like peptide 1 receptor. J. Physiol. 590 (20), 5245–5255 (2012).
Farzanegi, P. The effects of aerobic training and arbutin on GLP-1 and GLP-1R in diabetes rats. Indian J. Fun Appl. Life Sci. 4 (4), 356–362 (2014).
Kullman, E. L. et al. Short-term aerobic exercise training improves gut peptide regulation in nonalcoholic fatty liver disease. J. Appl. Physiol. 120, 1159–1164 (2016).
Chanoine, J. P. et al. GLP-1 and appetite responses to a meal in lean and overweight adolescents following exercise. Obesity (Silver Spring) 16 (1), 202–204. https://doi.org/10.1038/oby.2007.39 (2008).
Funding
The work presented in this article was supported by grants from the Tianjin Education Commission Research Program Project (No. 2022KJ002) and Key Research and Development Program Projects in Hebei Province (No. 22377713D).
Author information
Authors and Affiliations
Contributions
Huan Cai researched data and wrote the manuscript. Jingqin Liu contributed to data analysis and discussion. Chengye Dai conducted animal experiments. Shuchun Chen reviewed and edited the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All the experimental procedures were performed in strict accordance with the Guidelines on the Care and Use of Laboratory Animals issued by the Chinese Council on Animal Research and Guidelines of Animal Care. Furthermore, the Animal Ethical and Welfare Committee of Tianjin University of Sport approved these experiments.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cai, H., Dai, C., Liu, J. et al. Liraglutide combined with HIIT preserves contractile apparatus and blunts the progression of heart failure in diabetic cardiomyopathy rats. Sci Rep 15, 5051 (2025). https://doi.org/10.1038/s41598-025-85699-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-85699-4